1. Setup of the Schlenk Line
For a more detailed procedure, please review the "Schlenk Lines Transfer of Solvent" video in the Essentials of Organic Chemistry series. Schlenk line safety should be reviewed prior to conducting this experiment. Glassware should be inspected for star cracks before use. Care should be taken to ensure that O2 is not condensed in the Schlenk line trap if using liquid N2. At liquid N2 temperature, O2 condenses and is explosive in the presence of organic solvents. If it is suspected that O2 has been condensed or a blue liquid is observed in the cold trap, leave the trap cold under dynamic vacuum. Do NOT remove the liquid N2 trap or turn off the vacuum pump. Over time the liquid O2 will sublime into the pump - it is only safe to remove the liquid N2 trap once all of the O2 has sublimed.
- Close the pressure release valve.
- Turn on the N2 gas and the vacuum pump.
- As the Schlenk line vacuum reaches its minimum pressure, prepare the cold trap with either liquid N2 or dry ice/acetone.
- Assemble the cold trap.
2. Preparation of the Solid Reactants
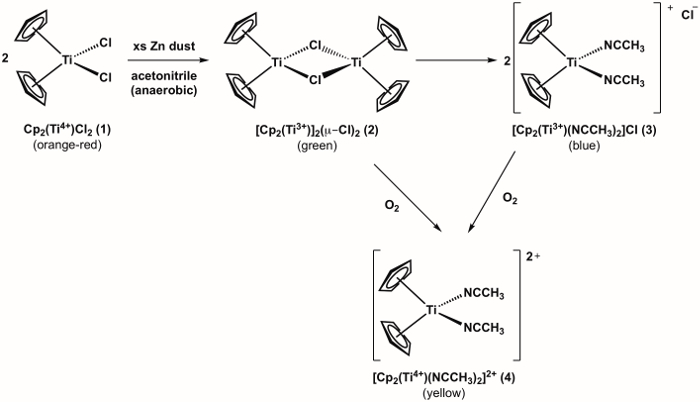
- Weigh 100 mg (0.40 mmol) of solid dicyclopentadienyltitanium(IV) dichloride (compound 1, Figure 1) and 78 mg (1.2 mmol) zinc dust into a Schlenk flask (Schlenk flask A).
- Fit Schlenk flask A with a greased glass stopper and attach the Schlenk flask side arm to the Schlenk line with Tygon tubing.
- Open the stopcock of the Schlenk line tube attached to Schlenk flask A to vacuum. Slowly open the stopcock on Schlenk flask A. Evacuate Schlenk flask A for 5 min.
- Repressurize Schlenk flask A with N2 by first closing the stopcock on the Schlenk flask. Slowly repressurize the Schlenk line tubing with N2 by turning the Schlenk line stopcock to N2. Make several (at least 5) quick 180 ° turns on the Schlenk flask stopcock, making sure the stopcock is closed after each turn. Slowly open the stopcock to finish filling Schlenk flask A with N2.
- Close the Schlenk flask A stopcock.
- Repeat steps 2.3-2.5 two more times. On the last cycle, leave the stopcock to the Schlenk flask A open.
3. Preparation of the Solvent
NOTE: Since the reaction is not water sensitive, glassware and solvents do not need to be dried. However, if the preparation is for use in the glovebox, all glassware and solvents should be appropriately dried.
- Measure 15 mL of acetonitrile and transfer the solvent to a new Schlenk flask (Schlenk flask B). Fit Schlenk flask B with a septum.
- Connect Schlenk flask B to the Schlenk line using Tygon tubing. Evacuate the tubing for 5 min and refill the tubing with N2 (the stopcock to the Schlenk flask should remain closed). Repeat the evacuation/refill cycles two more times. Leave the tubing under N2.
- Purge one of the unused Tygon tubes on the Schlenk line with N2, fitted with a long needle.
- Insert the needle into the septum of Schlenk flask B and lower the needle into the acetonitrile.
- Insert a second needle (not attached to the Schlenk line) into the septum of Schlenk flask B. This is the vent needle. Upon insertion of the vent needle, N2 should start bubbling through the acetonitrile.
- Allow the acetonitrile to degas for 15 min.
- Open the stopcock to Schlenk flask B.
- Remove the vent needle, followed by the needle connected to the Schlenk line. Close the stopcock on the Schlenk line that is connected to the long needle.
4. Addition of Solvent via Cannula (Figure 3)
- Make sure that the stopcocks to both of the Schlenk flasks (A & B) are open to N2.
- Replace the glass stopper on Schlenk flask A with a rubber septum.
- Insert one end of the cannula through the septum on Schlenk flask B (the donor flask). Do NOT put the needle into the acetonitrile.
- Ensure N2 is flowing through the cannula by putting the opposite end of the cannula close to the skin of the arm.
- Insert the other end of the cannula into Schlenk flask A (the receiving flask).
- Close the stopcock to Schlenk flask A.
- Lower the cannula in Schlenk flask B so that the tip reaches the bottom of the acetonitrile.
- Insert a vent needle in the septum of Schlenk flask A. Solvent should begin to flow. If no solvent is flowing, try increasing the N2 flow or raising the solvent flask above the height of the receiving flask.
- Transfer all 15 mL of the acetonitrile from Schlenk flask B to A. If only a portion of the solvent is desired, simply remove the cannula tip from the solvent in Schlenk flask B to stop the flow of liquid.
- Remove the vent needle from the septum and open the stopcock to Schlenk flask A.
- Remove the cannula from Schlenk flask A.
- Remove the cannula from Schlenk flask B.
5. Synthesis of Ti(III) Metallocene (Compound 3)
- Vigorously stir the solution for 15 min (or until the reaction mixture turns blue).
- If a green color persists, add more zinc dust (1-2 additional equivalents). To add more zinc dust to the system without introducing O2, make sure that the Schlenk flask stopcock is open to positive N2 pressure. Remove the rubber septum and add the solid to the flask. Re-attach the rubber septum. If the addition of excess zinc dust does not effect the desired color change to blue, O2 was likely introduced into the system.
6. Addition of Solvent via Syringe
- Degas 10 mL of acetonitrile as described in step 3 in Schlenk flask B.
- Make sure that both Schlenk flask A & B stopcocks are open to N2 and are fitted with rubber septa.
- Insert the syringe needle into either flask and pull N2 gas into the syringe.
- Remove the needle and eject the N2 into the hood.
- Repeat steps 6.3-6.4 two more times.
- Insert the syringe needle fitted to a 10 mL syringe into Schlenk flask B and pull up the desired volume of solvent (5 mL).
- Remove the needle from the solvent but leave the needle in the Schlenk flask. Bend the needle so that the syringe is pointing up (the needle should form an arch) and pull ~1 mL of N2 gas into the needle. There should be a gas "bubble" at the top of the syringe.
- While keeping the needle arched, remove the needle from Schlenk flask B. The syringe should still be pointed up with the bubble of N2 at the tip of the syringe where the needle is attached. The N2 bubble will prevent acetonitrile from leaking out of the syringe.
- With the needle still arched and the syringe pointing up, insert the needle into the septum of Schlenk flask A.
- Slowly add acetonitrile to Schlenk flask A. At this point, the position of the syringe is irrelevant.
- When solvent addition is complete, remove the syringe needle from Schlenk flask A.