A subscription to JoVE is required to view this content. Sign in or start your free trial.
Laser Cell Ablation in Intact Drosophila Larvae Reveals Synaptic Competition
In This Article
Summary
This protocol demonstrates the laser cell ablation of individual neurons in intact Drosophila larvae. The method enables the study of the effect of reducing competition between neurons in the developing nervous system.
Abstract
The protocol describes single-neuron ablation with a 2-photon laser system in the central nervous system (CNS) of intact Drosophila melanogaster larvae. Using this non-invasive method, the developing nervous system can be manipulated in a cell-specific manner. Disrupting the development of individual neurons in a network can be used to study how the nervous system can compensate for the loss of synaptic input. Individual neurons were specifically ablated in the giant fiber system of Drosophila, with a focus on two neurons: the presynaptic giant fiber (GF) and the postsynaptic tergotrochanteral motor neuron (TTMn). The GF synapses with the ipsilateral TTMn, which is crucial to the escape response. Ablating one of the GFs in the 3rd instar brain, just after the GF starts axonal growth, permanently removes the cell during the development of the CNS. The remaining GF reacts to the absent neighbor and forms an ectopic synaptic terminal to the contralateral TTMn. This atypical, bilaterally symmetric terminal innervates both TTMns, as demonstrated by dye coupling, and drives both motor neurons, as demonstrated by electrophysiological assays. In summary, the ablation of a single interneuron demonstrates synaptic competition between a bilateral pair of neurons that can compensate for the loss of one neuron and restore normal responses to the escape circuit.
Introduction
Laser ablation is a preferred tool for dissecting neural circuits in a wide variety of organisms. Developed in model genetic systems like worms and flies, it has been applied across the animal kingdom to study the structure, function, and development of the nervous system1,2,3. Here, single-neuron ablation was employed to investigate how neurons interact during circuit assembly in Drosophila. The escape system of the fly is a favorite circuit for analysis because it contains the largest neurons and the largest synapses in the adult fly, and the circuit has been well-characterized in the past decades4. The role neuron-neuron interactions play in the assembly of the Giant Fiber circuit is a focal point of this research.
One type of interaction that has been a focal point in neuroscience since the work of Hubel and Wiesel in the 1960s is "synaptic competition"5,6. In this protocol, laser ablation was used to revisit the role of competition through single-cell ablation in the giant fiber system (GFS) of Drosophila, where the molecular underpinnings of the phenomena might be discovered.
Ablation of neurons in the developing fly has been difficult for a variety of reasons, including visualizing the target neurons, the precision of the ablation method, and the survival of the specimen. To overcome these problems in the GFS, the UAS/Gal4 system7 was used to label neurons of interest, and a two-photon microscope was used to remove the presynaptic giant fiber or the postsynaptic jump motor neuron (TTMn).
In this study, to determine the role that neighboring bilateral neurons play in adjusting synaptic connectivity and synaptic strength in the GFS, one of the bilateral pairs of neurons (either presynaptic GF or postsynaptic motor neuron) was deleted just before pupal development. At this developmental stage, GF axonogenesis has not been completed8. The GF structure and function of the synaptic circuit in the adult were then examined, with particular attention given to the output of the remaining GF.
Protocol
All animals used for the protocol were of the species Drosophila melanogaster. There are no ethical issues surrounding the use of this species. Ethical clearance was not necessary to carry out this work. The details of the Drosophila species, reagents, and equipment used in the study are listed in the Table of Materials.
1. Breeding Drosophila and selecting the correct larval stage
- Choose a Gal4 driver line that drives expression in the cells that are to be ablated and recombine it with or cross it to a UAS-GFP reporter line. Raise flies on standard fly food at 25 °C.
NOTE: For giant fiber (GF) ablation, R91H05-Gal4 or A307-Gal4 was recombined with UAS-GFP. ShakB(lethal)-GAL4 recombined with UAS-GFP was used for TTMn ablations (see Table of Materials). - Select larvae that have started to emerge from the food and crawl up the side of the food vial. Those larvae are in the wandering stage, which is the preferred stage for performing GF cell ablation.
NOTE: Ablation can be performed at any larval stage. The last larval stage was important for the manipulation of the development of the GFS because the GF cell bodies can be easily identified in the brain, but the GF axons have not made connections to their targets at this stage.
2. Preparing the 3rd instar larva
NOTE: Larvae were anesthetized using a method similar to Burra et al.9. To keep procedure times short, only prepare one larva at a time. Short exposure to anesthetic will enhance the survival of experimental animals10.
- Prepare a glass dish with a tightly fitting lid. Place a cotton ball in the dish, and under a fume hood, add about 3-5 ml of ethyl ether (dangerous, flammable liquid, lab coat, gloves, goggles). Cover the dish with the lid.
- Collect individual 3rd instar larvae from fly vials. At the 3rd larval stage, larvae leave the food medium and crawl up along the side of the food vial11.
- Ensure that larvae are still moving and haven't started puparium formation. Larvae that are stationary and show a shortening of the body are starting pupation and are unsuitable for ablation. Pick larvae up with a small paintbrush.
NOTE: The 3rd larval stage starts after the second molt at 72 h of development (after egg laying), and lasts until puparium formation at 120 h for flies raised at 25 °C.
- Ensure that larvae are still moving and haven't started puparium formation. Larvae that are stationary and show a shortening of the body are starting pupation and are unsuitable for ablation. Pick larvae up with a small paintbrush.
- Transfer one larva into a small, open container. Place the container in the glass dish with ethyl ether and close the dish tightly. When opening the glass dish, place it under a snorkel or in a fume hood to prevent fumes from escaping.
- In intervals of 30 s, remove the lid with the larva from the dish and check the larva for mobility under a dissection microscope. The larva will generally be immobilized within 1 min. Once the mouth hooks stop twitching, the larva is ready for mounting. Discard any larvae that are not fully immobilized after 3 min.
NOTE: The top of an upside-down lid of a microcentrifuge tube (lid removed from the tube) can be used as a container.
- In intervals of 30 s, remove the lid with the larva from the dish and check the larva for mobility under a dissection microscope. The larva will generally be immobilized within 1 min. Once the mouth hooks stop twitching, the larva is ready for mounting. Discard any larvae that are not fully immobilized after 3 min.
3. Mounting larvae on slides for ablation
- Place the anesthetized larva on a glass microscope slide. Submerge the larva in a drop of insect saline; this will wash away some of the food debris that might stick to the larva.
- Under the dissection microscope, remove most of the saline with a paper tissue. Position the larva dorsal side up for GF ablation or the ventral side up for TTMn ablation. Slowly lower a glass coverslip onto the larva. Add saline to the side of the coverslip to fill in the space between the glass slide and the cover slip.
- For GF ablation, check the positioning of the brain under high magnification on the dissection microscope. Ensure that the brain is lying level and is visible through the cuticle. Often, the brain will be covered with fat tissue, making visualization and cell ablation impossible.
- To displace any fat tissue covering the brain, apply slight pressure to the coverslip with forceps and move the coverslip from side to side. If the fat tissue can't be moved away from the brain this way, use a different larva instead.
4. Locating the target cells
NOTE: The multi-photon system used for this study was mounted on an upright microscope. System-specific software was used to control acquisition and laser stimulation settings. The system was equipped with an epifluorescence light source to locate the samples. The objective lens was a water immersion lens with 25x magnification, a long 2 mm working distance, and an NA of 1.10.
- Place the sample on the multi-photon microscope's stage and use the GFP filter to locate the sample in epifluorescence mode. For GF ablation, the cell bodies can be identified in the brain, as seen in Figure 1A,C,D. For TTMn ablation, a cluster of four cells can be identified from the ventral side in the ventral nerve cord (VNC), as shown in Figure 1A,B. When done focusing, center the cells in the field of view and switch to 2-photon mode.
5. Setting up the ablation parameters
- Adjust laser settings and detector gain to view the GFP-expressing cells with the Galvano scanner. A wavelength of 870 nm works best for visualizing the GFP signal. Center the cell in the field of view.
- Use a circular region of interest (ROI) to define the area for ablation. Ensure that the ROI covers most of the surface of the cell (Figure 1E,G).
- Set up the ablation protocol in the software. Set one frame of acquisition, then stimulation, followed by another acquisition frame.
- Start the stimulation laser power setting at a lower value (10%-20 %) and run the stimulation protocol. A successful ablation can be recognized by a clear circle in the center of the cell soma at the location of the ROI and an increase in fluorescence surrounding the ROI (Figure 1F).
- If the ablation was not successful and merely bleached the cell (Figure 1H), increase the laser power by increments of 5% or the number of loops one at a time and run the protocol again. The applied laser power was between 10% and 40% for the system used in the protocol.
NOTE: Laser power settings for ablation depend greatly on how deep the cells are located in the tissue and other factors such as cuticle folding. If cells are bright and clearly visible, laser power will be on the lower end of the range. Laser power will be different for each system depending on the laser model and the age of the laser.
- If the ablation was not successful and merely bleached the cell (Figure 1H), increase the laser power by increments of 5% or the number of loops one at a time and run the protocol again. The applied laser power was between 10% and 40% for the system used in the protocol.
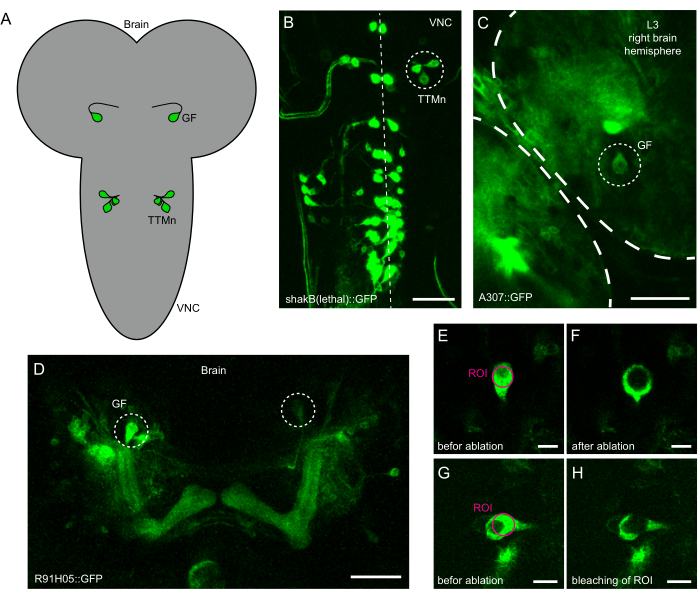
Figure 1: Identification of neurons for laser ablation in intact Drosophila L3 larvae. (A) Schematic of the location of giant fiber (GF) soma in the brain and cluster of motor neurons containing the tergotrochanteral motorneuron (TTMn) in the ventral nerve cord (VNC). (B) Maximum intensity projection of shakB(lethal)-Gal4 driving expression of GFP in the larval VNC. The midline is indicated by the dotted line. The circle indicates the cluster of neurons containing the TTMn that are targeted for laser ablation. Scale bar: 50 µm. (C) Partial projection view of the larval brain expressing GFP under the control of A307-Gal4. The circle indicates the GF soma targeted for laser ablation. Scale bar: 50 µm. (D) Maximum intensity projection of the brain with R91H05-Gal4 driving UAS-GFP. Both GFs (circles) are identifiable by location, shape, and size. Scale bar: 50 µm. (E) GF soma enlarged view before laser ablation. The magenta circle indicates the target region for laser ablation. Scale bar: 10 µm. (F) GF soma enlarged view after delivery of localized laser power and successful ablation. Scale bar: 10 µm. (G) GF soma enlarged view before laser ablation. The magenta circle indicates the target region for laser ablation. Scale bar: 10 µm. (H) GF soma enlarged view after delivery of localized laser power and unsuccessful ablation (bleaching). Scale bar: 10 µm. Please click here to view a larger version of this figure.
6. Recovering the larvae
- After successfully ablating the cell, remove the coverslip and gently pick up the larvae from the slide with a paintbrush. Place the larvae in a food vial. Larvae will start crawling around again within 30 min.
NOTE: Keeping procedure time short improves the survival rates of animals. With experience, the experiment can be performed in just under 10 min. - Continue raising larvae at standard conditions until they eclose from the pupal casing.
7. Testing the functionality of the GFS in adult flies
NOTE: The following steps are explained in detail in Allan and Godenschwege12, and Augustin et al.13.
- Test adult flies at age 2-5 days after eclosion. Anesthetize flies with CO2 and mount them on dental wax by pushing the legs into the wax. Spread the wings at a 90° angle and fix them in place with the wax. Secure the head by pushing the proboscis into the wax.
- Place stimulation wires into both eyes, a ground wire into the abdomen, and glass recording electrodes into the jump muscles (TTMs) on each side.
- Stimulate the GF circuit with single stimuli to measure response latency for both muscles. Stimulate the circuit with 100 Hz and 200 Hz trains of stimuli, respectively, to measure the following frequency for both muscles.
8. Dissection and labeling of the giant fiber system for confocal imaging
NOTE: Fly CNS dissection and dye injection are detailed in Boerner and Godenschwege14.
- Gently remove the fly from the dental wax with forceps and transfer it to a silicone elastomer-lined Petri dish under a dissection scope.
- Remove the legs, wings, and proboscis with scissors and use insect pins to secure the fly dorsal side up in the elastomer coating.
- Make a lateral incision along the midline from the middle abdomen, through the entire thorax, and up the neck, connecting the thorax to the head. Open the fly with insect pins. Remove organs and glands from the fly without damaging the nervous system.
NOTE: At this point, GF axons can be injected with dyes to label the GFs and electrically coupled neurons13. - Remove the head capsule from the head to expose the brain.
9. Immunohistochemistry of the nervous system
- After CNS dissection, fix the fly in 4% PFA in PBS for 45 min at room temperature (RT). Wash for 20 min in PBS at RT (3 times).
- Block the sample for 1 h at RT in 3% BSA in PBS with 0.5% detergent solution.
- Incubate for 2-3 nights at 4 °C in 3% BSA in PBS with 0.3% detergent solution and rabbit anti-GFP (1:500). Wash for 20 min in PBS at RT (6 times).
- Incubate overnight at 4 °C in PBS with anti-rabbit Alexa 488. Wash for 20 min in PBS at RT (6 times).
- Dehydrate samples with an ascending ethanol series (50%, 70%, 90%, 100% ethanol) for 10 min each step. Remove the pins and trim off the abdomen and the flight muscles. Transfer the rest of the thorax containing the nervous system onto a slide and cover it with a mounting medium like methyl salicylate. Place a coverslip on the sample and seal it with nail polish.
- Image the nervous system on a confocal microscope to analyze the anatomy of the GF terminal.
Results
This method can be used to manipulate the development of specific neuronal networks in the nervous system of Drosophila. The primary research question here was the formation of synaptic connections. Removing either the presynaptic GF or the postsynaptic TTMn enabled the investigation of reactive synaptogenesis at this central synapse and the molecular mechanisms crucial for synaptic function and development. As described in the protocol, laser cell ablation of one of the GFs or one of the TTMns was performed, an...
Discussion
Cell ablation with a 2-photon microscope proved to be a highly successful method to manipulate neuronal circuit development in Drosophila. Since this method is non-invasive, it causes minimal damage to the animal. The data support the usefulness of this cell-specific manipulation of known circuits.
Crucial for the success of the ablation was selecting the most appropriate Gal4 driver. Since the GFS is well studied, many specific Gal4 driver lines have been described7...
Disclosures
The authors have nothing to disclose.
Acknowledgements
Experiments on the 2-photon microscope were performed in the FAU Stiles-Nicholson Brain Institute Advanced Cell Imaging Core. We would like to thank the Jupiter Life Science Initiative for financial support.
Materials
| Name | Company | Catalog Number | Comments |
| Alexa Fluor 488 AffiniPure Goat Anti-Rabbit IgG (H+L) | Jaxkson ImmunoResearch | 111-545-003 | |
| Anti-green fluorescent protein, rabbit | Fisher Scientific | A11122 | 1:500 concentration |
| Apo LWD 25x/1.10W Objective | Nikon | MRD77220 | water immersion long working distance |
| Bovine Serum Albumin (BSA) | Sigma | B4287-25G | |
| Chameleon Ti:Sapphire Vision II Laser | Coherent | ||
| Cotton Ball | Genesee Scientific | 51-101 | |
| Dextra, Tetramethylrhodamine, 10,000 MW, Lysine Fixable (fluoro-Ruby) | Fisher Scientific | D1817 | |
| Drosophila saline | recipe from Gu and O'Dowd, 2006 | ||
| Ethyl Ether | Fisher Scientific | E134-1 | Danger, Flammable liquid |
| Fly food B (Bloomington recipe) | LabExpress | 7001-NV | |
| Methyl salicylate | Fisher Scientific | O3695-500 | |
| Microcentrifuge tube 1.5 mL | Eppendorf | 22363204 | |
| Microscope cover-slip 18x18 #1.5 | Fisher Scientific | 12-541A | |
| Neurobiotin Tracer | Vector Laboratories | SP-1120 | |
| Nikon A1R multi-photon microscope | Nikon | on an upright FN1 microsope stand | |
| NIS Elements Advanced Research | Nikon | Acquisition and data analysis software | |
| Paraformaldehyde (PFA) | Fisher Scientific | T353-500 | |
| PBS (Phosphate Buffered Salin) | Fisher BioReagents | BP2944-100 | Tablets |
| R91H05-Gal4 | Bloomington Drosophila Stock Center | 40594 | |
| shakB(lethal)-GAl4 | Bloomington Drosophila Stock Center | 51633 | |
| Superfrost microscope glass slide | Fisher Scientific | 12-550-143 | |
| Triton X-100 | Fisher Scientific | 422355000 | detergent solution |
| UAS-10xGFP | Bloomington Drosophila Stock Center | 32185 |
References
- Chung, S. H., Mazur, E. Femtosecond laser ablation of neurons in C. elegans for behavioral studies. Appl Phys A Mater Sci Process. 96 (2), 335-341 (2009).
- Bower, D. V., et al. Airway branching has conserved needs for local parasympathetic innervation but not neurotransmission. BMC Biol. 12, 92 (2014).
- Angelo, J. R., Tremblay, K. D. Laser-mediated cell ablation during post-implantation mouse development. Dev Dyn. 242 (10), 1202-1209 (2013).
- Allen, M. J., Godenschwege, T. A., Tanouye, M. A., Phelan, P. Making an escape: Development and function of the Drosophila giant fibre system. Semin Cell Dev Biol. 17 (1), 31-41 (2006).
- Hubel, D. H., Wiesel, T. N. Binocular interaction in striate cortex of kittens reared with artificial squint. J Neurophysiol. 28 (6), 1041-1059 (1965).
- Wiesel, T. N., Hubel, D. H. Comparison of the effects of unilateral and bilateral eye closure on cortical unit responses in kittens. J Neurophysiol. 28 (6), 1029-1040 (1965).
- Brand, A. H., Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 118 (2), 401-415 (1993).
- Allen, M. J., Drummond, J. A., Moffat, K. G. Development of the giant fiber neuron of Drosophila melanogaster. J Comp Neurol. 397 (4), 519-531 (1998).
- Burra, S., Wang, Y., Brock, A. R., Galko, M. J. Using Drosophila larvae to study epidermal wound closure and inflammation. Methods Mol Biol. 1037, 449-461 (2013).
- Kakanj, P., Eming, S. A., Partridge, L., Leptin, M. Long-term in vivo imaging of Drosophila larvae. Nat Protoc. 15 (3), 1158-1187 (2020).
- Bainbridge, S. P., Bownes, M. Staging the metamorphosis of Drosophila melanogaster. J Embryol Exp Morphol. 66, 57-80 (1981).
- Allen, M. J., Godenschwege, T. A. Electrophysiological recordings from the Drosophila giant fiber system (GFs). Cold Spring Harb Protoc. 2010 (7), 5453 (2010).
- Augustin, H., Allen, M. J., Partridge, L. Electrophysiological recordings from the giant fiber pathway of d. Melanogaster. J Vis Exp. (47), e2412 (2011).
- Boerner, J., Godenschwege, T. A. Whole mount preparation of the adult Drosophila ventral nerve cord for giant fiber dye injection. J Vis Exp. (52), e3080 (2011).
- Blagburn, J. M., Alexopoulos, H., Davies, J. A., Bacon, J. P. Null mutation in shaking-b eliminates electrical, but not chemical, synapses in the Drosophila giant fiber system: A structural study. J Comp Neurol. 404 (4), 449-458 (1999).
- Kennedy, T., Broadie, K. Newly identified electrically coupled neurons support development of the Drosophila giant fiber model circuit. eNeuro. 5 (6), 0346 (2018).
- Mcfarland, B. W., et al. Axon arrival times and physical occupancy establish visual projection neuron integration on developing dendrites in the Drosophila optic glomeruli. bioRxiv. , (2024).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved