A subscription to JoVE is required to view this content. Sign in or start your free trial.
Preparation and Immunofluorescence Staining of Bundles and Single Fiber Cells from the Cortex and Nucleus of the Eye Lens
In This Article
Summary
This protocol describes methods to prepare peripheral, mature, and nuclear eye lens fiber cells for immunofluorescence staining to study complex cell-to-cell interdigitations and the membrane architecture.
Abstract
The lens is a transparent and ellipsoid organ in the anterior chamber of the eye that changes shape to finely focus light onto the retina to form a clear image. The bulk of this tissue comprises specialized, differentiated fiber cells that have a hexagonal cross section and extend from the anterior to the posterior poles of the lens. These long and skinny cells are tightly opposed to neighboring cells and have complex interdigitations along the length of the cell. The specialized interlocking structures are required for normal biomechanical properties of the lens and have been extensively described using electron microscopy techniques. This protocol demonstrates the first method to preserve and immunostain singular as well as bundles of mouse lens fiber cells to allow the detailed localization of proteins within these complexly shaped cells. The representative data show staining of the peripheral, differentiating, mature, and nuclear fiber cells across all regions of the lens. This method can potentially be used on fiber cells isolated from lenses of other species.
Introduction
The lens is a clear and ovoid tissue in the anterior chamber of the eye that is made up of two cell types, epithelial and fiber cells1 (Figure 1). There is a monolayer of epithelial cells that covers the anterior hemisphere of the lens. Fiber cells are differentiated from epithelial cells and make up the bulk of the lens. The highly specialized fiber cells undergo an elongation, differentiation, and maturation programming, marked by distinct changes in cell membrane morphology from the lens periphery to the lens center2,3,4,5,6,7,8,9,10,11,12, also known as the lens nucleus. The function of the lens to fine-focus light coming from various distances onto the retina depends on its biomechanical properties, including stiffness and elasticity13,14,15,16,17,18,19. The complex interdigitations of lens fibers have been hypothesized20,21 and recently shown to be important for lens stiffness22,23.
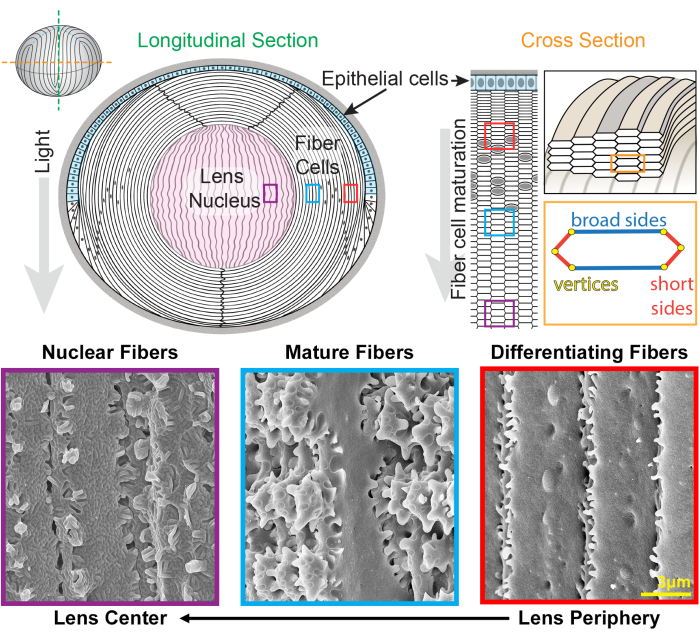
Figure 1: Lens anatomy diagrams and representative scanning electron microscopy (SEM) images from lens fibers. The cartoon shows a longitudinal (anterior to posterior from top to bottom) view of the anterior monolayer of epithelial cells (shaded in light blue) and a bulk mass of lens fiber cells (white). The center of the lens (shaded in pink) is known as the nucleus and comprises highly compacted fiber cells. On the right, a cross-section cartoon reveals the elongated hexagon cell shape of lens fibers that are packed into a honeycomb pattern. Fiber cells have two broad sides and four short sides. Representative SEM images along the bottom show the complex membrane interdigitations between lens fiber cells at different depths of the lens. From the right, newly formed lens fibers at the lens periphery have small protrusions along the short sides and balls-and-sockets along the broad side (red boxes). During maturation, lens fibers develop large paddle domains that are decorated by small protrusions along the short sides (blue boxes). Mature fiber cells possess large paddle domains illustrated by small protrusions. These interlocking domains are important for lens biomechanical properties. Fiber cells in the lens nucleus have fewer small protrusions along their short sides and have complex tongue-and-groove interdigitations (purple boxes). The broad sides of the cell display a globular membrane morphology. The cartoon was modified from22,32 and not drawn to scale. Scale bar = 3 µm. Please click here to view a larger version of this figure.
The lens grows by adding shells of new fiber cells overlaid on top of previous generations of fibers24,25. Fiber cells have an elongated, hexagonal cross section shape with two broad sides and four short sides. These cells extend from the anterior to the posterior pole of the lens, and depending on the species, the lens fibers can be several millimeters in length. To support the structure of these elongated and skinny cells, specialized interdigitations along the broad and short sides create interlocking structures to maintain the lens shape and biomechanical properties. Changes in cell membrane shape during fiber cell differentiation and maturation have been extensively documented by electron microscopy (EM) studies2,3,4,5,6,7,8,9,10,20,26,27,28,29. Newly formed fiber cells have balls-and-sockets along their broad sides with very small protrusions along their short sides, while mature fibers have interlocking protrusions and paddles along their short sides. Nuclear fibers display tongue-and-groove interdigitations and globular membrane morphology. Little is known about the proteins that are required for these complex interlocking membranes. Previous studies on protein localization in fiber cells have relied on lens tissue sections, which do not allow clear visualization of the complex cell architecture.
This work has created and perfected a novel method to fix single and bundles of lens fiber cells to preserve the complex morphology and to allow immunostaining for proteins at the cell membrane and within the cytoplasm. This method faithfully preserves cell membrane architecture, comparable to data from EM studies, and allows staining with primary antibodies for specific proteins. We have previously immunostained cortical lens fibers undergoing differentiation and maturation22,23. In this protocol, there is also a new method to stain fiber cells from the lens nucleus. This protocol opens the door to understanding the mechanisms for formation and changes in membrane interdigitations during fiber cell maturation and lens nucleus compaction.
Protocol
Mice have been cared for based on an animal protocol approved by the Institutional Animal Care and Use Committee at Indiana University Bloomington. The mice used to generate representative data were control (wild-type) animals in the C57BL6/J background, female, and 8-12 weeks old. Both male and female mice can be used for this experiment, since the sex of the mice is very unlikely to affect the experiment's outcome.
1. Lens dissection and decapsulation
- Euthanize mice following the National Institutes of Health's "Guide for the Care and Use of Laboratory Animals" as well as animal use protocols approved by the institution.
NOTE: For the present study, mice were euthanized by CO2 overdose followed by cervical dislocation in accordance with an approved animal protocol (Indiana University). - Enucleate the eyes from the mice using curved forceps by depressing the tissue around the eyes with one side of the forceps to displace the eye out of the socket. Next, close the forceps underneath the eye and lift to remove the eye from the socket. Transfer the eyes to fresh 1x phosphate-buffered saline (PBS) in a dissection tray.
- Cut the optic nerve with ultra-fine scissors as close as possible to the eyeball. Carefully insert fine-tip, straight tweezers into the eyeball through the optic nerve's exit at the posterior of the eye.
- Carefully insert scissors at the same location as the tweezers in step 1.3 and begin cutting an incision from the posterior toward the corneal-scleral junction.
NOTE: Rodent lenses occupy ~30% of their eyes. Accidental damage will occur if the tweezers or scissors are inserted too deep into the eye. - Continue cutting along the corneal-scleral junction until at least half of the junction has been separated.
- Use tweezers to gently push on the cornea so that the lens can exit through the incision made with steps 1.4 and 1.5.
- Carefully remove any large pieces of tissue from the lens using fine-tip, straight tweezers. Inspect the lens to find the equatorial region.
- Shallowly pierce the lens using fine-tip, straight tweezers, and then remove the lens capsule. The mass of lens fiber cells will remain intact, and the lens epithelial monolayer will remain attached to the lens capsule. Discard the lens capsule.
2. Lens single fiber cell staining
- Transfer the lens fiber cell mass to a well plate with 500 µL of freshly made 1% paraformaldehyde (PFA; in 1x PBS) solution and incubate the samples at 4 °C overnight with gentle nutation (Figure 2A).
NOTE: The volumes for the solutions given are optimized for 48-well plates. If another size well-plate or tubes are used, adjust the volumes of the solutions accordingly. Fixation, blocking, and washing (1x PTX, 0.1% Triton X-100 in 1x PBS) solutions were made with 10x PBS and double distilled water (ddH2O) to a final concentration of 1x PBS. - Transfer the lens fiber cells to a 60 mm dish with 1% PFA. Use a sharp scalpel to split the ball of fiber cells in half along its anterior-posterior axis (Figure 2B). Cut the halves in half again along the same axis to produce quarters.
NOTE: The anterior-posterior axis is easily recognized by the direction of the fiber cells in the tissue mass. - Use straight tweezers to remove the nucleus region from the lens fiber cell quarters (Figure 2C).
NOTE: The lens nucleus region is rigid in rodent lenses, and the center of the lens will separate from the softer cortical fibers easily. - Post-fix the quarters of the lens cortex region in 200 µL of 1% PFA for 15 min at room temperature (RT) with gentle shaking (300 rpm on a plate shaker).
- Wash the tissue quarters twice in 750 µL of 1x PBS for 5 min each with gentle shaking at RT.
- Block the samples using 200 µL of blocking solution (5% serum, 0.3% Triton X-100, 1x PBS) for 1 h at RT with gentle shaking.
NOTE: For the representative data in this protocol, the samples were not stained with primary or secondary antibodies. After the blocking step, the samples were incubated with wheat germ agglutinin (WGA; 1:100) and phalloidin (1:100) (see Table of Materials) for 3 h at RT with gentle shaking and protection from light. Primary antibody staining has been demonstrated in previous publications22,23. - Incubate with 100 µL of primary antibody solution overnight at 4 °C with gentle shaking.
NOTE: The antibodies are diluted in blocking solution. Compared to slides with tissue sections, there are more cells in this type of preparation. Primary antibody concentrations should be increased to provide ample antibodies to stain more cells. Doubling the antibody concentration from what is used on tissue sections is recommended. The same applies to secondary antibodies. - Wash the tissue quarters three times with 1x PTX (0.1% Triton X-100, 1x PBS) for 5 min each at RT with gentle shaking.
- Incubate the fiber cells with 100 µL of secondary antibody/dye solution for 3 h at RT with gentle shaking. Protect the samples from light during this and subsequent steps.
NOTE: WGA, phalloidin, and other fluorescent dyes can be added to the secondary antibody solution for simultaneous labeling of the cell membrane, cytoskeleton, or other organelles while also labeling the primary antibody. - Wash the fiber cells four times with 1x PTX for 5 min each at RT with gentle shaking.
- Add one drop or 50 µL of mounting media onto a plus-charged microscope slide before transferring the tissue quarters to the slide.
- Use tweezers to gently separate the fiber cells from each other and try to limit overlapping of the cell bundles.
- Gently apply a #1.5 coverslip on top of the sample in mounting media. The mounting media should spread to the edge of the coverslip; if this does not occur, add some additional mounting media to the edge of the coverslip. Aspirate away any excess mounting media around the edge of the coverslip and use nail polish to seal the edges of the coverslip on the slide.
NOTE: Any type of mounting medium that is formulated for confocal microscopy can be used for these experiments.
3. Lens nucleus single fiber cell staining
- Complete the dissection outlined in section 1.
- Mechanically remove the cortical fibers from the ball of the lens fiber cells, leaving the lens nucleus, by gently transferring the fiber cell mass to wet, gloved fingertips and gently rolling the tissue mass.
NOTE: In mouse lenses, rolling the ball of lens fiber cells between gloved fingertips is an effective method for removal of the cortical fiber cells from the hard lens nucleus28,30. For lenses where this mechanical method is not effective, careful dissection or a vortexing method29 may be used to remove the cortical fiber cells. - Transfer the lens nucleus to freshly made 1% PFA solution in a well-plate and incubate overnight at 4 °C with gentle shaking.
- Transfer the sample to a 60 mm dish with 1% PFA and use a sharp scalpel to split the lens nucleus along the anterior-posterior axis. Halve the tissue samples again to produce nucleus quarters.
- Follow the process outlined in steps 2.4-2.13 for immunostaining and sample mounting.
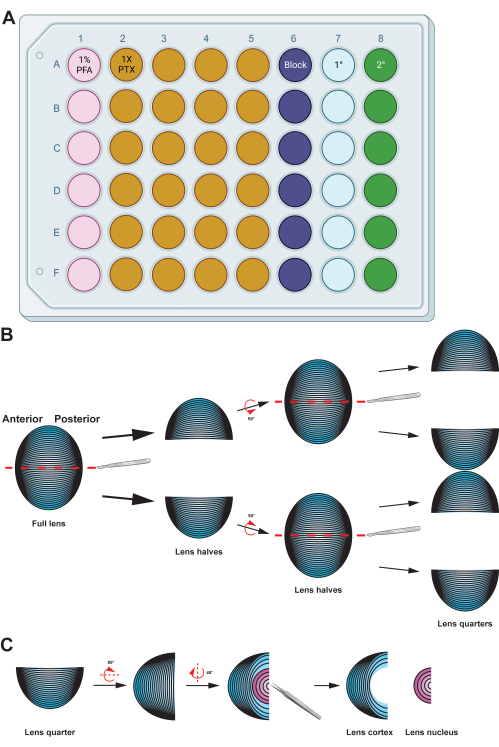
Figure 2: Graphical summary detailing the preparation and immunostaining of lens fiber cells. (A) This 48-well plate has been color-coded by column to demonstrate a sample plate setup for the described methods, allowing easy transfer of samples between the various immunostaining steps by gentle handling using forceps. While the representative data for this protocol is not incubated with a primary antibody, the diagram includes a column for primary antibody incubation, and the wells for washing can be reused after removing used wash buffers by aspiration. (B) After fixation of the lens fiber cell mass, the tissue is split along the anterior-posterior axis (red dashed lines) to preserve the original structure of the cells. Once the tissue mass has been halved, the samples are rotated and the halves split into quarters along the anterior-posterior axis (red dashed lines). (C) Removing the lens nucleus region (in pink) is easily done using tweezers to dig out the dense central tissue from the cortical fiber cells (in blue). Cartoon diagrams were partially created using BioRender.com and not drawn to scale. Please click here to view a larger version of this figure.
Results
Lens fiber cells are prepared from the lens cortex (differentiating fibers and mature fibers) and the nucleus, and the cells are stained with phalloidin for F-actin and WGA for the cell membrane. A mixture of bundles of cells or single lens fibers (Figure 3) are observed and imaged. From the lens cortex, two types of cells (Figure 3A) are found. Differentiating fiber cells in the lens periphery are straight, with very small protrusions along their short sides. A...
Discussion
This protocol has demonstrated the fixation, preservation, and immunostaining methods that faithfully preserve the 3D membrane morphology of bundles or singular lens fiber cells from various depths in the lens. The stained lens fibers are compared with SEM preparations that have long been used to study lens fiber cell morphology. The results show comparable membrane structures between both preparations. EM remains the gold standard for studying cell morphology, but immunolabeling is more challenging in SEM samples for lo...
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grant R01 EY032056 (to CC) from the National Eye Institute. The authors thank Dr. Theresa Fassel and Kimberly Vanderpool at the Scripps Research Core Microscopy Facility for their assistance with the electron microscope images.
