A subscription to JoVE is required to view this content. Sign in or start your free trial.
Advances in Nanoscale Infrared Spectroscopy to Explore Multiphase Polymeric Systems
In This Article
Summary
This protocol describes the application of atomic force microscopy and nanoscale infrared spectroscopy to evaluate the performance of photothermal nanoscale infrared spectroscopy in the characterization of three-dimensional multi-polymeric samples.
Abstract
Multiphase polymeric systems encompass local domains with dimensions that can vary from a few tens of nanometers to several micrometers. Their composition is commonly assessed using infrared spectroscopy, which provides an average fingerprint of the various materials contained in the volume probed. However, this approach does not offer any details on the arrangement of the phases in the material. Interfacial regions between two polymeric phases, often in the nanoscale range, are also challenging to access. Photothermal nanoscale infrared spectroscopy monitors the local response of materials excited by infrared light with the sensitive probe of an atomic force microscope (AFM). While the technique is suitable for interrogating small features, such as individual proteins on pristine gold surfaces, the characterization of three-dimensional multicomponent materials is more elusive. This is due to a relatively large volume of material undergoing photothermal expansion, defined by the laser focalization onto the sample and by the thermal properties of the polymeric constituents, compared to the nanoscale region probed by the AFM tip. Using a polystyrene (PS) bead and a polyvinyl alcohol (PVA) film, we evaluate the spatial footprint of photothermal nanoscale infrared spectroscopy for surface analysis as a function of the position of PS in the PVA film. The effect of the feature position on the nanoscale infrared images is investigated, and spectra are acquired. Some perspectives on the future advances in the field of photothermal nanoscale infrared spectroscopy are provided, considering the characterization of complex systems with embedded polymeric structures.
Introduction
Atomic force microscopy (AFM) has become essential to image and characterize the morphology of a wide variety of samples with nanoscale resolution1,2,3. By measuring the deflection of an AFM cantilever resulting from the interaction of the sharp tip with the sample surface, nanoscale functional imaging protocols for local stiffness measurements and tip-sample adhesion have been developed4,5. For soft condensed matter and polymer analysis, AFM measurements exploring the nanomechanical and nanochemical properties of local domains are highly sought after6,7,8. Before the emergence of nanoscale infrared (nanoIR) spectroscopy, AFM tips were chemically modified to assess the presence of different domains from the AFM force curve and deduct the nature of the tip-sample interaction. For instance, this approach was used to unveil the transformation of microdomains of poly(tert-butyl acrylate) at the surface of cyclohexane-treated polystyrene-block-poly(tert-butyl acrylate)block copolymer thin films at the sub 50 nm level9.
The combination of infrared (IR) light with AFM has had a significant impact on the field of polymer science6. Conventional IR spectroscopy is a widely used technique for studying the chemical structure of polymeric materials10,11, but it fails to provide information on individual phases and interphase behavior, as the regions are too small compared to the size of the IR beam used to probe the sample. The problem holds with IR microspectroscopy, as it is restricted by the optical diffraction limit6. Such measurements average the contributions of the entire region excited by the IR light; the signals resulting from the presence of nanoscale phases inside the probed region either exhibit complex fingerprints that should be deconvoluted during post-processing or are lost due to a signal level below the detectable level. Hence, it is essential to develop tools capable of nanoscale spatial resolution and high IR sensitivity to explore nanoscale chemical features in complex media.
Schemes to achieve nanoIR spectroscopy have been developed, first using a metallic AFM tip as a nanoantenna12,13, and more recently exploiting the AFM cantilever's ability to monitor changes in the photothermal expansion incurred during IR illumination of the sample12,14,15. The latter uses a pulsed, tunable IR light source tuned to an absorption band of the material probed, which causes the sample to absorb radiation and undergo photothermal expansion. This approach is well-suited for organic and polymeric materials. The pulsed excitation makes the effect detectable by the AFM cantilever in contact with the sample surface in the form of an oscillation. The amplitude of one of the contact resonances of the system observed in the frequency spectrum is then monitored as a function of illumination wavelength, which constitutes the nanoIR absorption spectrum of the material beneath the AFM tip15. The spatial resolution of nanoIR imaging and spectroscopy is limited by various effects of the photothermal expansion of the material. It has been evaluated that photothermal nanoIR spectroscopy using contact mode AFM can acquire the vibrational absorption spectra properties of materials with sub 50 nm scale spatial resolution14, with recent work demonstrating the detection of monomers and dimers of α-synuclein16,17. However, quantitative studies of the performance of nanoIR measurements on heterogeneous polymeric materials assembled in various configurations, such as the case of absorbers of finite dimensions embedded in the volume of various polymeric films, remain limited.
This article aims to create a polymeric assembly with an embedded feature of a known dimension to evaluate the sensitivity of photothermal expansion and spatial resolution of nanoIR during surface analysis. The protocol covers the preparation of a polyvinyl alcohol (PVA) polymer thin film on a silicon substrate and the placement of a three-dimensional polystyrene (PS) bead onto or embedded in the PVA film, which constitutes the formation of the model system. NanoIR imaging and spectroscopy measurements are described in the context of evaluating the signals generated by the same PS bead positioned on or beneath the PVA film. The influence of the bead position on the nanoIR signals is evaluated. Methods to assess the spatial footprint of the bead in the nanoIR map are discussed, and the effects of several parameters are considered.
Protocol
1. Making polyvinyl alcohol (PVA) solution
- Measure water and PVA polymer pellets (see Table of Materials) to create a 10 mL solution at a 20% PVA to water ratio by weight.
- Heat the water in the glassware over a hot plate set to 100 °C.
- Place the PVA polymer pellets into the heated water. Insert a magnetic stir bar.
- Reduce the heat to 80 °C and stir until the PVA fully dissolves.
- Cover the top of the glassware to prevent contamination.
- Once fully dissolved, place the 20% PVA solution into an appropriate storage container for storage at room temperature.
2. Preparing PVA-coated silicon (Si) wafers
- Cut a silicon (Si) wafer (see Table of Materials) into ~10 x 10 mm2 squares.
- Clean the Si substrate using isopropyl alcohol and let it dry.
- Place the clean Si wafer on the chuck of the spin coater (see Table of Materials).
- Drop cast approximately 10 µL of PVA solution onto the center of the Si substrate. Try to avoid bubble formation.
- Coat the Si substrate with a uniform PVA film by spin coating for 30 s at 1,500 rotations per minute (rpm).
NOTE: The specified volume of liquid and spin coating parameters create a uniform layer of PVA across the surface of the substrate with sufficient thickness to prevent rapid drying between the spin coating and placing the PS beads onto the PVA surface in the next step.
- Remove the sample from the spin coater and place it into a clean sample container to prevent contamination before transferring the PS beads.
3. Placing the PS beads onto the PVA-coated surface
- Clean a Si substrate using isopropyl alcohol and let it dry.
- Using a pipette, place 1 µL of PS beads suspended in water onto the center of the substrate.
- Let the water evaporate by placing the sample in a storage compartment containing bentonite clay desiccant.
NOTE: This step preserves the sample by reducing exposure to humidity. - Place the PVA-coated substrate (step 2.4) and the substrate with the dried PS beads (step 3.3) under an optical microscope. Depending on their size, a single bead will be visible using simple binoculars or will require higher optical magnification.
- Gently loosen the beads using ultrafine tweezers (see Table of Materials). Use a fine hair paintbrush to collect a few loose beads and lightly tap the hairs of the paintbrush over the freshly PVA-coated wafer. Multiple sweeps should allow the beads to accumulate within the hairs of the brush. Tap the top of the paintbrush hairs to disturb the PS bead powder to release beads onto the tacky PVA surface.
NOTE: It is important that the paintbrush is of high quality and clean to avoid releasing fibers and contaminants onto the PVA film's surface. Moving quickly during this step is essential so that the PVA does not completely dry. - Repeat this step until it is confirmed by optical microscopy inspection that individual PS beads adhere to the PVA surface.
- Store the sample in a clean container. Allow the sample to dry fully.
NOTE: The sample should be allowed to cool and dry completely before further analysis attempts are performed. AFM height measurements or surface profiler measurements can assess the thickness of successive PVA films.
4. Loading the sample for AFM characterization
NOTE: The protocol described is based on standard operating procedures of a nanoIR2 (see Table of Materials) platform, but should be adapted according to the AFM model used for the measurement.
- Mount the PVA and PS bead sample onto the AFM stage using a metallic AFM disk and adhesive tabs, so that the sample is firmly attached to the sample holder.
- Mount a nanoIR probe (e.g., FORTGG) onto the AFM probe holder.
NOTE: The AFM cantilever is 225 µm long, 27 µm wide, and 2.7 µm thick, with a tip radius of less than 10 nm. The cantilever is coated with 45 nm thick gold film on both sides to limit its response to the top-side IR illumination of the sample (see Table of Materials). For nanoIR spectroscopy measurements, preferably use a cantilever that has been stored in a polydimethylsiloxane-free environment prior to use. - Align the read-out laser at the free end of the cantilever beam by turning the laser alignment knobs (x and y adjustments of the laser position and vertical adjustment of the detector position).
- Maximize the SUM signal of the detector.
- Adjust the position of the detector by turning the deflection knob so that the laser is aligned with the center of the position-sensitive detector of the AFM read-out system, corresponding to a vertical deflection signal of ~0 V.
- Click on the Load icon in the AFM "Probe" control panel.
- Follow the prompts within the wizard screen. Use the focus arrows to determine the focal plane of the nanoIR cantilever. Use the XY-displacement controls to position the cantilever in the center of the screen (aligned with the white cross).
- Next, click on the focus arrows to find the focal plane of the surface of the sample.
- Use the optical view of the system and the XY-displacement controls to position the cantilever tip above the bead of interest and click on Next.
- On the engage screen, set the "standoff" to 50 µm and click on Approach Only.
- Initiate the Engage procedure to approach the tip for imaging.
5. Creating topographical and nanoIR images of the multipolymer sample
- Acquire topography images in standard "Contact Mode". Once the position of the cantilever with respect to the PS bead is set, initiate the approach by clicking on the engage icon in the AFM "Probe" control panel. An engage setpoint of a deflection differential of 0.2 V is used for the entire study here, corresponding to a force of ~100 nN.
In the AFM "Scan" control panel, set the scan rate to 0.8 Hz, scan size (height and width), and the number of points per line and number of lines per image to use for imaging (512 x 512 was used here). Click on Scan to acquire the topography image.
NOTE: Calibration of the cantilever18 is done by determining the deflection sensitivity (in nm/V) from the slope of the deflection-distance curve obtained with the cantilever interacting with a sapphire substrate (Supplementary Figure 1A). The cantilever stiffness is determined from thermal tuning19 (Supplementary Figure 1B). The resonance of the cantilever is fitted using a Lorentzian function. The cantilever stiffness (in N/m) is determined using the equipartition theorem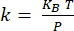 , where KB is the Boltzmann constant, T is the temperature (T = 295K), and P is the area of the power spectrum of the thermal fluctuations of the cantilever determined by integrating the Lorentzian fit of the thermal tuning data20.
, where KB is the Boltzmann constant, T is the temperature (T = 295K), and P is the area of the power spectrum of the thermal fluctuations of the cantilever determined by integrating the Lorentzian fit of the thermal tuning data20. - For nanoIR measurements, position the AFM tip on the feature of interest identified from the topography image.
- Select the tuning fork icon in the nanoIR control panel to determine the contact resonance frequencies of the cantilever. Set an illumination wavenumber that will excite photothermal expansion in the material. Set a range of laser pulse frequency to sweep and set the duty cycle of the nanoIR laser. Select Acquire within the "Laser Pulse Tune Window".
- Select the second contact resonance of the tip-sample system for nanoIR measurements by positioning the marker bar (green vertical line) at the peak of the second contact resonance.
NOTE: The selection of the contact resonance mode can vary depending on the type of cantilever and sample. - Click on Optimize to align the center of the IR laser focal region with the position of the cantilever tip. Alignment is done at a selected IR illumination wavenumber, corresponding to an absorption band of the material probed. The cantilever is positioned at the center of the laser footprint (Supplementary Figure 2).
NOTE: Alignment can vary for different wavenumbers depending on the laser model. - Acquire a background of the IR laser illumination. This consists of measuring the output of the IR quantum cascade laser (QCL) in the wavelength range of emission at the pulse frequency selected (Supplementary Figure 3). This is important for background correction of the nanoIR spectra.
- Acquire the nanoIR spectrum by selecting the wavenumber range (here, Start and Stop are set to 1,530 cm-1 and 1,800 cm-1, respectively), the step size (2 cm-1), and the number of averages used for the measurement. Perform background correction of the spectra displayed by dividing the photothermal amplitude measured by the attenuated background, which consists of multiplying the background collected in step 5.2.4 by the percentage of power selected for the measurement.
- For nanoIR imaging, select the region of interest for imaging.
- Enable phase-locked loop (PLL) auto-tune in the "Laser Pulse Tune Window" (accessed by the Tuning fork icon).
- Adjust the minimum and maximum frequency to create a sweep range centered at the second resonance mode in the general control panel.
- Zero the phase by clicking on zero in the PLL control panel and then click on OK in the "Laser Pulse Tune window".
- Select IR Imaging Enabled by putting a checkmark in the box within the nanoIR control panel.
- In the "Imaging View" control panel, choose Height (Imaging View 1), Amplitude 2 (Imaging View 2), and Phase 2 (Imaging View 3) to acquire the topographical and chemical images of the sample. Set the acquisition direction to Trace (or Retrace). A line fit Line is often required to observe the topography image of the sample being acquired. Capture fit should be set to None.
NOTE: Scanning preferences, such as scan direction captured or color palette used, can be adjusted as needed. - In the AFM "Scan" control panel, select the Scan icon.
- To save the image, select the Now or End of frame icon in the "Capture" control panel.
- To export the data, right-click on the image or spectrum file names within the data lists. Select Export and choose the format of the file to export. Save the file in the desired computer folder location.
Results
PS ((C8H8)n) beads were deposited on a clean Si substrate (Figure 1A) and on PVA ((CH2CHOH)n) (Figure 1B,C). Due to the poor adhesion of the bead on Si, nanoIR imaging in contact mode could not be acquired for this sample. Instead, using the optical view of the sample on nanoIR, the gold-coated AFM probe was engaged on top of the PS bead in contact mode, with an estimated force of about 100 ...
Discussion
AFM combined with nanoIR spectroscopy can provide nanoscale chemical information using a cantilever in contact mode and a pulsed tunable IR light source. Model systems, such as embedding an absorber with finite dimensions in the volume of a polymeric material, are important to improve the understanding of image formation mechanisms and to determine the performance of the tool. In the case of the PS/PVA configuration presented here, optimization was carried out to obtain a stable PS bead positioned above or below the surf...
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Science Foundation (NSF CHE-1847830).
Materials
| Name | Company | Catalog Number | Comments |
| 10|0 2200 Golden Taklon Round | Zem | ||
| 5357-8NM Tweezers | Pelco | ||
| Adhesive Tabs | Ted Pella | 16079 | |
| AFM metal specimen disks | Ted Pella | 16208 | |
| Binocular | AmScope | ||
| Cantilever for nanoIR measurements | AppNano | FORTGG | |
| Cell culture dishes | Greiner bio-one GmbH | ||
| Desiccator | |||
| Floating optical table | Newport | RS 4000 | |
| Hotplate | VWR | ||
| Isopropanol | |||
| Kimwipes | KIMTECH | ||
| Magnetic stir bar | |||
| Microparticles based on polystyrene size: 5 µm | SIGMA-ALDRICH | 79633 | |
| nanoIR2 microscope | Bruker | Contact mode NanoIR2 | |
| Nitrogen Tank | Airgas | ||
| Petri dishes | Greiner bio-one GmbH | ||
| Polyvinyl Alcohol | SIGMA-ALDRICH | 363170 | this polymer was only 87%-89% hydrolyzed, which explains the presence of residual C=O at 1730 cm-1 |
| Quantum Cascade Laser | Daylight Solutions | 1550-1800 cm-1 range | |
| Silicon wafer | MEMC St. Peters | #901319343000 | |
| Spin coater | Oscilla |
References
- Dufrêne, Y. F., Viljoen, A., Mignolet, J., Mathelié-Guinlet, M. AFM in cellular and molecular microbiology. Cellular Microbiology. 23 (7), e13324 (2021).
- Sharma, A., Rout, C. S. Probe-based techniques for 2D layered materials. Advanced Analytical Techniques for Characterization of 2D Materials. , 1-14 (2022).
- Zhong, J., Yan, J. Seeing is believing: atomic force microscopy imaging for nanomaterial research. RSC Advances. 6 (2), 1103-1121 (2016).
- Olubowale, O. H., et al. 34;May the force be with you!" Force-volume mapping with atomic force microscopy. ACS Omega. 6 (40), 25860-25875 (2021).
- Dokukin, M. E., Sokolov, I. Quantitative mapping of the elastic modulus of soft materials with HarmoniX and PeakForce QNM AFM modes. Langmuir. 28 (46), 16060-16071 (2012).
- Nguyen-Tri, P., et al. Recent applications of advanced atomic force microscopy in polymer science: a review. Polymers. 12 (5), 1142 (2020).
- Vitry, P., et al. Mode-synthesizing atomic force microscopy for 3D reconstruction of embedded low-density dielectric nanostructures. Nano Research. 8 (7), 2199-2205 (2015).
- Coste, R., Pernes, M., Tetard, L., Molinari, M., Chabbert, B. Effect of the interplay of composition and environmental humidity on the nanomechanical properties of hemp fibers. ACS Sustainable Chemistry & Engineering. 8 (16), 6381-6390 (2020).
- Schönherr, H., Feng, C. L., Tomczak, N., Vancso, G. J. Compositional mapping of polymer surfaces by chemical force microscopy down to the nanometer scale: reactions in block copolymer microdomains. Macromolecular Symposia. 230 (1), 149-157 (2005).
- Holland-Moritz, K., Siesler, H. W. Infrared spectroscopy of polymers. Applied Spectroscopy Reviews. 11 (1), 1-55 (1976).
- Bhargava, R., Wang, S. Q., Koenig, J. L. FTIR microscpectrscopy of polymeric systems. Liquid Chromatography/FTIR Microspectroscopy/Microwave Assisted Synthesis. , 137-191 (2003).
- Rao, V. J., et al. AFM-IR and IR-SNOM for the characterization of small molecule organic semiconductors. The Journal of Physical Chemistry C. 124 (9), 5331-5344 (2020).
- Wang, H., Wang, L., Xu, X. G. Scattering-type scanning near-field optical microscopy with low-repetition-rate pulsed light source through phase-domain sampling. Nature Communications. 7 (1), 13212 (2016).
- Dazzi, A., Prater, C. B. AFM-IR: Technology and applications in nanoscale infrared spectroscopy and chemical imaging. Chemical Reviews. 117 (7), 5146-5173 (2017).
- Mathurin, J., et al. Photothermal AFM-IR spectroscopy and imaging: Status, challenges, and trends. Journal of Applied Physics. 131 (1), 010901 (2022).
- Miller, A., et al. Enhanced surface nanoanalytics of transient biomolecular processes. Science Advances. 9 (2), 3151 (2023).
- Emin, D., et al. Small soluble α-synuclein aggregates are the toxic species in Parkinson's disease. Nature Communications. 13 (1), 5512 (2022).
- Jardine, K., Dove, A., Tetard, L. AFM force measurements to explore grain contacts with relevance for planetary materials. The Planetary Science Journal. 3 (12), 273 (2022).
- Sader, J. E., Chon, J. W. M., Mulvaney, P. Calibration of rectangular atomic force microscope cantilevers. Review of Scientific Instruments. 70 (10), 3967-3969 (1999).
- Higgins, M. J., et al. Noninvasive determination of optical lever sensitivity in atomic force microscopy. Review of Scientific Instruments. 77 (1), 013701 (2006).
- Smith, B. The infrared spectra of polymers III: hydrocarbon polymers. Spectroscopy. 36 (11), 22-25 (2021).
- Korbag, I., Mohamed Saleh, S. Studies on the formation of intermolecular interactions and structural characterization of polyvinyl alcohol/lignin film. International Journal of Environmental Studies. 73 (2), 226-235 (2016).
- Tetard, L., Passian, A., Farahi, R. H., Thundat, T., Davison, B. H. Opto-nanomechanical spectroscopic material characterization. Nature Nanotechnology. 10 (10), 870-877 (2015).
- Bai, Y., Yin, J., Cheng, J. -. X. Bond-selective imaging by optically sensing the mid-infrared photothermal effect. Science Advances. 7 (20), (2021).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved