A subscription to JoVE is required to view this content. Sign in or start your free trial.
A Low-Cost Method of Measuring the In Situ Primary Productivity of Periphyton Communities of Lentic Waters
In This Article
Summary
Presented here is a cost-effective and transportable method/facility for measuring the primary productivity of microbial mats under actual in situ environmental temperature and light conditions. The experimental setup is based on widely available materials and can be used under various conditions while offering the advantages of laboratory-based models.
Abstract
Measuring the in situ primary productivity of periphyton during the growing season gradient can elucidate the quantitative effect of environmental drivers (mainly phosphorus concentration and light intensity) and species composition on primary productivity. Primary productivity is mainly driven by light intensity, temperature, availability of nutrients, and distribution of the ionic species of the carbonate system in the respective depths of the euphotic zone. It is a complex system that is very difficult to simulate in the laboratory. This cheap, transportable, and easy-to-build floating barge allows measuring the primary productivity accurately-directly under the actual natural conditions. The methodology is based on measuring the primary productivity in real time using noninvasive oxygen sensors integrated into tightly sealed glass jars, enabling online oxygen flux monitoring and providing new insights into metabolic activities. Detailed seasonal in situ measurements of gross primary productivity of microbial mats (or other benthic organisms) can improve current knowledge of the processes controlling primary productivity dynamics in lentic waters.
Introduction
Primary productivity is the only entry of autochthonous carbon into the aquatic systems that form the entire system food web1. Hence, the accurate estimation of primary productivity is an essential step toward understanding the functioning of aquatic ecosystems. Littoral zones are areas of high primary productivity and biodiversity. In addition to phytoplankton, periphyton (hereafter referred to as microbial mats) and macroalgae are assumed to significantly contribute to primary productivity in littoral zones2. Due to their sessile lifestyle and significant spatial heterogeneity, quantification of primary productivity is not trivial.
Primary productivity is driven mainly by light intensity, temperature, availability of nutrients, and distribution of the ionic species of the carbonate system in the respective depths of euphotic zones3,4. The depth markedly influences the spatial distribution of microbial mats. Microbial communities must cope with the adverse effects of high irradiation and pronounced seasonal temperature variations in shallow depths and with lower light intensity at greater depths. In addition to the depth gradient, dynamic trophic interactions generate multiple and complex spatial patterns at different scales5. This complex system is complicated to simulate in the laboratory. The most accurate way to infer the metabolic activity of individual primary producers from littoral zones is to set up in situ experiments.
The methodology introduced in this paper is based on the traditional chamber method2,6,7, together with a transportable and easy-to-build low-cost floating barge. This allows the measurement of primary productivity at different depths under the natural light spectrum, temperature, and different distribution of the ionic species of the carbonate system with the depth. The method is based on the principle of light versus dark bottle oxygen, which was first employed to measure phytoplankton photosynthesis6 and is still commonly used6,7. It compares the rate of change in oxygen in bottles kept in the light (which includes the effects of primary productivity and respiration) with those held in the dark (respiration only)8. The method uses oxygen evolution (photosynthesis) as a proxy for primary productivity. The measured variables are net ecosystem productivity (NEP, as a change in O2 concentration over time in light conditions) and ecosystem respiration (RE, as a change in O2 concentration over time in the dark). Gross ecosystem productivity (GEP) is the calculation of the difference between the two (Table 1). The term "ecosystem" is used here to denote that the periphyton is composed of autotrophic and heterotrophic organisms. The most significant improvement of this traditional chamber method is using noninvasive oxygen optical sensors and optimization of this primarily planktonic method for measuring periphytic primary productivity.
The technique is described in the example of measuring microbial mats in the littoral zone of newly emerged post-mining lakes in the Czech Republic-Milada, Most, and Medar. The metabolic activity of microbial mats is determined using direct in situ measurement of O2 fluxes performed directly at specific depths, where the studied communities naturally occur. Heterotrophic and phototrophic activity is measured in closed glass bottles equipped with noninvasive optical oxygen sensors. These sensors detect the partial pressure of oxygen using the fluorescence of light-sensitive dyes. The bottles with microbial mats are suspended and incubated on a floating device at the appropriate depths. The oxygen concentration inside the bottles was continuously measured during the daylight period from the small boat.
Samples of intact microbial mats are collected and placed in gas-tight incubation bottles at designated depths by scuba divers. Each bottle is equipped with a noninvasive optical oxygen microsensor, which monitors the O2 productivity/consumption over time. All measurements are done in five replicate dark/light pairs in each depth. The temperature and photosynthetically active radiation (PHAR) intensities are measured at respective depths throughout the incubation. After 6 h of in situ incubation (daylight hours), the microbial mats are harvested from the bottles and dried. O2 fluxes are normalized to microbial biomass. As a control, fluxes are corrected for changes in O2 concentration in separate light and dark gas-tight bottles (blank controls) containing lake water without microbial mat biomass. Below are detailed instructions for building the floating barge and performing the whole experiment step-by-step. This paper also presents representative results from the measurements of microbial mats at two depths (1 m and 2 m), with five replicates at each depth. Actual temperature and light intensity were measured during the whole experiment using dataloggers.
Protocol
NOTE: Before sampling, determine the degree of replications based on the overall project needs, statistical design, or expected amount of sample variability.Five replicate pairs of light and dark incubation bottles are suggested for precise statistical analysis and to account for potential sample loss or breakage. The described floating experimental barge is designed to carry five replicates plus one pair of blank controls; see Figure 1 for a technical drawing of the experimental barge.
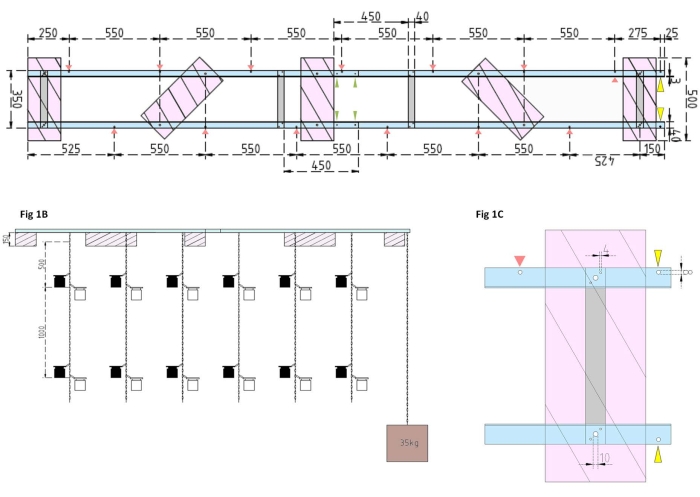
Figure 1: Technical drawings of the experimental barge and the side float. (A) Top view: the frame of the barge consists of four Aluminum angle L profile pieces (blue) that are joined together by four Aluminum flat bars (grey). XPS floats (pink) are mounted to the frame at two points, each one on the parallel aluminum pieces. Chains for incubation bottles are attached to the frame on both sides using snap hooks in predrilled holes (red arrows) with 550 mm of separation between them. Chains were provided with snap-hooks at 1 m and 2 m distances for incubation bottle attachment (choose the snap-hooks position according to the experimental depth). The concrete anchor is secured to the barge's bow, where an overhang of 25 mm allows for two predrilled holes (yellow arrowheads) to serve as an attachment point for the anchor's chain and research vessel. The frame is assembled or disassembled easily via the parallel joints between the four aluminum angle pieces (green arrowheads). (B) The side view shows the suspended chains with hanging incubation bottles and concrete anchor (brown square). (C) The side XPS float: Parallelaluminum angle L pieces (blue) are joined by vertical aluminum flat bars (grey). Below the crossbar section, the XPS float (pink) is mounted with the necessary hole sizes indicated (4 mm). The suspended chains are attached with snap hooks in 8 mm holes (red arrowhead). At the barge's bow, two 8 mm holes are drilled into the overhanging aluminum, one for securing the anchor to the barge (yellow arrowhead) and another for mooring the research vessel to the barge (blue). Please click here to view a larger version of this figure.
1. Construction of the experimental barge
NOTE: The floating barge consists of two equal sections mounted together, allowing easy assembly/disassembly. All used parts can be purchased at any hobby market or store selling building materials.
- First, assemble the frame of the barge by joining four aluminum angle L profile pieces (40 mm x 40 mm x 3 mm; length of 2,000 mm) together using four aluminum flat bars (40 mm x 3 mm x 350 mm), 16 screws (4 mm x 15 mm with hexagonal nuts), and 32 washers (4 mm x 10 mm).
NOTE: The distance and position of the flat bars are shown in the technical drawing in Figure 1A. The detailed attachment of the floats to the side flat bars is shown in Figure 1B. - To join the two equal sections of the frame, use four 4 mm x 15 mm screws with wing nuts and eight 4 mm x 10 mm washers to screw the aluminum angle L profiles together at the ends (Figure 1A, green arrows).
- Use five pieces of extruded polystyrene (XPS) material (500 mm x 200 mm x 150 mm), ten 10 mm x 170 mm screws with hexagonal nuts, and twenty 10 mm x 50 mm washers to prepare five extruded polystyrene floats (500 mm x 200 mm x 150 mm each). Attach the floats to the frame at five points shown in the technical drawing (Figure 1A).
- Drill holes into the frame (see red arrows in Figure 1A marking the positions and distances of the holes for the chains). Attach the 12 m steel chains (wire diameter of 3 mm, inside link of 5.5 mm x 26 mm) for the incubation bottles to the holes in the frame using steel carabine hooks (50 mm x 5 mm). Provide each chain with pairs of snap hooks (50 mm x 5 mm) for installing the incubation bottles to the desired depth according to the experimental design. In this case, they were seated at 1 m and 2 m depths.
- For the anchor, fill the 15 L bucket with concrete. Insert an eye bolt into the concrete and allow it to dry undisturbed. Fasten the 5 m steel chain to the hook. Secure the anchor to the predrilled hole on the barge's bow (marked by yellow arrows in Figure 1A,B).
NOTE: Technical drawings with the assembly description are shown in Figure 1A-C. Figure 2 shows the photo of the assembled experimental barge. Figure 3 shows the attachment of incubation bottles to the chain.
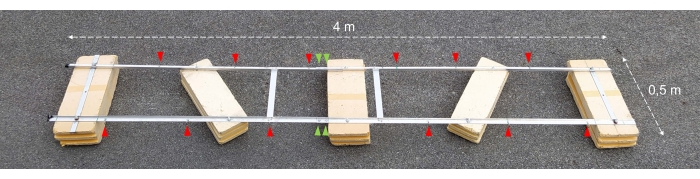
Figure 2: Assembled experimental barge. Photograph of the assembled experimental barge. Red arrowheads show the holes for the attachment of chains with incubation bottles. The green arrowheads point to where the two halves of the float are joined together. Please click here to view a larger version of this figure.

Figure 3: Incubation bottles. Photo of two pairs of dark and light incubation bottles hanging at a depth of 1 m. One pair of bottles contains the sample of intact microbial mats still growing on the stone (red arrowhead). The second one is the blank bottle with the lake water from the respective depth. A yellow arrowhead points to the oxygen sensor spot attached to the inner wall of the incubation bottle. Please click here to view a larger version of this figure.
2. Installation in the field
- Use of an inflatable kayak is suggested for barge placement and conducting experiments as it is easily transportable.
- Select a place with an ideal depth to anchor the float. Choose the depth so that the lower incubation bottles are at least 2 m above the bottom to avoid disturbing the sediment into the water column around the incubation bottles.
- Attach the assembled barge behind the stern of the boat. Carefully lower the anchor along the side of the boat and align it so that it hangs slightly below the water surface so that the float can be easily towed along with the anchor to the spot with the required depth.
- Untie the anchor from the boat, lower it to the bottom, and secure the barge to the anchor chain.
- Secure the chains for attaching the incubation bottles to the barge.
3. Incubation bottle preparation
- Use the transparent wide-necked 0.5 L bottles with gas-tight seals.
NOTE: It is possible to adjust the size of the bottles but remember to increase the floatation of the barge with more polystyrene sheets as well. The barge described here can reliably carry 24 0.5 L glass bottles. - Attach the oxygen optical sensor spots to the inner wall of each bottle.
- Add an opaque layer to the dark treatment bottles by wrapping them with black electrical tape.
- Cut a tiny hole in the spot with the optical sensor. To prevent light from entering the bottle, make the hole slightly smaller than the diameter of the sensor.
NOTE: Any opaque layer that prevents light from entering the bottle will also work. The advantage of black electrical tape is that it resists abrasion and does not peel off in water.
4. Sample collection and handling
NOTE: Divers carry out the manual collection of samples in deeper water. In shallow water, it can be done by snorkeling or wading.
- Place the incubation bottles in the portable box.
- Dive with the box to the respective depth. Avoid disturbing the sediment in the surrounding water.
- Fill the incubation bottles with the samples carefully. Try to disturb the biomass of the sample as little as possible, for example, by using long tweezers. If the microbial mats grow on a solid surface, such as a small stone, carefully transfer the whole stone with intact biomass into the bottle.
NOTE: Avoid collecting big stones when sampling mats grow on stones-the glass bottles can be broken during further manipulation. - Fill one pair of light/dark bottles with clean water from the respective depths to serve as blank controls.
NOTE: The bottles without the periphyton sample serve as a control determining the oxygen production/consumption of ambient water organisms. It ensures that the calculated net or gross primary productivity of periphyton is unbiased. - Ensure that the water in all incubation bottles is clean and contains no disturbing sediment.
- Close the bottles and bring them to the boat anchored to the floating barge.
5. Measuring the primary productivity
NOTE: The person sitting in the boat takes the box from the diver and performs the following steps.
- Attach the first two pairs of incubation bottles to the snap-hooks on the first chain.
- Measure the initial oxygen concentration in each bottle using the fiber-optic oxygen meter. Attach the optical cable of the meter to the oxygen sensor mounted inside the bottle and immediately (within a few seconds) read out the O2 concentration contactless (through the bottle wall). Record the measured value.
NOTE: The handling time is short; from taking the bottles from the divers to the initial setting to the respective depth, it takes just a few minutes. - Immediately afterward, carefully lower the chain with the attached bottles back into the water. Ensure that the incubation bottles are placed at the same depth as the biomass that was placed in them was sampled.
- Make another measurement from the ship after 1 h (see NOTE below). Carefully pull each chain with the bottles into the boat, read the oxygen value by attaching the optical cable to the sensor, and lower the samples into the water again.
NOTE: Adjust the time between individual measurements during the incubation according to the intensity of O2 productivity/consumption of samples to avoid oversaturation of bottles. - Repeat this procedure at least four or five times with all pairs of bottles.
NOTE: The whole setup of the experiment in the field is shown in Figure 4.
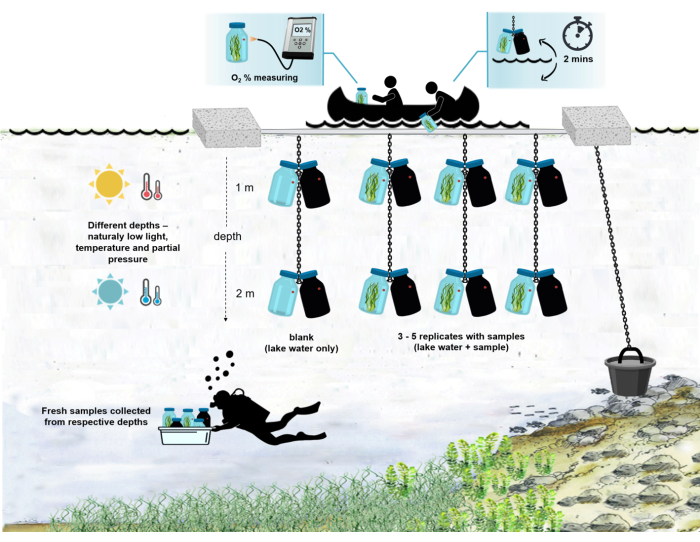
Figure 4: Schema of the experimental setup in the field. Illustration of the anchored experimental barge on the lake surface. The incubation bottles (0.5 L) with microbial mat biomass are hung at two different depths (1 m and 2 m). The divers collected samples of microbial mats directly into the incubation bottles at the appropriate depths. Oxygen concentration in individual bottles is measured from the ship. The bottles are pulled out of the water. The oxygen concentration value is measured in a few seconds by attaching an optical cable to the oxygen sensor. The bottles are then carefully lowered back into the water. The whole procedure of measuring two pairs of incubation bottles from two depths takes ~2 min. Please click here to view a larger version of this figure.
6. Sample analyses
- After the end of the measurements, take the samples directly from the bottles, and transfer the biomass of the microbial mat to the small plastic flasks. If the mats grow on solid substrates (e.g., stones), scrub them with a toothbrush or a small knife.
- In the laboratory, filter each replicate through pre-weighed glass fiber filters to determine the dry weight9.
7. Data analyses
- During the incubation period, measure the oxygen concentration in the light and dark bottles and compare it with the oxygen concentration in the water column when the bottles are filled.
NOTE: The change in oxygen in the light bottle over time is the combined result of gross ecosystem productivity (GEP) and respiration by all the organisms in the bottle (autotrophs and heterotrophs from the ambient water and the periphytic community). The decrease in oxygen in the dark bottle measures the respiratory losses of both autotrophs and heterotrophs. The change in oxygen concentration in the control (i.e., bottles without the periphyton) is only the product of heterotrophic or autotrophic organisms in the ambient water. Periphyton productivity and respiration were estimated by subtracting ambient water productivity and respiration measured in blank incubation bottles. - Note down the O2 concentration in percent oxygen saturation (i.e., calibrate the oxygen sensors to 0% and 100% oxygen saturation). Before estimating the primary productivity, convert the raw data (
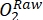 ) to some reasonable unit.
) to some reasonable unit.
NOTE: In this study, the data are converted to mmol of O2 per gram of organic matter (OM) of the periphyton mass at the dry weight basis, according to Benson and Krause10.- Calculate the conversion using equation (1):
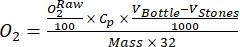 (1)
(1)
Where Cp is O2 concentration in water (mg (O2) L-1) when it is fully saturated by O2, VBottle is the volume of the bottle in mL, VStones is the volume occupied by the stone in mL, is the weight of the periphyton mass in g, and 32 is the molar weight of O2. - Calculate Cp using equation (2):
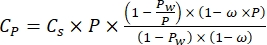 (2)
(2)
Where Cs is the standard O2 concentration, P is the atmospheric pressure at the lake surface, Pw is the partial pressure of the water vapor at the lake surface, and ω is water density. - Calculate Cs, ω, P, and Pw from previously defined empirical equations (3-6) when lake elevation (h in km) and water temperature at the lake surface (t in °C) are known:
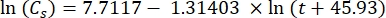 (3)
(3)
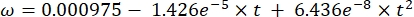 (4)
(4)
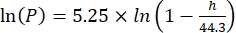 (5)
(5)
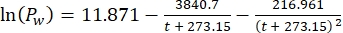 (6)
(6)
NOTE: From equations (1-6), it is evident that the calculated O2 concentration is most accurate for shallow depths. As the depth increases, the calculated concentration gets more biased in terms of absolute concentration. It is optimal when the rate of change of oxygen concentration along the depth is known for each lake so that the absolute O2 concentration can be corrected if necessary. Once the O2 concentration is calculated, its change over time can be used to calculate two different fluxes of O2 at two different conditions. Under light conditions, net ecosystem productivity (NEP) is directly proportional to the change in O2 concentration over time (see below). The term "ecosystem" is used here to denote that the periphyton is composed of autotrophic and heterotrophic organisms. In the dark, the change in O2 concentration over time is proportional to the sum of respiratory losses of autotrophic and heterotrophic organisms, thus defining ecosystem respiration (RE). The difference between NEP and RE defines gross ecosystem productivity (GEP). If the respiratory losses of the heterotrophic part of the community are negligible, GEP becomes equal to gross ecosystem productivity.
- Calculate the conversion using equation (1):
- Determine the rate of change of O2 concentration over time by third-degree polynomial regression, as shown in equation (7).
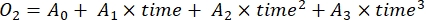 (7)
(7)
Where A0 is O2 concentration at time zero and A1-A2 are coefficients of polynomial regression.
NOTE: The polynomial function is used because it can serve as an approximation of any differential equation. Thus, it is not necessary to know the accurate functional relationship between O2 concentration and time. Therefore, any assumption associated with the functional relationship (e.g., linearity) does not need to be controlled. By definition, the second term of polynomial regression, A1 defines the rate of change of O2 concentration at time zero (i.e., instant rate), which is independent of A0 and, thus, the absolute O2 concentration at time zero. For that reason, the estimate of O2 flux is not affected by the bias in absolute O2 concentration calculations caused by pressure change across the depth gradient. A1 has units O2 (mmol g(OM)-1) per time (1 h in this study).- Calculate A1, calculated separately for bottles with and without exposure to light and containing microbial mat biomass (i.e., VStones > 0) and for control bottles with and without exposure to light and containing free water (i.e., VStones = 0).
NOTE: Assigning these different regression coefficients notations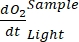 ,
, 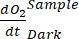 ,
, 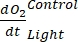 , and
, and 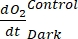 , respectively, the equation (8) for productivity GEP calculation can be written as:
, respectively, the equation (8) for productivity GEP calculation can be written as:
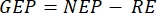 (8.1)
(8.1)
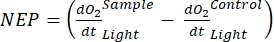 (8.2)
(8.2)
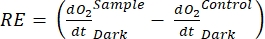 (8.3)
(8.3)
The term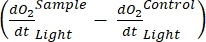 defines net ecosystem productivity (Figure 5A; i.e., net oxygen productivity ), and the term
defines net ecosystem productivity (Figure 5A; i.e., net oxygen productivity ), and the term 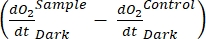 represents the sum of autotrophic and heterotrophic respiration (Figure 5B; RE, i.e., assuming both respirations are similar under dark and light conditions).
represents the sum of autotrophic and heterotrophic respiration (Figure 5B; RE, i.e., assuming both respirations are similar under dark and light conditions). - Subtract R from NEP to obtain GEP (Figure 5C).
NOTE: Equation (8) implicitly assumes that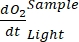 and
and 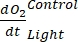 are both positive, and
are both positive, and 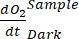 and
and 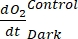 are both negative. If
are both negative. If 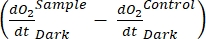 is positive, check the raw data carefully for outliers .
is positive, check the raw data carefully for outliers . 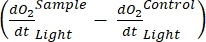 can be theoretically negative because the respiratory losses caused by heterotrophic activity can be higher than photosynthetic activity.
can be theoretically negative because the respiratory losses caused by heterotrophic activity can be higher than photosynthetic activity.
- Calculate A1, calculated separately for bottles with and without exposure to light and containing microbial mat biomass (i.e., VStones > 0) and for control bottles with and without exposure to light and containing free water (i.e., VStones = 0).
Results
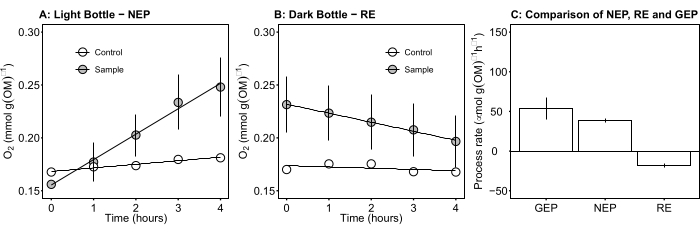
Figure 5: Net and gross ecosystem productivity of microbial mats during daylight. (A) Light bottle-net ecosystem productivity: time course data of net oxygen productivity of microbial mats from the light bottles. The oxygen concentration change in incubation bottles was measured after 1 h during daylight. Grey circles: bottles with samples of m...
Discussion
The methodology described in this paper is based on the principle of the light and dark bottle oxygen technique in combination with the noninvasive technique of measuring O2 concentration using optical oxygen sensors. This system allows the parallel measurement of different incubation settings as the optical fiber for measuring O2 can be moved quickly from bottle to bottle. The benthic communities from various depths can differ in taxonomic composition and productivity; simultaneously measuring them...
Disclosures
The authors confirm that they have no conflicts of interest to disclose.
Acknowledgements
This study was supported by the Czech Science Foundation (GACR 19-05791S), RVO 67985939, and by the CAS within the program of the Strategy AV 21, Land save and recovery. Many thanks to Ondřej Sihelský for taking the shots in the field - without him, the filming would have been complete hell. The project would not be possible without tight cooperation with companies, Palivový Kombinát Ústí s.p. and Sokolovská Uhelná, who provided access to the studied localities.
Materials
| Name | Company | Catalog Number | Comments |
| Aluminum angle L profile 40 x 40 mm x 3 mm, length 2,000 mm | |||
| Aluminum flat bar 40 x 3 x 350 mm | |||
| Bucket 15 L with concrete infill | |||
| Carabine hook with screw lock 50 x 5 mm | |||
| electric tape black | |||
| Extruded polystyrene (XPS) material 500 x 200 x 150 mm | |||
| Fibox 3 LCD trace | PreSens Precision Sensing GmbH | stand-alone fiber optic oxygen meter | |
| Hondex PS-7 Portable Depth Sounder | Hondex - Honda Electronics | to measures distances through water - to bottom depth measurement; https://www.honda-el.net/industry/ps-7e | |
| KORKEN - glass tight-seal jar 0.5 L | IKEA | incubation bottles; https://www.ikea.com/cz/en/p/korken-jar-with-lid-clear-glass-70213545/ | |
| metal hook | |||
| Oxygen Sensor Spot SP-PSt3-NAU-D5 | PreSens Precision Sensing GmbH | non-invasive optical oxygen sensor for measurements under Real Conditions | |
| SCOUT infantable canoe | GUMOTEX | https://www.gumotexboats.com/en/scout-standard#0000-044667-021-13/11C | |
| Screw 10 x 170 mm with hexagonal nuts | |||
| Screw 4 x 15 mm with hexagonal nuts | |||
| Screw 4 x 15 mm with wing nuts | |||
| Snap hooks 50 x 5 mm | |||
| Steel Carabine hook 50 x 5 mm | |||
| Steel chain with wire diameter 3 mm, inside link 5.5 x 26 mm | |||
| Steel chain, 5 m | |||
| toothbrush | |||
| tweezer | |||
| Washer 10 x 50 mm | |||
| Washer 4 x 10 mm | |||
| Washer 4 x 10 mm |
References
- Blachart, J. L., et al. Potential consequences of climate change for primary production and fish production in large marine ecosystems. Philosophical Transactions of the Royal Society B: Biological Sciences. 367 (1605), 2979-2989 (2012).
- Howarth, R. W., Michaels, A. F., Sala, O. E., Jackson, R. B., Mooney, H. A., Howarth, R. W. The Measurement of primary production in aquatic ecosystems. Methods in Ecosystem Science. , 72-85 (2000).
- Vadenbecouer, Y. E. G., Peterson, M. J., Vander, Z., Kalff, J. Benthic algal production across lake size gradients: Interactions among morphometry, nutrients, and light. Ecology. 89 (9), 2542-2552 (2008).
- Reimer, A., Landmann, G., Kempe, S. Lake Van, eastern Anatolia, hydrochemistry and history. Aquatic Geochemistry. 15 (1), 195-222 (2009).
- Cantonati, M., Lowe, R. L. Lake benthic algae: toward an understanding of their ecology. Freshwater Sciences. 33 (2), 475-486 (2014).
- Gaarder, T., Gran, H. H. Investigation of the production of plankton in the Oslo Fjord. Rapports et Proces-verbaux des Réunions. Conseil International pour l'Éxploration de la Mer. 42, 1-48 (1927).
- Hall, R. O., Thomas, S., Gaiser, E. E., Fahey, T. J., Knapp, A. K. Measuring Freshwater Primary Productivity and Respiration. Principles and Standards for Measuring Primary Productivity. , (2007).
- Howart, R., Michaels, A. Chapter 6 The Measurement of Primary Production in Aquatic Ecosystems. Springer Science and Business Media LLC. , (2000).
- Kopáček, J., Hejzlar, J. Semi-micro determination of total phosphorus in soils, sediments, and organic materials: a simplified perchloric acid digestion procedure. Communications in Soil Science and Plant Analysis. 26 (11-12), 1935-1946 (1995).
- Benson, B. B., Krause, D. The concentration and isotopic fractionation of oxygen dissolved in freshwater and seawater in equilibrium with the atmosphere1. Limnology and Oceanography. 29 (3), 620-632 (1984).
- Dodds, W. K., Biggs, B. J., Lowe, R. L. Photosynthesis-irradiance patterns in benthic microalgae: variations as a function of assemblage thickness and community structure. Journal of Phycology. 35 (1), 42-53 (1999).
- Bott, T. L., et al. An evaluation of techniques for measuring periphyton metabolism in chambers. Canadian Journal of Fisheries and Aquatic Sciences. 54 (3), 715-725 (1997).
- Blankenship, R. E. Structural and functional dynamics of photosynthetic antenna complexes. Proceedings of the National Academy of Sciences. 112 (45), 13751-13752 (2015).
- Hawes, I., Schwartz, A. -. M. Photosynthesis in an extreme shade environment, benthic microbial mats from Lake Hoare, a permanently ice-covered Antarctic lake. Journal of Phycology. 35 (3), 448-459 (1999).
- Aristegui, J., et al. Planktonic primary production and microbial respiration measured by 14C assimilation and dissolved oxygen changes in coastal waters of the Antarctic peninsula during austral summer: Implications for carbon flux studies. Marine Ecology-Progress Series. 132, 191-201 (1996).
- Steemann-Nielsen, C. The use of radioactive carbon (14C) for measuring organic production in the sea. Journal of Experimental Marine Biology and Ecology. 18 (2), 117-140 (1952).
- Sanz-Martín, M., et al. Relationship between carbon-and oxygen-based primary productivity in the Arctic Ocean, svalbard archipelago. Frontiers in Marine Science. 6, 468 (2019).
- Nielsen, E. S. Measurement of the production of organic matter in the sea by means of carbon-14. Nature. 167 (4252), 684-685 (1951).
- Jönsson, B. A 14C-incubation technique for measuring microphytobenthic primary productivity in intact sediment cores. Limnology and Oceanography. 36 (7), 1485-1492 (1991).
- Bender, M. L., et al. A comparison of four methods for determining planktonic community production. Limnology and Oceanography. 32 (5), 1085-1098 (1987).
- Šimek, K., et al. Spatio-temporal patterns of bacterioplankton productivity and community composition related to phytoplankton composition and protistan bacterivory in a dam reservoir. Aquatic Microbial Ecology. 51 (3), 249-262 (2008).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved