Detection of Neuritic Plaques in Alzheimer's Disease Mouse Model
* These authors contributed equally
In This Article
Summary
One of the pathological characteristics of AD is the formation of Amyloid β protein positive neuritic plaques. In this protocol we describe two methods to detect neuritic plaques in transgenic AD model mice: immunohistochemical detection using the ABC and DAB method and fluorescent detection using thioflavin S staining method.
Abstract
Alzheimer's disease (AD) is the most common neurodegenerative disorder leading to dementia. Neuritic plaque formation is one of the pathological hallmarks of Alzheimer's disease. The central component of neuritic plaques is a small filamentous protein called amyloid β protein (Aβ)1, which is derived from sequential proteolytic cleavage of the beta-amyloid precursor protein (APP) by β-secretase and γ-secretase. The amyloid hypothesis entails that Aγ-containing plaques as the underlying toxic mechanism in AD pathology2. The postmortem analysis of the presence of neuritic plaque confirms the diagnosis of AD. To further our understanding of Aγ neurobiology in AD pathogenesis, various mouse strains expressing AD-related mutations in the human APP genes were generated. Depending on the severity of the disease, these mice will develop neuritic plaques at different ages. These mice serve as invaluable tools for studying the pathogenesis and drug development that could affect the APP processing pathway and neuritic plaque formation. In this protocol, we employ an immunohistochemical method for specific detection of neuritic plaques in AD model mice. We will specifically discuss the preparation from extracting the half brain, paraformaldehyde fixation, cryosectioning, and two methods to detect neurotic plaques in AD transgenic mice: immunohistochemical detection using the ABC and DAB method and fluorescent detection using thiofalvin S staining method.
Protocol
1. Fixation of mouse brains and cryosectioning
- Transgenic mice are sacrificed using carbon dioxide gas, followed by decapitation and the brain extracted.
- The brain is dissected longitudinally at the center. Half of the brain will be flash frozen on dry ice for biochemical analysis. The other will be submerged in 4% paraformaldehyde (PFA) for at least 48 hours at 4°C.
- After fixation in 4% PFA for at least 48 hours, the hemi brain is transferred directly into 30% sucrose solution and placed at 4°C. At the point, the hemi brain should be floating in the sucrose solution. When the hemi brain sank to the bottom, usually requiring about 48 hours, it is ready to be embedded in OCT for cryosectioning.
- Before cryosectioning, mount a thin layer of OCT on the knob and allow to freeze at -20°C. Remove the cerebellum from the rest of the hemi brain and place onto the layer of frozen OCT. Immediately cover the hemi brain with OCT and allow to slowly freeze at -20°C. Once the OCT embedding the brain has turned opaque, it is ready to be sectioned.
- Set the thickness of each section to 30 μm. Collect each slice into a container with D'Olomos solution.
2. Immunohistochemistry procedure for neuritic plaques (free floating sections)
- Treat each section in 88% formic acid for 15 minutes. After formic acid treatment, the section will temporarily turn opaque and may appear to shrivel.
- Remove each section into a plate and wash with Tx-PBS (0.3% Triton X-100 in PBS) for 5 min X 3 with gentle shaking.
- Block Endogenous Peroxide blocking 0.5% H2O2 in Tx-PBS, RT 30 min with gentle shaking.
- Wash section with Tx-PBS for 10 min X 3 with gentle shaking.
- Block section with 5% non-fat skim milk dissolved in Tx-PBS at room temperature for 1 hour with gentle shaking.
- Incubate sections in primary antibody (Biotinylated 4G8, 1:500-1:2000, Signet) diluted in Tx-PBS containing 5% milk at 4°C overnight with gentle shaking.
- Wash section with Tx-PBS for 10 min X 3 with gentle shaking.
- Prepare ABC solution following manufacturer's protocol (Elite ABC, Vector Laboratories,). Incubate sections in ABC mixture for 30 min with gentle shaking.
- Wash section with Tx-PBS for 10 min X 3 with gentle shaking.
- Prepare 3,3'-diaminobenzidine tetrahydrochloride (DAB) (Vector Laboratories) following manufacturer's protocol. Incubate sections in DAB mixture for 5-10 min. At this point, you will observe a brownish color develop on each section.
- Wash section with Tx-PBS for 10 min X 3 with gentle shaking.
- Free-floating brain sections are mounted on gelatin coated slides, to help with tissue adhesion. Sections are then air dried followed by clearing with a series of xylene and mounted in Entellen.
- Plaques are visualized under light microscopy at 40X magnification and are quantified by assessing the mean plaque count per slice for each mouse.
3. Thioflavin S staining procedure for detection of neuritic plaques
- Mouse sacrifice and brain extraction procedure are as indicated in sections 1.1 to 1.5.
- Mount 4 to 5 hemi brain sections onto a glass slide and allow to completely air dry prior to staining.
- Wash slides with 70% Ethanol for 1 minute.
- Wash slides with 80% Ethanol for 1 minute.
- Incubate slides in filtered (0.2 μm filter) thioflavin S solution (1% in 80% of Ethanol) for 15 minutes. (Thioflavin S solution needs to be filtered before each use). Note: protect thioflavin S from light and protect stained slides from light.
- Wash slides with 80% Ethanol for 1 minute.
- Wash slides with 70% Ethanol 1 minute.
- Wash with distilled water twice.
- Mount slides in aqueous mounting media and allow slides to dry in the dark for at least 2 hour, followed by sealing coverslip with clear nail polish. Stained sections should be stored in the dark at 4°C.
- The green fluorescence stained plaques could be visualized with fluorescent microscropy. Analyze slides within one week because the staining will fade with time.
4. Representative Results
In our recent study on the efficacy of the anti-epileptic drug and mood stabilizer valproic acid (VPA) to inhibit neuritic plaque formation, we used the above described immunostaining and thioflavin S staining procedures to identify neuritic plaques in AD model mice (3). Figure 1 represents a typical 9 month old APP23 transgenic mouse, stained with anti-Aβ using biotinylated 4G8 and detected via the ABC method. Neuritic plaques are clearly labeled with the antibody and are indicated by the white arrows. Using thioflavin S staining, we detected neuritic plaque deposition 2 month old APP23xPS transgenic mice (Figure 2). Thioflavin S bound Aβ-containing neuritic plaques appears green under fluorescence microscopy (indicated by white arrows). We showed that with VPA treatment, neuritic plaque formation is significantly reduced (3). In another study, we studied the molecular link underlying hypoxia and AD pathogenesis. Again, using both 4G8 anti-Aβ immunohistochemistry staining and thioflavin S staining, we found that hypoxia facilitated the formation of Aβ containing neuritic plaques (4).
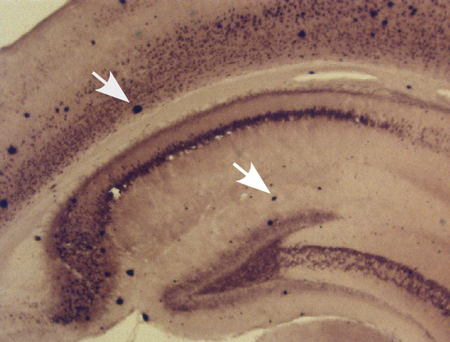
Figure 1. Immunohistochemical analysis of neuritic plaque formation in AD transgenic mice. APP23 mice at the age of 9 months were sacrificed and were dissected, fixed, and sectioned. Neuritic plaques in the hippocampal were detected using an anti-Aβ antibody 4G8 and were developed using the ABC and DAB methods. The plaques were visualized by light microscopy with 40X. White arrows point to Aβ neuritic plaques.
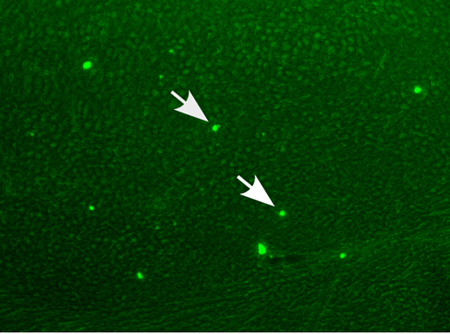
Figure 2. Thioflavin S staining of neuritic plaques in AD modeled mice. APP23xPS45 double transgenic mice at 8 weeks old were sacrificed and were dissected, fixed, and sectioned. Neuritic plagues in the hippocampal are were confirmed using thioflavin S fluorescent staining and visualized by microscopy at 40X magnification. White arrows indicate thioflavin S stained neuritic plaques.
Discussion
Immunohistochemistry using the biotinylated labeled 4G8 antibody stains neuritic plaques in AD modeled mice with specificity. The staining outcome allows quantification and comparison of plaque load between different treatment groups. There are several crucial steps that could affect the outcome. Because the hemi brains are not perfused prior to extraction from the skull, care should be used during the extraction process in order to prevent damages to the hemi brain. Moreover, since the hemi brain is passively perfused, we recommend incubating it in 4% PFA for at least 48 hours at 4°C prior to submerging in the 30% sucrose solution. Alternatively, transcardial perfusion could be done prior to extracting the brain. If the brain is fixed properly, it should have a rubbery texture. In the case where the brain is not fixed properly, the sections will easily tear in the D'Olomos solution or during the staining procedures.
The 4G8 monoclonal antibody is reactive to amino acid residues 17-24 of Aβ and recognized the epitope in the core sequence (VFFAE). Since 4G8 is derived from mouse species, it tends to give a higher background staining. Thus, we chose to incubate the sections at the indicated dilution with considerations to achieving a true signal while minimizing background staining.
In addition to immunostaining for neuritic plaques detection using the 4G8 antibody, a thioflavin S staining method is also used to identify plaques. Thioflavin S is a homogenous dye mixture that results from methylation of dehydrothiotoluidine with sulfonic acid. Thioflavin S non-selectively binds beta sheet contents of proteins, such as those in amyloid oligomers. Upon binding, thioflavin undergoes a characteristic blue shift of its emission spectrum. Conversely thioflavin S binding to the monomeric forms will not elicit a blue shift and could not be detected with florescent microscope. Thioflavin S staining provides a quick alternative to screen for amyloid as the intensity of fluorescence allows good visualization of small amounts of amyloid deposits. However, one drawback about thioflavin S staining is the lack of specificity. Many other tissue components including containing extensive beta sheets, such as fibrinoids, hyaline, keratin, etc, have a rather affinity for this dye.
Acknowledgements
This work was supported by the Canadian Institutes of Health Research (CIHR), the Townsend Family, and Jack Brown and Family Alzheimer's Research Foundation (to W. S.). W. S. is the holder of the Canada Research Chair in Alzheimer's Disease. P. T.T. L. is supported by the Natural Sciences and Engineering Research Counsel and the Michael Smith Foundation for Health Research Scholarship.
Materials
| Name | Company | Catalog Number | Comments |
| Name of the reagent and equipment | Company | Catalogue number | |
|---|---|---|---|
| Paraformaldehyde | Sigma-Aldrich | P6148-1KG | |
| Polyvinylpyrrolidene | Sigma-Aldrich | PVP40-100G | |
| Sucrose | Fisher Scientific | S5-3 | |
| Ethylene glycol | Sigma-Aldrich | 324558-2L | |
| Tissue-Tek O.C.T. Compound | Sakura | 4583 | |
| 88% formic acid | Fisher Scientific | A118P-500 | |
| Triton X-100 | ICN Biomedicals | 807426 | |
| H2O2 | Sigma-Aldrich | H-1009 | |
| Biotin Labeled 4G8 Antibody | Cedarlane | SIG-39240-500 | |
| Elite ABC kit | Vector | PK-6100 | |
| Imm PACT DAB | Vector | SK-4105 | |
| Xylene | Fisher Scientific | X4-4 | |
| Entellan | EM science | 65037-71 | |
| Superfrost* Plus Microscope Slides | Fisher Scientific | 12-550-15 | |
| Cryostat | Leica | CM-3050-S | |
| MX35 Premier microtome blade | Thermo Scientific | 3052835 | |
| Thioflavin S | Sigma-Aldrich | T1892-25G |
References
- Glenner, G. G., Wong, C. W. Alzheimer's disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun. 120, 885-890 (1984).
- Hardy, J., Selkoe, D. J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science. 297, 353-356 (2002).
- Qing, H., He, G., Ly, P. T., Fox, C. J., Staufenbiel, M., Cai, F., Zhang, Z., Wei, S., Sun, X., Chen, C. H., Zhou, W., Wang, K., Song, W. Valproic acid inhibits Abeta production, neuritic plaque formation, and behavioral deficits in Alzheimer's disease mouse models. J Exp Med. 205, 2781-2789 (2008).
- Sun, X., He, G., Qing, H., Zhou, W., Dobie, F., Cai, F., Staufenbiel, M., Huang, L. E., Song, W. Hypoxia facilitates Alzheimer's disease pathogenesis by up-regulating BACE1 gene expression. Proc Natl Acad Sci U S A. 103, 18727-18732 (2006).
This article has been published
Video Coming Soon
ABOUT JoVE
Copyright © 2024 MyJoVE Corporation. All rights reserved