Materials
| Name | Company | Catalog Number | Comments |
| 100% Triton X-100 | FisherScientific | BP151-500 | |
| 60mm plate | FisherScientific | FB0875713A | |
| 16% paraformaldehyde | Electron Microscopy Sciences | 15710 | |
| 10X phosphate buffered saline | ThermoFisher | 70011-044 | |
| 1X phosphate buffered saline | ThermoFisher | 14190136 | |
| 48-well plate | CytoOne | CC7672-7548 | |
| Cover slips (22 x 40 mm) | FisherScientific | 12-553-467 | |
| Curved tweezers | World Precision Instruments | 501981 | |
| Dissection microscope | Carl Zeiss | Stereo Discovery V8 | |
| Fine tip straight tweezers | Electron Microscopy Sciences | 72707-01 | |
| Fisherbrand Superfrost Plus Microscope Slides | FisherScientific | 12-550-15 | |
| LSM 800 confocal microscope with Airyscan (63X) and Zen 3.5 Software | Carl Zeiss | ||
| Nail polish | |||
| Normal donkey serum | Jackson ImmunoResearch | 017-000-121 | |
| Phalloidin (rhodamine) | ThermoFisher | R415 | |
| Primary antibody | |||
| Scalpel Feather Disposable, steril, No. 11 | VWR | 76241-186 | |
| Secondary antibody | |||
| Straight forceps | World Precision Instruments | 11252-40 | |
| Thermo Scientific Nunc MicroWell MiniTrays (dissection tray) | FisherScientific | 12-565-154 | |
| Ultra-fine scissors | World Precision Instruments | 501778 | |
| VECTASHIELD Antifade Mounting Medium with DAPI | Vector Laboratories | H-1200 | |
| Wheat germ agglutinin (fluorescein) | Vector Laboratories | FL-1021-5 |
References
- Lovicu, F. J., McAvoy, J. W. Growth factor regulation of lens development. Developmental Biology. 280 (1), 1-14 (2005).
- Kuszak, J., Alcala, J., Maisel, H. The surface morphology of embryonic and adult chick lens-fiber cells. The American Journal of Anatomy. 159 (4), 395-410 (1980).
- Kuszak, J. R. The ultrastructure of epithelial and fiber cells in the crystalline lens. International Review of Cytology. 163, 305-350 (1995).
- Kuszak, J. R., Macsai, M. S., Rae, J. L. Stereo scanning electron microscopy of the crystalline lens. Scanning Electron Microscopy. , 1415-1426 (1983).
- Lo, W. K., Harding, C. V. Square arrays and their role in ridge formation in human lens fibers. Journal of Ultrastructure Research. 86 (3), 228-245 (1984).
- Taylor, V. L., et al. Morphology of the normal human lens. Investigative Ophthalmology & Visual Science. 37 (7), 1396-1410 (1996).
- Vrensen, G. F. Aging of the human eye lens-a morphological point of view. Comparative Biochemistry and Physiology. Part A, Physiology. 111 (4), 519-532 (1995).
- Vrensen, G. F., Duindam, H. J. Maturation of fiber membranes in the human eye lens. Ultrastructural and Raman microspectroscopic observations. Ophthalmic Research. 27, 78-85 (1995).
- Willekens, B., Vrensen, G. The three-dimensional organization of lens fibers in the rabbit. A scanning electron microscopic reinvestigation. Albrecht von Graefe's Archive for Clinical and Experimental Opthalmology. 216 (4), 275-289 (1981).
- Willekens, B., Vrensen, G. The three-dimensional organization of lens fibers in the rhesus monkey. Graefe's Archive for Clinical and Experimental Ophthalmology. 219 (3), 112-120 (1982).
- Zhou, C. J., Lo, W. K. Association of clathrin, AP-2 adaptor and actin cytoskeleton with developing interlocking membrane domains of lens fibre cells. Experimental Eye Research. 77 (4), 423-432 (2003).
- Kuwabara, T. The maturation of the lens cell: a morphologic study. Experimental Eye Research. 20 (5), 427-443 (1975).
- Weeber, H. A., Eckert, G., Pechhold, W., vander Heijde, R. G. Stiffness gradient in the crystalline lens. Graefe's Archive for Clinical and Experimental Ophthalmology. 245 (9), 1357-1366 (2007).
- Weeber, H. A., et al. Dynamic mechanical properties of human lenses. Experimental Eye Research. 80 (3), 425-434 (2005).
- Weeber, H. A., vander Heijde, R. G. On the relationship between lens stiffness and accommodative amplitude. Experimental Eye Research. 85 (5), 602-607 (2007).
- Heys, K. R., Cram, S. L., Truscott, R. J. Massive increase in the stiffness of the human lens nucleus with age: the basis for presbyopia. Molecular Vision. 10, 956-963 (2004).
- Heys, K. R., Friedrich, M. G., Truscott, R. J. Presbyopia and heat: changes associated with aging of the human lens suggest a functional role for the small heat shock protein, alpha-crystallin, in maintaining lens flexibility. Aging Cell. 6 (6), 807-815 (2007).
- Glasser, A., Campbell, M. C. Biometric, optical and physical changes in the isolated human crystalline lens with age in relation to presbyopia. Vision Research. 39 (11), 1991-2015 (1999).
- Pierscionek, B. K. Age-related response of human lenses to stretching forces. Experimental Eye Research. 60 (3), 325-332 (1995).
- Biswas, S. K., Lee, J. E., Brako, L., Jiang, J. X., Lo, W. K. Gap junctions are selectively associated with interlocking ball-and-sockets but not protrusions in the lens. Molecular Vision. 16, 2328-2341 (2010).
- Lo, W. K., et al. Aquaporin-0 targets interlocking domains to control the integrity and transparency of the eye lens. Investigative Ophthalmology & Visual Science. 55 (3), 1202-1212 (2014).
- Cheng, C., et al. Tropomyosin 3.5 protects the F-actin networks required for tissue biomechanical properties. Journal of Cell Science. 131 (23), (2018).
- Cheng, C., et al. Tropomodulin 1 regulation of actin is required for the formation of large paddle protrusions between mature lens fiber cells. Investigative Ophthalmology & Visual Science. 57 (10), 4084-4099 (2016).
- Kuszak, J. R. The development of lens sutures. Progress in Retinal and Eye Research. 14 (2), 567-591 (1995).
- Bassnett, S., Costello, M. J. The cause and consequence of fiber cell compaction in the vertebrate lens. Experimental Eye Research. 156, 50-57 (2017).
- Biswas, S., Son, A., Yu, Q., Zhou, R., Lo, W. K. Breakdown of interlocking domains may contribute to formation of membranous globules and lens opacity in ephrin-A5(-/-) mice. Experimental Eye Research. 145, 130-139 (2016).
- Blankenship, T., Bradshaw, L., Shibata, B., Fitzgerald, P. Structural specializations emerging late in mouse lens fiber cell differentiation. Investigative Ophthalmology & Visual Science. 48 (7), 3269-3276 (2007).
- Cheng, C., et al. Age-related changes in eye lens biomechanics, morphology, refractive index and transparency. Aging. 11 (24), 12497-12531 (2019).
- Cheng, C., et al. EphA2 affects development of the eye lens nucleus and the gradient of refractive index. Investigative Ophthalmology & Visual Science. 63 (1), 2 (2022).
- Cheng, C., Gokhin, D. S., Nowak, R. B., Fowler, V. M. Sequential application of glass coverslips to assess the compressive stiffness of the mouse lens: strain and morphometric analyses. Journal of Visualized Experiments. (111), e53986 (2016).
- Forrester, J. V., Dick, A. D., McMenamin, P. G., Roberts, F., Pearlman, E., Saunders, W. B. Anatomy of the eye and orbit. The Eye (Fourth Edition). , 1 (2016).
- Cheng, C., Nowak, R. B., Fowler, V. M. The lens actin filament cytoskeleton: Diverse structures for complex functions. Experimental Eye Research. 156, 58-71 (2017).
- Goldberg, M. W., Fiserova, J. Immunogold labeling for scanning electron microscopy. Methods in Molecular Biology. 1474, 309-325 (2016).
- Goldberg, M. W. High-resolution scanning electron microscopy and immuno-gold labeling of the nuclear lamina and nuclear pore complex. Methods in Molecular Biology. 1411, 441-459 (2016).
- Hermann, R., Walther, P., Muller, M. Immunogold labeling in scanning electron microscopy. Histochemistry and Cell Biology. 106 (1), 31-39 (1996).
- Gokhin, D. S., et al. Tmod1 and CP49 synergize to control the fiber cell geometry, transparency, and mechanical stiffness of the mouse lens. PLoS One. 7 (11), e48734 (2012).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved