A subscription to JoVE is required to view this content. Sign in or start your free trial.
Analysis of Efferocytosis of Apoptotic Thymocytes by Peritoneal Macrophages
In This Article
Overview
This video demonstrates a procedure for analyzing peritoneal macrophage-mediated efferocytosis of apoptotic mouse thymocytes by fluorescence microscopy.
Protocol
All procedures involving animal models have been reviewed by the local institutional animal care committee and the JoVE veterinary review board.
1. Preparation of CFSE-labeled apoptotic thymocytes
- Euthanize two naïve C57/B6 mice by CO2 inhalation for 10 min and dissect to open the chest cavity, remove (pull out) the thymus with curved fine-tip forceps into tissue culture petri dish containing 10 mL of RPMI1640 medium.
- Obtain single-cell suspension by grinding the whole thymus against two frosted ends of the microscope slides and then filter the suspension through a 100 μm cell strainer.
- Collect the 10 mL thymus suspension into a 50 mL tube and centrifuge at 300 x g for 5 min.
- Remove the supernatant, resuspend in 40 mL of 1x PBS, and count cell numbers with a hemocytometer.
- Centrifuge at 300 x g for 5 min and remove the supernatant.
- Resuspend in 20 mL of 1x PBS in a 50 mL tube as a single cell suspension with up to 2 x 108 cells (if more than 2 x 108 cells, resuspend the rest of the cells in another 20 mL of 1 x PBS in a different 50 mL tube).
- Make 5 μM of CFSE in equal volume (20 mL) of 1x PBS in a separate 50 mL tube by pipetting 40 μL of CFSE stock solution (2.5 mM) into 20 mL of 1x PBS and mix well by inverting the tube 2 - 3 times.
- Add the 20 mL of CFSE from step 1.7 into the 20 mL of cell suspension from step 1.6 (the final concentration of CFSE is now 2.5 μM).
NOTE: For every 20 mL cell suspension, a 20 mL CFSE tube is needed. - Invert the cell and CFSE mixture tube 2 - 3 times and incubate the mixture in the dark at room temperature for a maximum of 2 min, then stop the reaction by adding 10 mL of heat-inactivated horse serum.
- Centrifuge the 50 mL tube mixture at 300 x g for 5 min at room temperature.
NOTE: If the CFSE labeling is successful, the cell pellet will become light yellow. - Remove the supernatant and resuspend the cell pellet in 40 mL of 1x PBS and count cell numbers with a hemocytometer.
- Centrifuge the cell suspension at 300 x g for 5 min and discard the supernatant.
- Wash the cell pellet again with 40 mL of RPMI1640 medium.
- Remove the supernatant and resuspend cells with RPMI1640 tissue culture medium (RPMI1640, 20 mM HEPES, 10% FBS (heat inactivated), 20 mM glutamine, and 1x Pen/Strep) at a concentration of 7 x 106 cells/mL in a 100 mm tissue culture dish.
NOTE: If more cells are obtained, a separate 100 mm tissue culture dish will be needed. - Add staurosporine into the cell suspension culture at a final concentration of 1 μM and culture for 4 h at 37 °C in a tissue culture incubator supplied with 5% CO2.
2. Preparation of peritoneal macrophages
- Inject two C57B6 mice (or any gene-manipulated mice in the lab) intraperitoneally with 1 mL of 3% aged thioglycollate at day 0.
- Euthanize the mice on day 5 as in step 1.1, cut and peel open the abdominal skin but leave the peritoneum intact. Flush the peritoneal cavity by quickly pushing 10 mL of wash buffer (RPMI1640, 2% FBS, 0.04% EDTA) into the peritoneal cavity using a 10 mL syringe attached to an 18 G needle.
- Retrieve the wash buffer slowly with the same needle/syringe and collect the wash buffer into a 50 mL tube.
- Wash the peritoneal gavage twice with 1x PBS, resuspend the peritoneal macrophages in RPMI1640 tissue culture medium at a density of 2 x 106 cells/mL and aliquot 500 μL into each well of the 24-well plate. Leave the plate in a tissue culture incubator for 2 h.
- Remove the floating cells by aspirating and replacing them with 500 μL of fresh culture medium, twice.
- Optionally, add TAM receptor tyrosine inhibitor, RXDX-106, at concentrations indicated in the figure legends into each well of the macrophage culture and incubate for another 2 h.
3. Co-culture of peritoneal macrophages with apoptotic thymocytes
- Collect apoptotic thymocytes from step 1 and wash three times with RPMI1640 medium. The efficiency of apoptotic induction can be measured at this stage with the Annexin V/7-AAD kit.
- Distribute 0 - 12 x 106 cells (in 500 μL medium) into each well of the macrophage cultures from protocol #2, according to the experimental arrangement (e.g., see Figure 1). This makes the whole culture volume of 1 mL in each well of the 24-well plate. Add the blocking antibody into the culture immediately before the addition of apoptotic thymocytes.
- Culture the cell mixture at 37 °C for 4 h in a tissue culture incubator supplied with 5% CO2.
- Wash each well of the culture with 1x PBS (containing 500 μM EDTA) twice to remove the free-floating apoptotic cells.
- Stain the plate-bound macrophages with CD11b-PE at this stage.
- Wash the plate-bound macrophages with a staining buffer (1x PBS, 1% BSA) once.
- Add 200 μL of staining buffer containing 2 μL CD11b-PE into each well.
- Incubate the plate at 4 °C for 20 min.
- Wash each well of the plate three times with the staining buffer.
- Add 200 μL of staining buffer and proceed with the plate for image analysis under the fluorescent microscope.
Results
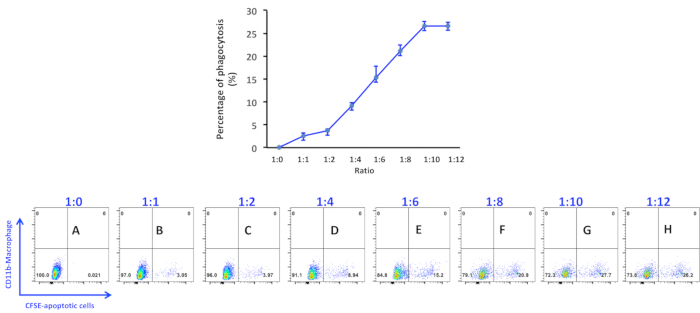
Figure 1. Percentage of phagocytosis of apoptotic thymocytes by peritoneal macrophages. Apoptotic thymocytes were induced by incubating with 1 μM of staurosporine for 4 h and added into macrophage culture at a ratio as indicated in the figure panels: (A) 1:0; (B) 1:1; (C) 1:2; (D) 1:4; (E) 1:6; (F) 1:8; (G) 1:10; (H) 1:12. Data were acquired with a flow cytometer. The percen...
Disclosures
Materials
| Name | Company | Catalog Number | Comments |
| Ack lysing buffer | GIBCO | A10492 | |
| Annexin V/7-AAD | BD Pharmingen | 559763 | |
| Anti-Mer antibody | R&D Systems | BAF591 | |
| CD11b-PE (clone M1/70) | BD Pharmingen | 553311 | |
| CFSE | Invitrogen | C1157 | |
| DMSO | Sigma-Aldrich | D-2650 | |
| EDTA (0.5 mM) | GIBCO | 15575-020 | |
| FACS tubes | BD Biosciences | 352017 | |
| Horse Serum (Heat-inactivated) | Invitrogen | 26050088 | |
| PBS, 1x | Corning | 21040CV | |
| RPMI-1640 | Corning | 10040CV | |
| RXDX-106 | Selleck Chemicals | CEP-40783 | |
| Staurosprine (100mg) | Fisher Scientific | BP2541-100 | Add 214.3 ml of DMSO into 100mg to make 1mM stocking solution |
This article has been published
Video Coming Soon
Source: Zhen, Y. et al., Experimental Analysis of Apoptotic Thymocyte Engulfment by Macrophages. J. Vis. Exp. (2019)
Copyright © 2025 MyJoVE Corporation. All rights reserved