A Low-Cost, Odor-Reward Association Task for Tests of Learning and Memory
In This Article
Summary
An odor-reward, associative learning task was used to investigate the differential effects of physiological manipulation on long-term and short-term memory.
Abstract
Robust and simple behavioral paradigms of appetitive, associative memory are crucial for researchers interested in cellular and molecular mechanisms of memory. In this paper, an effective and low-cost mouse behavioral protocol is described for examining the effects of physiological manipulation (such as the infusion of pharmacological agents) on the learning rate and duration of odor-reward memory. Representative results are provided from a study examining the differential role of tyrosine kinase receptor activity in short-term (STM) and long-term memory (LTM). Male mice were conditioned to associate a reward (sugar pellet) with one of the two odors, and their memory for the association was tested 2 or 48 h later. Immediately prior to the training, a tyrosine kinase (Trk) receptor inhibitor or vehicle infusions were delivered into the olfactory bulb (OB). Although there was no effect of the infusion on the learning rate, blockade of the Trk receptors in the OB selectively impaired LTM (48 h), and not short-term memory (STM; 2 h). The LTM impairment was attributed to the diminished odor selectivity as measured by the length of the digging time. The culmination of the results of this experiment showed that Trk receptor activation in the OB is the key in olfactory memory consolidation.
Introduction
The mechanisms of associative memory formation have previously been investigated predominantly based on one-trial fear conditioning studies. However, many mundane tasks typically have more complex acquisition patterns and rely on repeated encounters. The goal of this protocol is to provide a cost-effective rodent behavioral paradigm that is used to understand the cellular and molecular mechanisms of multi-trial appetitive learning and memory.
Odor learning that is dependent on the main olfactory bulb (OB) provides several advantages for the study of multi-trial appetitive memory. First, OB-dependent memories have varying durations (STM, LTM, and intermediate-term memory1) and rely on the same molecular2,3 and structural mechanism as elsewhere in the brain, including neuromodulation4, long-term potentiation5, and adult neurogenesis6,7,8. Second, in contrast to the higher-order regions, like the hippocampus, OB-dependent memories allow for observations of a more direct correspondence between experimenter manipulations of the perceptual environment and changes to the neural circuitry responsible for learning8,9,10,11. In this paper, an OB-dependent associative learning and memory paradigm, that can be used to study general molecular and structural mechanisms, is detailed. It was developed to allow researchers to access the advantages of olfactory learning for the study of cellular and molecular mechanisms of memory.
In our recent publication3, the protocol described here was used to demonstrate that the consolidation of appetitive odor learning is dependent upon Trk receptor activation within the OB. In the protocol below, areas where the behavioral paradigm can be adjusted for different experimental needs, are also discussed.
A total of 27 adult male CD-1 mice, 8 weeks old at the time of cannulation, were used in this study. For the precise group distributions and odor set use, see the methods section of our previous publication3. Male mice were used to avoid large fluctuations in estrogen levels because previous research12 showed that olfactory memory is enhanced by increased estrogen levels. These mice were always kept on a 12:12 h reverse light/dark cycle and had access to water. During the behavioral experiments, the diets of the mice were restricted to maintain them at ~90% of their free feeding weight. Diet restriction began 3 days before the commencement of the behavioral experiment. As will be described below, the same set of mice are presented with different odor sets in order to reach the appropriate levels of statistical power while minimizing the animal use. The statistical analysis section shows how to account for the random variance that may be introduced by this.
Protocol
The protocol below adheres to the animal care guidelines of the IACUC at Earlham College.
1. Olfactory bulb cannulation
NOTE: These surgeries do not require sterile technique since they do not require large incisions to be made. However, each institution may differ in their requirements. If experimenters are performing this surgery on immunocompromised mouse strains, additional considerations may be necessary. In all, experimenters are encouraged to discuss this protocol with their veterinarian and animal care team prior to use and to clean and disinfect all tools between each surgery.
- During the setup, soak cannulae and screws in a small beaker or Petri dish containing 32% chlorhexidine to keep them disinfected.
- Anesthetize mice with gaseous 4% isoflurane in pure oxygen and secure into a stereotaxic apparatus. During the surgery, ensure that the mice are maintained under 1.5-2% isoflurane anesthesia supplied through a nose cone. Monitor breathing throughout the surgery. Use eye ointment to prevent the dryness of eyes while under anesthesia.
- After the mouse is secured and no longer responds to a firm pinch of the hind foot, use 32% chlorhexidine to rub the top of the head in order to clean incision surface.
- Next, rub the top of the head in the rostral to caudal direction with lidocaine (topical analgesia).
- Press down firmly to make a single incision down the midline with a clean scalpel blade.
- Using a drill attached to the stereotaxic, drill two holes over the olfactory bulbs for the guide cannula (26 G) using coordinates AP +5.0 mm, ML +/-0.75 mm with respect to bregma.
- Drill two holes over the cerebellar formation.
NOTE: The location of these screws does not need to be precise, ensure that they are symmetrical along the midline. - Place screws into the two holes over the cerebellar formation and use a tissue adhesive to secure these screws to the skull.
- Use the stereotaxic to insert the guide cannula (26 G) into the holes drilled over the olfactory bulbs (step 1.6). Lower the cannula DV 1.0 mm.
- Mix the dental cement in a Petri dish. Use a small metal scoop to slowly pile the dental cement around the cannula. Let it dry for 5 s. Then, remove the arms of the stereotaxic device, being careful not to pull the guide cannula out in the process. Continue piling the dental cement over the entire incision until a small cap is formed (See Figure 1A).
- Place dummy plugs into the guide cannula at this point to prevent the blockage.
- Immediately after the surgery, inject ketoprofen (0.2 mg/kg) and saline (200 µL) to reduce pain and rehydrate. Provide softened food or hydrogel to the mice after surgery. Do not leave mice unattended until they have regained consciousness to maintain sternal recumbency.
NOTE: Mice are also singly housed from this point forward. - Inject saline (200 µL) and ketoprofen (0.2 mg/kg) once a day for two days after the surgery.
- For 2 days and up to 5 days, (as needed) after the surgery, weigh the mice and monitor their weights. If the weight does not return to pre-surgery levels within two or three days, consult with the veterinarian about the appropriate method of feeding.
- Allow mice to recover for at least 7 days before beginning the behavioral training.
2. Associative discrimination task
- Infusions
- Administer OB-specific infusions of the tyrosine kinase receptor inhibitor, K252a (50 μM; 5% DMSO in saline), or vehicle (5% DMSO in saline) to the mice as follows.
- Deliver the infusion into the OB of the mice bilaterally. Inject 2.0 µL final volume per bulb with 0.2 µL/min infusion rate and 10 min of total infusion time using a dual injector pump.
NOTE: Timing of the manipulation can be adjusted depending on the type of behavioral study being performed. Exact steps of infusion are specific to each injector pump and provided in the manufacturer’s manual. - Make sure to leave the injectors inside the cannulae for about 5 min after delivery to inhibit the backflow and promote diffusion. (i.e., plan for a total of 15 min for each infusion).
- Deliver the infusion into the OB of the mice bilaterally. Inject 2.0 µL final volume per bulb with 0.2 µL/min infusion rate and 10 min of total infusion time using a dual injector pump.
- Administer OB-specific infusions of the tyrosine kinase receptor inhibitor, K252a (50 μM; 5% DMSO in saline), or vehicle (5% DMSO in saline) to the mice as follows.
- Odor sets.
- Dilute all odorants in light mineral oil to a partial pressure of 1.0 Pa using a pre-calculated ratio based on vapor pressure (Table 1).
- Use the 5 separate odor pairs from Table 1 (numbers in the table indicate volume in µL to mix in 50 mL mineral oil for 1.0 Pa).
- To prepare the scented sand to be used during the behavioral steps (Section 2.3 and 3), mix 400 µL of the 1.0 Pa odorant from Step 2.2.2 for every 100 g of play sand.
- Shaping
NOTE: Mice should undergo shaping for a period of 10 days as described below.- Bring mice into the procedure room and handle them for 10 min per day for the first two days following recovery from surgery.
- On Day 3, place a Petri dish filled with (+)-limonene scented sand into the home cages of the mice and filled with about 10 sucrose pellets, each 5-mg in mass.
NOTE: Use 1.0 Pa (+)-limonene (mix 102 µL in 50 mL mineral oil) as the rewarded odor and plain mineral oil (the diluting agent for the testing odors) as the unrewarded odor. Choosing monomolecular odorants is also recommended since these are most likely to be novel to mice. - Replenish both the sand and the pellets on Day 4.
- On Days 5 and 6, acclimate the mice to the custom-made behavioral apparatus, by placing them into the apparatus and letting them explore the space (Figure 1B,C). Make the apparatus using a standard home cage, and poly (methyl methacrylate) to construct two lids and a black center divider. Ensure that both the lids and the center divider are 1-2 cm larger than the home cage.
- Prepare one Petri dish of limonene scented sand and another with sand containing mineral oil. After acclimation on both Day 5 and 6, place both the dishes of sand into the test chamber and mix 10 sucrose pellets into the limonene-scented dish to serve as the reward. Place each mouse into the test chamber for 10 min and allow to freely explore and consume the reward pellets.
- On Day 7, introduce mice to an abridged version of the final testing procedure, by placing the dishes containing limonene scented and mineral oil scented sand into the behavioral apparatus. This time include the center divider.
- Place a single reward on top of the limonene-scented sand and place the mouse into the resting chamber.
- Once the mouse has been placed in the resting chamber, lift the center divider to allow the mouse to enter the test chamber to investigate and dig in the sand-filled dishes. Return the mouse to the resting chamber either after it has retrieved the reward pellet or after 5 min has elapsed.
- Repeat this process for a total of 10 trials for each individual mouse. Counterbalance the placement of the rewarded dish of either on the left side or right side by employing a random number generator. There are no rest periods between trials.
- On Day 8, repeat the trials of Day 7, but also progressively bury the pellet deeper and deeper in the sand.
NOTE: Most mice should be digging for the unseen reward pellet by the 10th trial on Day 8. - On Day 9, increase the number of trials to 20 trials for each mouse with the full deep burial of sucrose pellet and introducing the mice to the testing chamber for 1 min per trial. Let mice dig in both dishes for the reward.
- On Day 10, repeat the 20 trials for each mouse, but if they dig in the unrewarded dish before they dig in the rewarded dish, and then begin the next trial. Allow the mice that first dug in the rewarded (limonene-scented) dish to retrieve the reward pellet before sending them back to the resting chamber.
3. Training and testing
NOTE: Once the mice have started digging reliably for the unseen, odor-cued reward pellets, the experiment can commence.
- Training
NOTE: The training phase begins two days after shaping is completed and consists of 20 trials for each mouse. Prior to the training, deliver the intrabulbar drug/vehicle infusions immediately (see Section 2.1 for infusion details) and begin the training immediately following the infusions.- Place a mouse into the resting chamber.
- Place two dishes of sand scented with a novel odor-pair in the test chamber, where a reward pellet is buried in one of the dishes.
- Once the testing chamber is ready, lift the opaque barrier and introduce the mouse into the test chamber. Immediately return the mouse to the resting chamber, if the mouse digs in the unrewarded dish first (record these trials as a "0"). If the mouse digs in the rewarding odor first, allow it to retrieve the pellet and return it to the resting chamber. Record these trials as a "1." If the trial lasts 1 min without the mouse retrieving the reward, send the mouse back to the resting chamber.
- Clean and refill dishes and begin the next trial. Repeat the same for 20 trials.
- Testing
NOTE: Memory testing can be performed at any duration of interest to the researcher. In this experiment, two separate groups of mice were tested 2 h (STM) or 48 h (LTM) following the training given the interest in the differential influence of K252a on STM and LTM.- Carry out testing using the same odors and procedure as described for the training (Section 3.1).
NOTE: Depending on the research question, the researcher may need to include control groups. For example, in the previously published experiment, the effects of Trk receptor blockade on memory consolidation was studied3. Therefore, a control group was infused with K252a prior to 48-h testing to show that the effects were not due to interference with retrieval.
- Carry out testing using the same odors and procedure as described for the training (Section 3.1).
- Statistical analysis
NOTE: SPSS 22.0 syntax for each step provided as a Supplemental File as an example.- Perform statistical analysis using linear mixed effects analysis. Unlike ANOVAs, linear mixed effects models can better account for random effects and repeated measures.
- Calculate the dependent measure: "proportion correct." Recall from 3.1.3 that a "1" was assigned to trials in which the mouse dug in the rewarding odor first, and a "0" if the mouse dug first in the unrewarding odor. Average every five trials to create four trial blocks (TB; e.g., trial block 1 or TB1 was the average of trials 1-5, trial block 2 or TB 2 was the average of trials 6-10 and so forth).
- Set the independent variables or fixed effects as drug groups (K252a or Vehicle; Section 2.1.1) and trial blocks (from 3.3.1). In the representative results below, the variables used for each analysis are specified.
- Include individual mouse and odor set nested within the mouse as "random effects" in order to compensate for intrinsic behavioral differences in the mice and any effects of using multiple odor sets.
- Perform a logit transformation on the proportion correct.
NOTE: The proportion correct is not a continuous, unbound dependent variable. Thus, it violates two assumptions for linear models. Therefore, the logit transformation is performed. - Use estimated marginal means to perform post hoc tests on significant interactions identified by the full model; multiple pairwise comparisons must be corrected for in post hoc testing. Bonferroni or Šidák are typically used.
Results
As described, this protocol allows researchers to assess the influence of some manipulation on learning, STM, and LTM. Sample results from Tong et al, 20183 are presented here. The results support the hypothesis that Trk receptor blockade selectively inhibits LTM, but not learning or STM.
Figure 2A shows the schematics of training, STM test, and LTM test. First, it was shown that K252a infusions did not affect the learning rate of an odor-reward association. Figure 2B (Section 3.1) shows the learning rate of both K252a and vehicle groups from Training. Statistical analysis using a linear mixed model was run with two fixed effects, drug group and trial block (TB); mouse and odor set nested within the mouse were random effects. Data from Testing (Section 3.2) were not included in the analysis. A significant main effect was seen from trial block (F(3, 183.692) = 43.735, p < 0.001), but no effect from drug group (F(1, 85.685) = 0.132, p = 0.717) and no significant interaction (F(3, 183.692) = 0.111, p = 0.954). Post hoc tests, using the Šidák adjustment, confirmed that the K252a and vehicle groups did not differ on any of the trial blocks during Training (p > 0.05 for all comparisons). TB2, TB3, and TB4 were significantly higher than TB1 for all comparisons (p ≤ 0.001 in all cases), demonstrating that both groups successfully learned the odor-reward association by the end of 20 Training trials.
Next, to examine the effects of the infusion on STM and LTM, the same analysis was run and included data from Testing (Section 3.2). It showed a significant interaction between drug group and trial block (F(2, 77.558) = 4.043, p = 0.021), with no significant main effects of drug group (F(1, 55.629) = 1.438, p = 0.236) or trial block (F(2, 69.979) = 1.360, p = 0.263). In order to specifically examine memory, post hoc pairwise comparisons with Šidák correction, compared differences between the last trial block of Training (Section 3.1) and the first trial block of Testing (Section 3.2) either 2 (STM) or 48 hours (LTM) later. For vehicle-infused mice, the comparisons showed retention of the associative memory at both 2- and 48 h post-training (p > 0.05 for all comparisons with training performance). For K252a-infused mice, the first trial block of the 2-hour test (STM) did not differ from the last trial block of Training (p > 0.05); however, their memory performance was significantly lower after 48 h (p = 0.018). In addition, memory at the 48-hour test was significantly reduced compared to memory at the 2 hours test (p = 0.009), and to the performance of the vehicle group at the 48-hour test (p = 0.006). There was no difference in STM between vehicle- and K252a-infused mice (p = 0.356). Together, the results show that K252a inhibition of Trk receptors in the olfactory bulb selectively disrupts long-term, but not short-term, odor memory (Figure 3).
| Odor set | Odor 1 | Odor 2 |
| 1 | pentanoic acid | butanoic acid |
| 225.1 | 63.6 | |
| 2 | hexanal | heptanal |
| 11.1 | 35.3 | |
| 3 | propyl acetate | butyl acetate |
| 3.1 | 10.9 | |
| 4 | 2-octanone | 2-heptanone |
| 87.4 | 28.7 | |
| 5 | pentanol | hexanol |
| 37.2 | 127.3 |
Table 1: Mixing volumes for odor sets. Each row shows two odors that can be used as a pair for the behavioral steps. For example, to use the first "odor set," make the pentanoic and butanoic acid mixtures. The numbers in the table indicate volume in µL to mix in 50 mL mineral oil for 1.0 Pa concentration of each odor. During training and testing, one Petri dish would be scented with pentanoic acid, the other with butanoic acid.

Figure 1: Cannulation placement and behavioral apparatus. (A) Shows the relative position of the cannula, dental cement cap, and screws to the head of the mouse. Note that the needles of the cannula reach into the two olfactory bulbs, the pedestal itself is embedded into the dental cement cap. The screws are placed into two holes drilled into the skull over the cerebellar formation. The screws do not touch the brain itself, but they act as a caudal anchor for the dental cement cap. The figure shows the relative size to make the cement cap. (B) Shows the assembled behavioral apparatus. The body is a typical mouse home cage. Lids with holes for air were made from plexiglass. The center divider is also made from black plexiglass. The lids should be made from heavy enough plexiglass that they can act as a tract for the center divider to be lifted and placed down during the trials. (C) Shows one side of the behavioral apparatus. Note that the center divider is slightly taller than the cage for ease of lifting. Petri dishes of sand can be placed along the edge. Please click here to view a larger version of this figure.
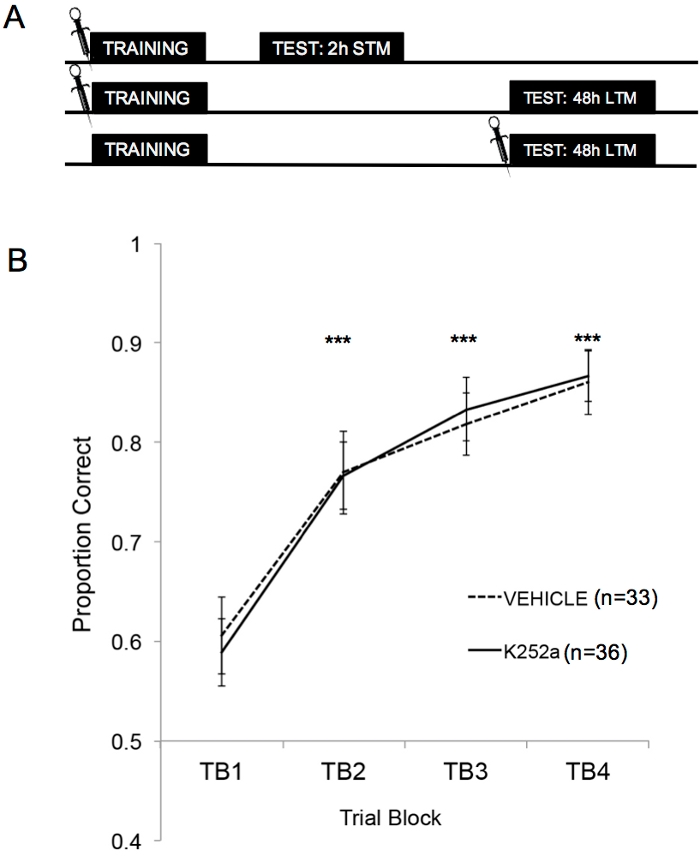
Figure 2: Study design and learning results. (A) Shows the schematic of the study design. Note that the STM and LTM groups were independent (i.e. different groups of mice). The symbols at the beginning of the blocks indicate the time when infusions were given. (B) Shows the proportion correct for Trials 1-20 during Training. The results indicate that K252a and vehicle groups did not differ in their learning rate (slope of the lines). Error bars represented the standard error of the mean (SEM). Asterisks show significant increases in proportion correct compared to TB1 for both vehicle and K252a cohorts (p ≤ 0.001 for all comparisons). This figure is adapted from Tong et al. 2018 with permission3. Please click here to view a larger version of this figure.
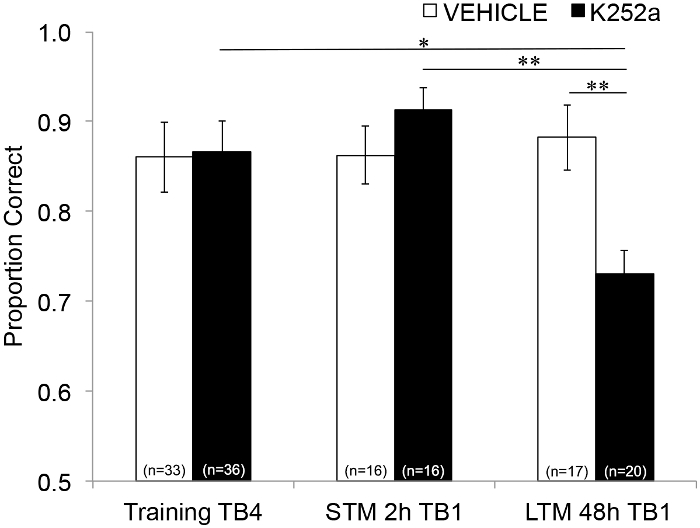
Figure 3: Differential effects of Trk receptor blockade on STM and LTM. Shows the proportion correct for Trial Block 4 of Training and Trial Block 1 of both the STM and LTM Testing. That is, a linear mixed model is used to compare the proportion correct during the last trial block of the training phase (Figure 2; Training-TB4) to those during the first trial blocks (Testing-TB1) of short-term (2-hours test) and long-term (48-hour test) memory test. The linear mixed model had two fixed effects: drug group and trial block (Training-TB4, STM-TB1, LTM-TB1). The random effects were mouse and odor set nested within the mouse. Post hoc comparisons found that K252a mice had significantly impaired LTM (comparison with training TB4; p = 0.018) but not STM (p > 0.05). LTM performance by K252a-infused mice was also significantly lower than the STM of K252a-infused mice (p = 0.009), and lower than LTM of vehicle mice (p = 0.006). Error bars represented the SEM. This figure is adapted from Tong et al. 2018 with permission3. Please click here to view a larger version of this figure.

Supplemental File: Syntax used for the statistical analysis. Please click here to download this file.
Discussion
One-trial fear conditioning is a powerful behavioral protocol for studying molecular and cellular dynamics of memory, but much of natural learning is incremental and best modeled through a paradigm like the one described above. The inhibition of Trk receptors in the OB prevented the consolidation of olfactory memory in a multi-trial, appetitive learning paradigm as shown previously by our group3. The finding opens new avenues for research into the differential timing of molecular mechanisms, like neurotrophins, in appetitive and aversive learning.
This experiment consisted of two critical parts: (1) the cannulation and (2) the associative discrimination task (subdivided into shaping, training, and testing). Experimenters can adapt this protocol to their specific research question. For example, we were primarily interested in the OB and this established protocol can be easily applied to other OB studies. For experimenters with other regions of interest, it will be important to validate the infusion sites in a pilot study. Experimenters may also need to consider the diffusion rate, spatial penetration, and bio-activity duration of what they are infusing.
The shaping steps outlined in the protocol have been used extensively by the authors of this protocol. Adhering to them as described appears to be important to have the mice learn the task in a timely manner. Using other timelines, the authors observed more variance amongst the mice in their familiarity with the task and this meant additional training to get all mice to a criterion for testing with experimental odors. For training and testing, the researcher has flexibility, depending on their research interests, with the number of trials, the concentration of the experimental odors, and the similarity of the odors to one another. We recommend the use of multiple odor sets when possible, as we’ve described, in order to reduce the number of animals used for the experiment. See the Statistical Analysis section for the guidance on how to account for the use of multiple order sets in the final analysis. In principle, the similarity of the odor pair can be varied to adjust the difficulty of the discrimination. In the previously published study from our group3, the odor pairs consist of two odorants of the same functional group but differed from each other by one carbon length. These discriminations are more difficult than pairs that differ by two or more carbon lengths, but easier than enantiomers (e.g., (+)-limonene and (-)-limonene). Odorants from different functional groups are highly different perceptually. Cleland et al13 discusses more stimulus variations and their effect on specific learning parameters.
One major limitation of this protocol is that it takes a much longer time to carry out compared to automated associative learning tasks where multiple animals could be tested in parallel. For a given test, it would take one researcher at least 20 minutes to complete 20 trials for one mouse. However, it is this lack of automation that means the protocol is more financially accessible, a priority for many institutions. Importantly, in the case of this experiment, it has been found that this protocol is highly tractable and effective for training undergraduate researchers with interests in behavioral neuroscience. In particular, these students develop strong animal handling skills in addition to the usual benefits of research participation.
Researchers who are interested in adopting this paradigm can vary several parameters. Most apparently, pharmacological manipulations of mechanisms are diverse, and this behavioral protocol can be used with chemogenetic techniques or various other ways of manipulating molecular and cellular pathways (e.g. optogenetics). The paradigm itself can be adjusted to vary the kind of learning and memory tested. For example, researchers can adjust the similarity of the two odors presented in order to control the learning rate. In our study3, the odor pairs consist of two odorants of the same functional group but differed from each other by one carbon length. These discriminations are more difficult than pairs that differ by two or more carbon lengths, but easier than enantiomers (e.g. (+)-limonene and (-)-limonene). Odorants from different functional groups are highly different perceptually. Cleland et al13 discusses more stimulus variations and their effect on specific learning parameters, concluding that more similar odors are more difficult to discriminate and therefore take longer to learn14. These manipulations would also affect the strength of memories. Along this vein, researchers may be interested in testing memory at different timepoints after learning. For example, two studies15,16 examined the role of BDNF in LTM persistence for a one-trial aversive learning task. BDNF is a ligand of TrkB. The studies showed that anti-BDNF antisense oligonucleotide infusion in the hippocampus 12 hours after learning blocked LTM 7 days later, but not 2 days later. This study shows that the timescale of molecular mechanisms after initial learning play interesting, and yet-to-be-understood, roles in LTM. This paper describes a behavioral protocol that would allow for the investigation of these timescales. Other parameters of interest for future application include the mouse model used. For example, it would be interesting to replace male mice (who have a significantly better studied neurobiology)17 with female mice in future studies to examine variations in the learning rate, STM, and LTM, as female mammals have a higher sensitivity and selectivity to odors than male mammals18. Of course, rodent models of diseases can also be used effectively with this protocol.
Disclosures
The authors of this paper had no competing financial interests.
Acknowledgements
The project was supported by the Scantland Summer Collaborative Research Gift and the Stephen and Sylvia Tregidga Burges Endowed Research Fund. The authors would like to acknowledge colleagues in the Department of Psychology and the Neuroscience Program at Earlham College for their support and guidance.
Materials
| Name | Company | Catalog Number | Comments |
| Double guide cannula | PlasticsOne | C235GS-5-1.5/SPC | Custom order |
| (-)-limonene | Sigma-Aldrich | 218367-50G | |
| (+)-limonene | Sigma-Aldrich | 183164-100ML | |
| 2-hetanone | Sigma-Aldrich | 537683 | |
| 2-octanone | Sigma-Aldrich | O4709 | |
| 5mg sucrose pellets | Test Diet | 1811560 | Custom size. Used for rewards |
| Butanoic acid | Sigma-Aldrich | B103500 | |
| butyl acetate | Sigma-Aldrich | 402842 | |
| Dental Cement Powder (Coral) | A-M Systems | 525000 | |
| Dental Cement Solvent | A-M Systems | 526000 | |
| Double connector assembly | PlasticsOne | C232C | |
| Double dummy cannula | PlasticsOne | C235DCS-5/SPC dummy dbl | Custom order |
| Double injector | PlasticsOne | C235IS-5/SPC | Custom order |
| Drill | Kopf Instruments | Model 1474 High Speed Stereotaxic Drill | This drill requires an additional "adaptor" piece in order to fit certain drill bits. We get by this problem by wrapping the drill bit with lab tape to increase the circumference of the drill it to fit. This may not be an option for surgeries requiring sterile technique. |
| Eye Ointment | Purchase from local pharmacy | ||
| Figure 1 illustration software | BioRender | ||
| heptanal | Sigma-Aldrich | W254002 | |
| hexanal | Sigma-Aldrich | 115606 | |
| hexanol | Sigma-Aldrich | H13303 | |
| Infusion pump model 11 | Harvard Apparatus | 4169D | Used pumps available via American Instrument Exchange |
| Isoflurane | Santa Cruz Animal Health | sc-363629Rx | Vet prescription needed for order |
| K252a | Sigma-Aldrich | K2015 | Mixed to 50uM in DMSO (5%) |
| Ketoprofen | Allivet | 25920 | Vet prescription needed for order |
| Lidocaine | Aspercreme | Purchased from Amazon | |
| Mounting Screws | PlasticsOne | 00-96 X 3/32 | |
| Mouse Anesthesia Mask | Kopf Instruments | Model 907 Mouse Anesthesia Mask | Used with the stereotaxic to allow oxygen and anesthesia while mouse in stereotax |
| Mouse Nose Adaptor | Kopf Instruments | Model 926 Mouse Adaptor | Used with the stereotaxic to allow for head of mouse to be secured. |
| Novalsan | Jeffers | 41375 | |
| Pentanoic acid | Sigma-Aldrich | 240370 | |
| pentanol | Sigma-Aldrich | 138975 | |
| Petri dish glass bottoms | VWR | 10754-804 | |
| Polycarbonate Café bottoms | Ancare | N10PCSEC | Use normal housing cages and custom fit a track in the middle to act as the track for an opaque plexiglass divider |
| propyl acetate | Sigma-Aldrich | 537438 | |
| Quikrete Premium Play Sand | Purchase from local hardware store | ||
| Saline | Insight Needles | N/A | Sterile saline for drug mixing |
| Stereotaxic apparatus | Kopf Instruments | Model 902 Small Animal Stereotaxic Instrument | |
| Testing chamber | Ancare | N10PCSEC | Our testing chambers are modified using the regular mouse housing cage. The manuscript details what was done. |
| Vetbond Tissue Adhesive | 3M | Purchased from Amazon |
References
- Grimes, M. T., Smith, M., Li, X., Darby-King, A., Harley, C. W., Mclean, J. H. Mammalian Intermediate-Term Memory: New Findings in Neonate Rat. Neurobiology of Learning and Memory. 95 (3), 385-391 (2011).
- Grimes, M. T., Harley, C. W., Darby-King, A., Mclean, J. H. PKA Increases in the Olfactory Bulb Act as Unconditioned Stimuli and Provide Evidence for Parallel Memory Systems: Pairing Odor with Increased PKA Creates Intermediate- and Long-Term, but not Short-Term, Memories. Learning and Memory. 19 (3), 107-115 (2012).
- Tong, M. T., Kim, T. Y. P., Cleland, T. A. Kinase Activity in the Olfactory Bulb is Required for Odor Memory Consolidation. Learning & Memory. 25 (5), 198-205 (2018).
- Devore, S., Lee, J., Linster, C. Odor Preferences Shape Discrimination Learning in Rats. Behavioral Neuroscience. 127 (4), 498-504 (2013).
- Gao, Y., Strowbridge, B. W. Long-Term Plasticity of Excitatory Inputs to Granule Cells in the Rat Olfactory Bulb. Nature Neuroscience. 12 (6), 731-733 (2009).
- Bath, K. G., Mandairon, N., et al. Variant Brain-Derived Neurotrophic Factor (Val66Met) Alters Adult Olfactory Bulb Neurogenesis and Spontaneous Olfactory Discrimination. Journal of Neuroscience. 28 (10), 2383-2393 (2008).
- Lazarini, F., Lledo, P. M. Is Adult Neurogenesis Essential for Olfaction. Trends in Neurosciences. 34 (1), 20-30 (2011).
- Mandairon, N., Peace, S., Karnow, A., Kim, J., Ennis, M., Linster, C. Noradrenergic Modulation in the Olfactory Bulb Influences Spontaneous and Reward-Motivated Discrimination, but not the Formation of Habituation Memory. European Journal of Neuroscience. 27 (5), 1210-1219 (2008).
- Guérin, D., Peace, S. T., Didier, A., Linster, C., Cleland, T. A. Noradrenergic Neuromodulation in the Olfactory Bulb Modulates Odor Habituation and Spontaneous Discrimination. Behavioral Neuroscience. 122 (4), 816-826 (2008).
- Moreno, M. M., Bath, K., Kuczewski, N., Sacquet, J., Didier, A., Mandairon, N. Action of the Noradrenergic System on Adult-Born Cells Is Required for Olfactory Learning in Mice. Journal of Neuroscience. 32 (11), 3748-3758 (2012).
- Vinera, J., Kermen, F., Sacquet, J., Didier, A., Mandairon, N., Richard, M. Olfactory Perceptual Learning Requires Action of Noradrenaline in the Olfactory Bulb: Comparison with Olfactory Associative Learning. Learning and Memory. 22 (3), 192-196 (2015).
- Dillon, T. S., Fox, L. C., Han, C., Linster, C. 17β-estradiol Enhances Memory Duration in the Main Olfactory Bulb in CD-1 Mice. Behavioral Neuroscience. 127 (6), 923 (2013).
- Cleland, T. A., Narla, V. A., Boudadi, K. Multiple Learning Parameters Differentially Regulate Olfactory Generalization. Behavioral Neuroscience. 123 (1), 26 (2009).
- Cleland, T. A., Morse, A., Yue, E. L., Linster, C. Behavioral Models of Odor Similarity. Behavioral Neuroscience. 116 (2), 222-231 (2002).
- Bekinschtein, P., Cammarota, M., Igaz, L., Bevilaqua, L., Izquierdo, I., Medina, J. Persistence of Long-Term Memory Storage Requires a Late Protein Synthesis- and BDNF- Dependent Phase in the Hippocampus. Neuron. 53 (2), 261-277 (2007).
- Bekinschtein, P., et al. BDNF is Essential to Promote Persistence of Long-Term Memory Storage. Proceedings of the National Academy of Sciences. 105 (7), 2711-2716 (2008).
- Andreano, J., Cahill, L. Sex influences on the neurobiology of learning and memory. Learning and Memory. 16 (2009), 248-266 (2009).
- Kass, M. D., Czarnecki, L. A., Moberly, A. H., Mcgann, J. P. Differences in Peripheral Sensory Input to the Olfactory Bulb Between Male and Female Mice. Scientific Reports. 7 (1), (2017).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved