Werner Heisenberg considered the limits of how accurately one can measure properties of an electron or other microscopic particles. He determined that there is a fundamental limit to how accurately one can measure both a particle’s position and its momentum simultaneously. The more accurate the measurement of the momentum of a particle is known, the less accurate the position at that time is known and vice versa. This is what is now called the Heisenberg uncertainty principle. He mathematically related the uncertainty in the position and the uncertainty in momentum to the quantity involving Planck’s constant.
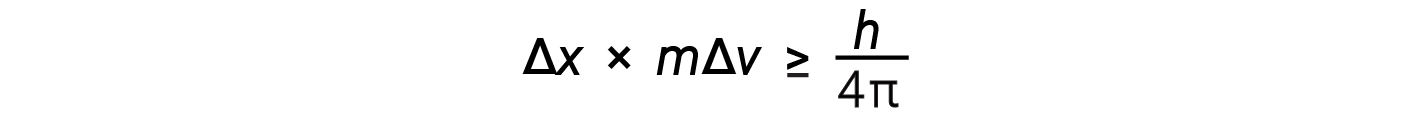
This equation calculates the limit to how precisely one can know both the simultaneous position of an object and its momentum.
Thus, the more accurate is the position of the electron, the less accurate is its velocity and vice versa. For example, one can predict where a baseball would land in the outfield by noting its initial position and velocity and by considering the effect of gravity and wind, etc. The trajectory of the baseball can be estimated.
For an electron, however, the position and velocity cannot be determined simultaneously. Therefore, a trajectory for the electron of an atom cannot be determined. This behavior is indeterminate. Instead of the precise location of an electron, one can talk in terms of the probability of finding an electron in a certain region of the atom, which is a probability density. It can be indicated as psi square (ψ2). The higher the probability of finding an electron in a particular region, the larger the value of psi square. Based on this, atoms are described as consisting of a nucleus surrounded by an electron cloud.
Heisenberg’s principle imposes ultimate limits on what is knowable in science. The uncertainty principle can be shown to be a consequence of wave–particle duality, which lies at the heart of what distinguishes modern quantum theory from classical mechanics.
This text is adapted from Openstax, Chemistry 2e, Section 6.3: Development of Quantum Theory.
From Chapter 7:

Now Playing
7.8 : The Uncertainty Principle
Electronic Structure of Atoms
22.0K Views

7.1 : The Wave Nature of Light
Electronic Structure of Atoms
46.9K Views

7.2 : The Electromagnetic Spectrum
Electronic Structure of Atoms
51.4K Views

7.3 : Interference and Diffraction
Electronic Structure of Atoms
27.4K Views

7.4 : Photoelectric Effect
Electronic Structure of Atoms
28.5K Views

7.5 : The Bohr Model
Electronic Structure of Atoms
46.6K Views

7.6 : Emission Spectra
Electronic Structure of Atoms
46.2K Views

7.7 : The de Broglie Wavelength
Electronic Structure of Atoms
24.8K Views

7.9 : The Quantum-Mechanical Model of an Atom
Electronic Structure of Atoms
40.4K Views

7.10 : Quantum Numbers
Electronic Structure of Atoms
33.0K Views

7.11 : Atomic Orbitals
Electronic Structure of Atoms
31.6K Views

7.12 : The Pauli Exclusion Principle
Electronic Structure of Atoms
30.3K Views

7.13 : The Energies of Atomic Orbitals
Electronic Structure of Atoms
23.0K Views

7.14 : The Aufbau Principle and Hund's Rule
Electronic Structure of Atoms
37.0K Views

7.15 : Electron Configuration of Multielectron Atoms
Electronic Structure of Atoms
34.4K Views
Copyright © 2025 MyJoVE Corporation. All rights reserved