A subscription to JoVE is required to view this content. Sign in or start your free trial.
An Automated Differential Nuclear Staining Assay for Accurate Determination of Mitocan Cytotoxicity
* These authors contributed equally
In This Article
Summary
The protocol describes a rapid, high-throughput, reliable, inexpensive, and unbiased assay for efficiently determining cellular viability. This assay is particularly useful when cells' mitochondria have been damaged, which interferes with other assays. The assay uses automated counting of cells stained with two nuclear dyes – Hoechst 33342 and propidium iodide.
Abstract
The contribution of mitochondria to oncogenic transformation is a subject of wide interest and active study. As the field of cancer metabolism becomes more complex, the goal of targeting mitochondria using various compounds that inflict mitochondrial damage (so-called mitocans) is becoming quite popular. Unfortunately, many existing cytotoxicity assays, such as those based on tetrazolium salts or resazurin require functional mitochondrial enzymes for their performance. The damage inflicted by compounds that target mitochondria often compromises the accuracy of these assays. Here, we describe a modified protocol based on differential staining with two fluorescent dyes, one of which is cell-permeant (Hoechst 33342) and the other of which is not (propidium iodide). The difference in staining allows living and dead cells to be discriminated. The assay is amenable to automated microscopy and image analysis, which increases throughput and reduces bias. This also allows the assay to be used in high-throughput fashion using 96-well plates, making it a viable option for drug discovery efforts, particularly when the drugs in question have some level of mitotoxicity. Importantly, results obtained by Hoechst/PI staining assay show increased consistency, both with trypan blue exclusion results and between biological replicates when the assay is compared to other methods.
Introduction
The first step to identifying effective cancer treatments is the selection of a robust, unbiased cytotoxicity assay that can be used to examine the effect of treatment. A common choice for low-throughput experiments is the exclusion of trypan blue dye from living cells. This method is favored because it allows a relatively unbiased method for quantifying cell survival. trypan blue passively diffuses into cells whose membranes are compromised, but it is effectively blocked from entering healthy cells1. The quotient of the living cells and the total cells represents the percent viability, which indicates the efficacy of the treatment. The most significant disadvantage of the trypan blue assay is that it is poorly suited for high-throughput methodologies. It has a relatively low signal-to-noise ratio and prolonged staining can result in artifacts due to the staining of viable cells. Consequently, trypan blue exclusion is typically, but not always2, relegated to manual counting. This makes it too slow and introduces the strong possibility of bias due to subjective judgment of the researcher (unless blinding or independent counts are used, which further reduce laboratory throughput). In general, the throughput of this assay is insufficient for modern drug discovery.
Viability assays, which generally have a much higher throughput, allow researchers to circumvent this limitation, but come with significant caveats (see Table 1). These methods generally fall into two groups. One group is comprised of colorimetric assays that are based on the function of cellular redox enzymes. Colorless or non-fluorescent substrates are converted into vibrant products that can be quantified using a spectrophotometer. Classic examples include tetrazolium salts (MTT, WST-1, XTT, etc.) and resazurin. This category also includes luminescent assays that utilize luciferin to evaluate ATP level. Assays of this type have the underlying limitation that they are measuring cellular metabolism, which is not cellular viability per se. It is quite common for cells to become quiescent under adverse conditions, but still retain the ability to divide3,4,5. For example, cancer stem cells are often relatively metabolically quiescent6,7,8,9, and are likely to be difficult to assay using these techniques. Effectiveness of treatments that damage mitochondrial function, such as most mitocans, is also likely to be significantly overestimated.
An alternative methodology leverages the chemical properties of various substances that allow them to either cross or not cross biological membranes. One example are nuclear stains such as SYTOX or propidium iodide (PI). This category also includes assays that are similar in concept but different in function, such as the lactate dehydrogenase (LDH) assay, which measures the release of LDH into the extracellular milieu as an indicator of cellular necrosis (Figure 1, Table 1). These assays are more capable of distinguishing between metabolically inactive and dead cells.
| Assay/dye | Type(s) of cell death detected | Necessary equipment | Key features |
| MTT, CKK-8, Alamar Blue (resazurin) | Apoptosis/Necrosis | Spectrophotometer | Inexpensive, rapid; endpoint assay; dependent on enzymes' activity (exclusively mitochondrial in case of MTT) and does not discriminate between modes of cell death1,10 |
| LDH release | Necrosis | Spectrophotometer | Rapid, independent of mitochondrial enzymes’ activity; expensive for high-throughput tests; detects necrotic cells with compromized plasma membrane11,12 |
| Trypan Blue (TB) | Apoptosis/Necrosis | Microscope | Cell-impermeant; does not discriminate between modes of cell death; laborous and not suitable for high-throughput screening; more difficult to use with adherent cells; prone to subjective judgment of the user, but is considered the standard cell viability measurement method13 |
| Acridine orange (AO) | Apoptosis/Necrosis/ | Fluorescence microscope | A nucleic acid dye with unique spectral properties, can distinguish between apoptosis and necrosis/necroptosis14 |
| Necroptosis | |||
| Hoechst 33342, DAPI | Apoptosis | Fluorescence microscope or flow cytometer | Cell-permeable; inappropriate on its own to monitor cell death; useful for co-staining; can be used to assess chromatin condensation and nuclei fragmentation in early apoptosis; can be paired with propidium iodide to distinguish apoptosis from necrosis15,16 |
| Propidium Iodide (PI) | Late apoptosis/Necrosis | Fluorescence microscope or flow cytometer | Cell-impermeant intercalator; detects both late apoptosis and necrosis modes of cell death17. Toxic and permeable after long incubation times18 |
Table 1. List of cytotoxicity assays. Cytotoxicity assays, some of which were used in this study, listed along with the brief description of their key features.
Recent studies have demonstrated that mitochondrial metabolism is altered in some cancers19,20,21,22,23,24,25. For example, acute myeloid leukemias (AML) have been shown to upregulate their mitochondrial mass, mtDNA content, and mitochondrial respiration to meet their energy demands19,26,27. On the other hand, some solid tumors are characterized by mitochondrial dysfunction, or rather “metabolic reprogramming”, such as downregulation of mitochondrial proteins involved in OXPHOS or decreased mtDNA content, that has been associated with tumor invasiveness, metastatic potential and resistance to apoptosis-inducing drugs28,29. Furthermore, recently, there has been an increased interest in using mechanistically diverse compounds that affect mitochondrial function (generally called mitocans30), as potential therapies for particular cancers. These drugs target ATP synthesis, mitochondrial DNA, OXPHOS, and ROS production, as well as pro-apoptotic and anti-apoptotic proteins associated with mitochondria30,31. Several studies have shown that this approach has significant promise19,32,33,34. However, these metabolic deviations in cancer cell biology or mitochondria-targeting treatments may significantly affect conventional viability assays that are based on mitochondrial functionality.
Here, an optimized protocol for a differential nuclear staining assay is described. The protocol allows fast and accurate determination of cytotoxicity of mitocans or their combinations with other compounds. Hoechst 33342 is a cell-permeant nuclear dye that readily crosses cell membranes to stain DNA, allowing the total cell count to be obtained. By co-staining with PI, which only enters the nuclei of dead cells, the proportion of living (Hoechst only) and dead (stained with both) cells can be accurately determined. This protocol refines the published assay35 by adding a step for the optimization of the dye concentration (by cross-referencing results with orthogonal trypan blue method) and centrifugation of the plate prior to imaging. Since many cell lines are semi-adherent or suspended, centrifugation increases the proportion of cells that are imaged and strongly improves accuracy. The assay has several advantages, including that staining does not require removal of media or washing. The dye mixture is also inexpensive, easy to prepare, and compatible with multichannel/robotic pipetting systems.
After cells have been stained, they are imaged with an automated microscope. This has the added advantage of creating a permanent record of the images that can be re-analyzed later and the effects of particular compounds can be re-evaluated by visual inspection of captured images. Once images have been obtained, cells can be counted either manually or by using any of several software packages, including both free (e.g., ImageJ, CellProfiler, etc) and commercial software (e.g., Metamorph, Gen5, etc). Automated cell counting is generally preferable since properly developed automated cell counting pipelines are more accurate and less biased than manual counts. They also more effectively disregard cell debris or insoluble complexes. Development of these pipelines is generally straightforward and is simplified by the efficiency of the stains used. The output is quantitative since the actual number of dead cells is automatically calculated with respect to total cell number, and different thresholds can be applied to increase or decrease the stringency of detection35. For convenience, optimized parameters for counting cells using Gen5 v. 3.00 software compatible Cytation 5 Cell Imaging Multi-Mode Reader are included.
Protocol
1. Cytotoxicity Assay: Setup
- Prepare solutions of compounds of interest at desired concentrations in the appropriate media (serum-free or 1, 2.5, or 5% FBS RPMI-1640).
- To measure cytotoxicity of a single compound (e.g., to determine effective doses), prepare compounds at 2x final concentration.
- To measure cytotoxicity of compound combinations, prepare compounds at 4x final concentration.
- Prepare solvent-only controls by mixing the same amount of solvent with the appropriate medium. For example, if testing compounds dissolved in DMSO and methanol, make solvent-only control for each solvent.
- Collect cells from culture dish or flask into a 15 mL conical tube.
- Transfer 10 µL of cell suspension into a microcentrifuge tube and stain with 10 µL 0.4% trypan blue. Use a hemocytometer to count viable and non-viable cells for each cell source.
- Pellet cells at 200 g for 5 min. Aspirate or decant supernatant.
- Resuspend cell pellet in assay-appropriate media (serum-free or 1, 2.5, 5% FBS RPMI-1640) at a cell density of 3*105 cells/mL.
NOTE 1: Cell density of 3*105 cells/mL provides a seeding density of 15,000 cells/well. Seeding density is an important parameter and ideally should be pre-defined prior to the experiment. Seeding density should take into account 1) cell size – usually larger cells are seeded at a lower density; 2) treatment duration – cells are typically seeded at a lower density for experiments that will last longer; and 3) cell division rate – cells with a higher rate of division are seeded at a lower density. Specific examples of optimized seeding densities: K562 cells, larger, 24 h duration – 10,000 cells/well; MOLM-13 cells, moderate size, 24 h treatment – 15,000/well; MOLM-13 cells, 48 h treatment – 8,000/well; small healthy peripheral blood mononuclear cells (PBMCs), 24 h treatment – 50,000/well; primary AML cells, 24 h treatment – 15,000-20,000/well.
NOTE 2: The presence of FBS in media may affect the activity of the compounds. Reducing the concentration of FBS may make assay results simpler to interpret, but it also reduces the physiological accuracy. - Seed 50 µL of cell suspension from step 1.5 into each well of a 96-well plate using a multichannel pipette.
- Add compounds as follows:
- For single compound assays, add 50 µL of 2x compound solution into each well. For solvent-control wells, add 50 µL of test media containing the solvent at the 2x concentration.
- For combination assays, add 25 µL of each of the compound (4x solutions) into each well. For single compound-control wells, add 25 µL of 4x compound solution and 25 µL of test medium. For solvent-control wells, add 50 µL of test medium or test medium containing the solvent.
NOTE 1: The final concentration of DMSO should not exceed 0.5%.
NOTE 2: It is recommended to add the media containing the compounds with the pipette touching the wall of each well due to low volume.
- Gently tap the plate to ensure mixing of the contents of the wells.
- Incubate plates at 37 °C in a humidified 5% CO2 atmosphere for an appropriate time, e.g., 24 h.
2. Cytotoxicity Assay: staining with Hoechst 33342 and propidium Iodide
- Prepare 10x staining solution. This solution needs to be prepared fresh before each experiment, it cannot be stored. Final dye concentrations should be determined prior to the experiment.
- For leukemia cell lines and primary leukemia cells, 1 mL of 10x staining buffer contains 10 µL of 20 mM Hoechst 33342 and 50 µL of 1 mg/mL propidium iodide in sterile PBS (final concentrations: Hoechst 33342 20 µM, PI 5 µg/mL).
- For healthy PBMCs, 1 mL of 10x staining buffer contains 10 µL of 20 mM Hoechst 33342 and 10 µL of 1 mg/mL propidium iodide in sterile PBS (final concentrations: Hoechst 33342 20 µM, PI 1 µg/mL).
NOTE: The final concentration of propidium iodide has to be determined prior to the experiments. Cells should be tested using a range of PI concentrations (1, 2.5, 5 µg/mL), and then Hoechst/PI-calculated viability should be compared with viability measured via trypan blue. The PI concentrations listed above were chosen based on target cell viability in media-control wells (above ~90% for leukemia cell lines, above ~70% for healthy PBMCs).
CAUTION: Hoechst 33342 and propidium iodide are potential carcinogens. Wear appropriate personal protective equipment when handling them.
- After incubation, use a multichannel pipette to add 10 µL of 10x staining buffer to each well.
NOTE: To prevent cross-contamination, make sure that the pipette tips do not touch the media. - Gently tap the plate to mix and clear bubbles. Stain at 37 °C for 15 min.
- Centrifuge the plate at 200 g for 4 min to bring all of the cells to the bottom of the plate. Carefully wipe the bottom of the plate with a damp kimwipe to remove fibers and/or debris that will interfere with imaging.
NOTE 1: Centrifugation of the plate ensures the highest chances for all of the cells to be captured in the image. Often dead cells will detach and float, providing misleading values of cytotoxicity. Centrifugation mitigates this effect.
NOTE 2: The plate should be imaged as quickly as possible after centrifugation, ideally, within 15 min. Centrifugation can reduce the selectivity of PI staining and may allow cells to slowly accrue propidium iodide. The improvement in accuracy gained by visualizing the dead cells outweighs the slight increase in PI staining. It is recommended to finish imaging within 1 h of centrifugation.
3. Cytotoxicity Assay: data acquisition
- Set the software for the automated microscope/plate imager to detect fluorescence for both Hoechst 33342 (excitation maximum 350 nm, emission maximum 461 nm) and PI (excitation maximum 493 nm, emission maximum 636 nm). Acquire images for each well in both channels.
- Using the software (such as CellProfiler, a free image analysis studio based on MatLab http://cellprofiler.org/, ImageJ, or proprietary software such as Gen5) count the cells in each well in each channel.
NOTE: Since every cell should be stained with Hoechst 33342, and dead cells should be stained with PI, the ratio of dead to all represents the fraction of cells that are dead. For example, if the automated count in untreated sample shows 467 cells stained with PI (dead cells) and 2335 cells stained with Hoechst 33342 (total cells), the fraction dead is 0.2 or 20%. This value is then compared to an identically handled sample where treatment was used. - Detailed description of Hoechst/PI data acquisition using Cytation5 Cell Imaging Multi-Mode Reader and Gen5 v. 3.00 software:
- Set the Cytation5 multimode plate reader/imager to image cells in flat bottom, 96-well, generic black plastic plates.
- Set an imaging protocol to utilize the standard DAPI and Texas Red filter sets. Take images at the well center using a 4x magnification objective. Use no offset (X/Y or Z). Use the following imaging settings: DAPI – LED - 10, integration time - 99, gain - 0; Texas Red – LED - 8, integration time - 950, gain - 18. Perform autofocusing using the DAPI signal; there should be no offset in focusing between channels.
- Perform image analysis using Gen5 software v 3.00. In the software settings, define cells as shapes between 5 and 25 µm in their size. Exclude primary edge objects and split touching objects by turning on special option “Split touching objects”. Next, process the image to remove the background (dark background subtraction), apply a nuclear mask (threshold value DAPI >= 6000 AU), and count objects. Perform a subpopulation analysis based on PI staining (threshold value Texas Red >= 5000 AU), and count objects. Cell viability % is defined as (1 -
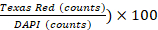 .
.
Results
The aforementioned protocol has been developed using OCI-AML2 cells, which were taken as a representative acute myeloid leukemia cell line. AML is characterized by abnormal proliferation of undifferentiated and non-functional hematopoietic cells in the bone marrow26. Despite recent developments in AML targeted therapy, the standard of care has remained unchanged for several decades, and consists of induction therapy (typically comprised of three days of anthracycline, e.g., daunorubicin, idar...
Discussion
Although the protocol for Hoechst/PI cytotoxicity assay is robust and requires comparatively little hands-on time, there are several experimental details that are very important to ensure accurate results. First, it is essential to make sure that the concentration of DMSO remains below 0.5% (v/v). It is generally agreed that exposure to even low doses of DMSO can substantially alter the morphology and attachment of cells and significantly delay cell cycle progression39,
Disclosures
The authors have nothing to disclose.
Acknowledgements
NVK, a CPRIT scholar in Cancer Research, thanks the Cancer Prevention and Research Institute of Texas (CPRIT) for their generous support, CPRIT grant RR150044. This work was also supported by the Welch Foundation Research Grant C-1930, and by the National Institutes of Health R35 GM129294 awarded to NVK. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Materials
| Name | Company | Catalog Number | Comments |
| 2-Deoxy-D-glucose/2-DG | Chem-Impex | 50-519-067 | |
| 3-bromo-pyruvate | Alfa Aesar | 1113-59-3 | |
| 96-Well plates | Greiner Bio-One | 655090 | Black or clear flat-bottomed 96-well plates |
| Alamar blue HS cell viability reagent (100mL) | Thermo Fisher | A50101 | |
| Countess II automated cell counter | Thermo Fisher | ||
| Cytation 5 Cell Imaging Multi-Mode Reader | BioTek | ||
| Hoechst 33342 | Thermo Fisher | 62249 | 20 mM solution; final concentration 1:1,000 |
| HyClone fetal bovine serum | GE Healthcare | #25-514 | |
| m-chlorophenylhydrazone/CCCP | Sigma Aldrich | C2759 | |
| PBS tablets | Thermo Fisher | BP2944100 | 1 tablet + 200 mL of sterile water = 1x PBS solution |
| Penicillin-Streptomycin-Glutamine (100X) | Gibco | 10378016 | |
| Pierce LDH assay kit | Thermo Fisher | 50-103-5952 | |
| Propidium Iodide | Thermo Fisher | 50-596-072 | Dry powder; stock 1 mg/mL in PBS; final concentration 5 µg/mL (leukemia cells), 1 µg/mL (normal PBMCs) |
| Rotenone | Ark Pharm | AK115691 | |
| RPMI-1640 Medium With L-glutamine and sodium bicarbonate, liquid, sterile-filtered, suitable for cell culture | Sigma Aldrich | R8758-500ML | |
| Thiazolyl blue tetrazolium bromide | ACROS Organics | AC158990010 | |
| Trypan blue stain (0.4%) | Gibco | 15250-061 | |
| Cell lines | |||
| K562 | ATCC | CCL-243 | CML cell line |
| MOLM-13 | ATCC | AML cell line | |
| MOLT-4 | ATCC | CRL-1582 | ALL cell line |
| OCI-AML2 | ATCC | AML cell line | |
References
- Ramirez, C. N., Antczak, C., Djaballah, H. Cell viability assessment: toward content-rich platforms. Expert Opinion on Drug Discovery. 5 (3), 223-233 (2010).
- Melzer, S., et al. Trypan blue as an affordable marker for automated live-dead cell analysis in image cytometry. Scanning. 38 (6), 857-863 (2016).
- Sikora, E., Mosieniak, G., Sliwinska, M. A. Morphological and Functional Characteristic of Senescent Cancer Cells. Current Cancer Drug Targets. 17 (4), 377-387 (2016).
- Coppé, J. P., Desprez, P. Y., Krtolica, A., Campisi, J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annual Review of Pathology. 5, 99-118 (2010).
- Castro-Vega, L. J., et al. The senescent microenvironment promotes the emergence of heterogeneous cancer stem-like cells. Carcinogenesis. 36 (10), 1180-1192 (2015).
- Weihua, Z., Lin, Q., Ramoth, A. J., Fan, D., Fidler, I. J. Formation of solid tumors by a single multinucleated cancer cell. Cancer. 117 (17), 4092-4099 (2011).
- Osisami, M., Keller, E. T. Mechanisms of Metastatic Tumor Dormancy. Clinical Medicine. 2 (3), 136-150 (2013).
- Zhang, S., et al. Generation of cancer stem-like cells through the formation of polyploid giant cancer cells. Oncogene. 33 (1), 116-128 (2014).
- Mittal, K., et al. Multinucleated polyploidy drives resistance to Docetaxel chemotherapy in prostate cancer. British Journal of Cancer. 116 (9), 1186-1194 (2017).
- McKeague, A. L., Wilson, D. J., Nelson, J. Staurosporine-induced apoptosis and hydrogen peroxide-induced necrosis in two human breast cell lines. British Journal of Cancer. 88 (1), 125-131 (2003).
- Kaja, S., et al. An optimized lactate dehydrogenase release assay for screening of drug candidates in neuroscience. Journal of Pharmacological and Toxicological Methods. 73, 1-6 (2015).
- Chan, F. K., Moriwaki, K., De Rosa, M. J. Detection of necrosis by release of lactate dehydrogenase activity. Methods in Molecular Biology. 979, 65-70 (2013).
- Piccinini, F., Tesei, A., Arienti, C., Bevilacqua, A. Cell Counting and Viability Assessment of 2D and 3D Cell Cultures: Expected Reliability of the Trypan Blue Assay. Biological Procedures Online. 19, 8 (2017).
- Plemel, J. R., et al. Unique spectral signatures of the nucleic acid dye acridine orange can distinguish cell death by apoptosis and necroptosis. Journal of Cell Biology. 216 (4), 1163-1181 (2017).
- Galluzzi, L., et al. Guidelines for the use and interpretation of assays for monitoring cell death in higher eukaryotes. Cell Death & Differentiation. 16 (8), 1093-1107 (2009).
- Cummings, B. S., Schnellmann, R. G. Measurement of cell death in mammalian cells. Current Protocols in Pharmacology. , (2004).
- Brauchle, E., Thude, S., Brucker, S. Y., Schenke-Layland, K. Cell death stages in single apoptotic and necrotic cells monitored by Raman microspectroscopy. Scientific Reports. 4, 4698 (2014).
- Chiaraviglio, L., Kirby, J. E. Evaluation of impermeant, DNA-binding dye fluorescence as a real-time readout of eukaryotic cell toxicity in a high throughput screening format. Assay and Drug Development Technologies. 12 (4), 219-228 (2014).
- Panina, S. B., Baran, N., Brasil da Costa, F. H., Konopleva, M., Kirienko, N. V. A mechanism for increased sensitivity of acute myeloid leukemia to mitotoxic drugs. Cell Death & Disease. 10 (8), 617 (2019).
- Caro, P., et al. Metabolic signatures uncover distinct targets in molecular subsets of diffuse large B cell lymphoma. Cancer Cell. 22 (4), 547-560 (2012).
- Lagadinou, E. D., et al. BCL-2 inhibition targets oxidative phosphorylation and selectively eradicates quiescent human leukemia stem cells. Cell Stem Cell. 12 (3), 329-341 (2013).
- Senft, D., Ronai, Z. A. Regulators of mitochondrial dynamics in cancer. Current Opinion in Cell Biology. 39, 43-52 (2016).
- Vazquez, F., et al. PGC1α expression defines a subset of human melanoma tumors with increased mitochondrial capacity and resistance to oxidative stress. Cancer Cell. 23 (3), 287-301 (2013).
- Caino, M. C., Altieri, D. C. Cancer cells exploit adaptive mitochondrial dynamics to increase tumor cell invasion. Cell Cycle. 14 (20), 3242-3247 (2015).
- Ralph, S. J., Rodríguez-Enríquez, S., Neuzil, J., Saavedra, E., Moreno-Sánchez, R. The causes of cancer revisited: "mitochondrial malignancy" and ROS-induced oncogenic transformation - why mitochondria are targets for cancer therapy. Molecular Aspects of Medicine. 31 (2), 145-170 (2010).
- Kreitz, J., et al. Metabolic Plasticity of Acute Myeloid Leukemia. Cells. 8 (8), (2019).
- Sriskanthadevan, S., et al. AML cells have low spare reserve capacity in their respiratory chain that renders them susceptible to oxidative metabolic stress. Blood. 125 (13), 2120-2130 (2015).
- Guerra, F., et al. Mitochondrial Dysfunction: A Novel Potential Driver of Epithelial-to-Mesenchymal Transition in Cancer. Frontiers in Oncology. 7, 295 (2017).
- Guerra, F., Arbini, A. A., Moro, L. Mitochondria and cancer chemoresistance. Biochimica et Biophysica Acta. 1858 (8), 686-699 (2017).
- Neuzil, J., Dong, L. F., Rohlena, J., Truksa, J., Ralph, S. J. Classification of mitocans, anti-cancer drugs acting on mitochondria. Mitochondrion. 13 (3), 199-208 (2013).
- Ubah, O. C., Wallace, H. M. Cancer therapy: Targeting mitochondria and other sub-cellular organelles. Current Pharmaceutical Design. 20 (2), 201-222 (2014).
- Yamaguchi, R., et al. Efficient elimination of cancer cells by deoxyglucose-ABT-263/737 combination therapy. PLoS One. 6 (9), 24102 (2011).
- Hahn, T., et al. Use of anti-cancer drugs, mitocans, to enhance the immune responses against tumors. Current Pharmaceutical Biotechnology. 14 (3), 357-376 (2013).
- Panina, S. B., Pei, J., Baran, N., Konopleva, M., Kirienko, N. V. Utilizing Synergistic Potential of Mitochondria-Targeting Drugs for Leukemia Therapy. Frontiers in Oncology. 10, 435 (2020).
- Lema, C., Varela-Ramirez, A., Aguilera, R. J. Differential nuclear staining assay for high-throughput screening to identify cytotoxic compounds. Current Cellular Biochemistry. 1 (1), 1-14 (2011).
- Döhner, H., et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 115 (3), 453-474 (2010).
- Heinz, S., et al. Mechanistic Investigations of the Mitochondrial Complex I Inhibitor Rotenone in the Context of Pharmacological and Safety Evaluation. Scientific Reports. 7, 45465 (2017).
- Fan, T., et al. Tumor Energy Metabolism and Potential of 3-Bromopyruvate as an Inhibitor of Aerobic Glycolysis: Implications in Tumor Treatment. Cancers (Basel). 11 (3), (2019).
- Pal, R., Mamidi, M. K., Das, A. K., Bhonde, R. Diverse effects of dimethyl sulfoxide (DMSO) on the differentiation potential of human embryonic stem cells. Archives of Toxicology. 86 (4), 651-661 (2012).
- Tunçer, S., et al. Low dose dimethyl sulfoxide driven gross molecular changes have the potential to interfere with various cellular processes. Scientific Reports. 8 (1), 14828 (2018).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved