Establishment of an Animal Training Model of Duodenoscopy, Choledochoscopy, and Laparoscopy for Cholelithiasis
In This Article
Summary
Here, we present a protocol for establishing a multi-scopy combined animal training model to enhance the proficiency and comprehension of young doctors in multi-scopy combined technology.
Abstract
Cholelithiasis is a condition characterized by the formation of stones in the gallbladder and/or bile ducts. Proficient use of duodenoscopy, laparoscopy, and choledochoscopy techniques plays a pivotal role in managing cholelithiasis. To enhance young doctors' proficiency in multiendoscope technology application, a training model for duodenoscopy, choledochoscopy, and laparoscopy was established using a live pig model. Initially, the duodenoscope was used to access the duodenum through passage through the mouth, esophagus, and stomach to observe the duodenal papilla. Subsequently, the tube was inserted into the common bile duct, followed by the placement of the guide wire. Trocars were subsequently inserted to establish pneumoperitoneum and facilitate the performance of laparoscopic cholecystectomy. The common bile duct was explored and opened with the retrieval of the guide wire found within it. The choledochoscope was then used to explore both distal common bile duct and intrahepatic bile ducts. Finally, under duodenoscopy guidance, either nasobiliary tube or plastic biliary stent placement occurred before primary suture closure of the common bile duct. Establishing a multi-scopy combined animal model can further enhance young doctors' proficiency in multi-scopy combined technology through practical experience.
Introduction
Cholelithiasis is a common disease of the digestive system, characterized by the formation of stones in the gallbladder and/or bile duct. The incidence of gallstones in adults is estimated to be between 10% and 20%1,2, with approximately 5% to 15% of patients also experiencing choledocholithiasis or hepatolithiasis. Choledocholithiasis can lead to serious complications such as acute cholangitis or biliary pancreatitis.
Common bile duct stones (CBDS) often occur due to migration from the gallbladder, although a few may develop directly in the bile duct. Coexistent common bile duct stones are found in 3%-16% of patients with symptomatic gallbladder stones3,4. According to the evidence-based clinical practice guidelines for cholelithiasis 2021, endoscopic CBDS removal plus surgical cholecystectomy and surgical CBDS removal plus cholecystectomy are recommended for CBDS with gallbladder stones5,6. For minimally invasive and fast recovery, endoscopic retrograde cholangiography (ERCP) plus laparoscopic cholecystectomy (LC) or laparoscopic common bile duct exploration T-tube choledochotomy (LCBDE) plus LC are the usual management in the clinic. These treatments require using multi-scopy combined techniques such as ERCP technique combined with laparoscopic technique or laparoscopic technique combined with choledochoscopic technique. These techniques are now mainly therapeutic procedures, and lack of experience leads to a higher risk of adverse events7,8. Thus, training and teaching multi-scopy combined techniques have gained importance9. However, there is no good model for training and teaching multi-scopy combined techniques in place currently.
Here, we present a training protocol for multi-scopy combined techniques involving duodenoscopy, choledochoscopy, and laparoscopy using a live pig model. The establishment of a multi-scopy combined animal model can enhance the proficiency and comprehension of young doctors in multi-scopy combined technology through hands-on experience. This will contribute to their overall skill development and understanding in this field.
Protocol
The procedure of live pig experiments was performed in accordance with international rules and was approved by the Committee of Animal Care (WebLab020-FY202403).
1. Model preparation
- Subject the mini-pig to a fasting period of 12 h and withhold access to drinking water on the morning of the procedure.
- Perform endotracheal intubation and provide 0.5-2% isoflurane in a 1:1 ratio with oxygen at a flow rate of 5-10 mL/kg/min to initiate general anesthesia. Keep the eyes of the pig closed with film dressing.
NOTE: The somatokinetic response is used to evaluate the depth of anesthesia. This assessment is deemed appropriate when the pig exhibits non-responsiveness while both heart rate and respiration remain stable during the operation. - Position the pig on an operating table in the supine position for experiments.
- Monitor the heart rate and oxygen saturation using an electrocardiogram monitor.
- Sterilize the abdominal skin with iodophor, then cover it with sterile towels.
2. ERCP procedure (Figure 1)
- Check the duodenoscope in conjunction with the imaging system and its functionalities, including water draw, CO2 intake, and suction capabilities.
- Hold the duodenoscope and insert it into the mouth, pass through the esophagus and into the stomach cavity (Figure 1A, B).
- Navigate through the pylorus (Figure 1C) and into the duodenum to observe the duodenal papilla (Figure 1D).
NOTE: Usually, passing through the pylorus is hard and easy to slip back because the pyloric muscle is hypertrophic, and the position of the duodenal papilla is near the pylorus. The person holding and controlling the duodenoscope should be well-trained in this procedure. - Use a sphincterotome with a biliary guidewire to cannulate the duodenal papilla (Figure 1E).
- Cannulate the common bile duct (CBD) with a guidewire and retain it in the CBD (Figure 1F).
3. Establishment of pneumoperitoneum
- Incise the skin above the umbilicus with 10 mm, then use two towel forceps to clamp and lift the skin on both sides of the incision. Insert the Veress needle into the abdominal cavity through the incision.
- If it fails, incise the abdominal wall layer by layer to the depth to reach the abdominal cavity.
- Inject carbon dioxide gas through the Veress needle (connected to a carbon dioxide gas pump) and maintain a pneumoperitoneum pressure of 12 mmHg.
4. Trocar placement
- Position the observation port in proximity to the umbilicus to facilitate a comprehensive exploration of the abdominal cavity.
- Insert a 12 mm trocar about 2 cm below the xiphoid process after incising the skin.
- Insert a 5 mm trocar roughly 2 cm below the costal margin of the right midclavicular line after incising the skin.
- Insert a 5 mm trocar 2 cm below the costal margin of the right anterior axillary line after incising the skin.
NOTE: Care should be taken to avoid damaging any intestines that may be adhered to the abdominal wall.
5. LC procedure (Figure 2)
- Hold back the liver lobe to expose the gallbladder with a stomach clamp.
- Grasp the gallbladder ampulla to expose the gallbladder triangle by a separate clamp.
- Dissect the cystic duct and cystic artery carefully. Then, ligature and disconnect them (Figure 2B).
- Gently detach the gallbladder from behind the liver until completely removed (Figure 2A).
6. LCBDE procedure (Figure 3)
- Hold back the liver lobe to expose the position of CBD by a stomach clamp.
- Separate the CBD until the complete dissociation using an electric hook (Figure 3A).
- Dissect the CBD longitudinally to expose the guidewire in the CBD (Figure 3B).
- Employ a choledochoscope to explore the lower part of the CBD and duodenal papilla downward (Figure 3C, E), and the common hepatic duct and intrahepatic bile duct upward (Figure 3D, F).
NOTE: In cases where residual stones are present, endoscopic basket extraction may be performed. Consider either primary closure or T-tube placement for optimal management, depending on the diameter of the CBD.
7. Postoperation
- Suture the abdominal incisions and euthanize the pig with 20 mL of potassium chloride injection under anesthesia.
Results
The training protocol for multi-scopy combined techniques was performed with a 35 kg female domesticated mini-pig. After the pig was anesthetized and monitored, the abdomen skin was sterilized and covered with towels. The duodenoscope was inserted into the mouth, passed through the esophagus and into the stomach cavity (Figure1A,B), guided through the pylorus (Figure 1C) and into the duodenum, where the duodenal papilla (Figure 1D) could be observed. The CBD was selectively cannulated with a sphincterotome and guidewire (Figure 1E), which was then kept in place (Figure 1F). Following the establishment of pneumoperitoneum and placement of trocars, laparoscopic cholecystectomy was performed (Figure 2). The CBD was separated with an electric hook (Figure 3A) and dissected longitudinally with the guidewire exposed (Figure 3B). The whole CBD and intrahepatic bile duct were explored using a choledochoscope (Figure 3C-F). Primary closure was then performed before removal of equipment and suturing of incisions. Finally, under anesthesia, euthanasia of the pig took place.
We conducted this procedure on five pigs. Initially, we perfomed the procedure on a single pig, which yielded successful results. Following this, we assembled 20 trainees to present the protocol and procedural videos, providing training with an additional four pigs. Among these trainees, four experienced ERCP practitioners successfully completed the ERCP procedure within 30 min. In contrast, eight trainees with little or no prior experience in ERCP encountered difficulties in initial 30-min timeframe; however, four of them were able to complete the ERCP procedure after two or three trials with assistance from tutors in the subsequent 30 min. Additionally, four trainees successfully performed the LC procedure while other four trainees accomplished the LCBDE procedure in 30 min. Thus, a total of four trainee groups successfully completed the protocol (Table 1).
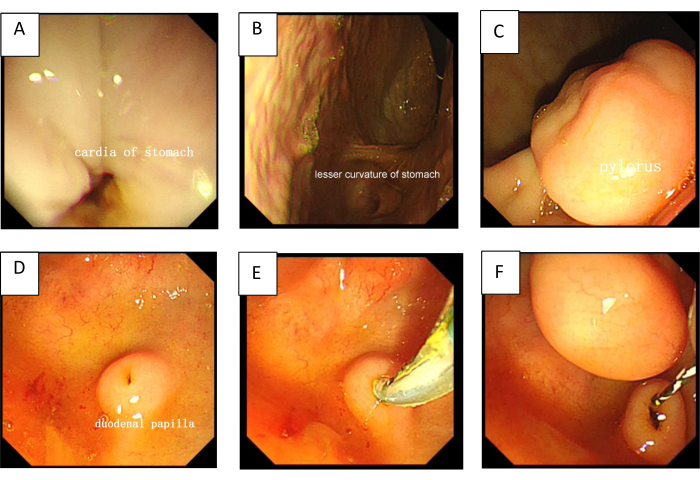
Figure 1: The procedure of ERCP. (A) The cardia of the stomach. (B) The lesser curvature of the stomach. (C) The pylorus. (D) The duodenal papilla. (E) A sphincterotome with a biliary guidewire to cannulate the duodenal papilla. (F) After selectively cannulating the common bile duct (CBD), the guidewire was retained in the common bile duct. Please click here to view a larger version of this figure.
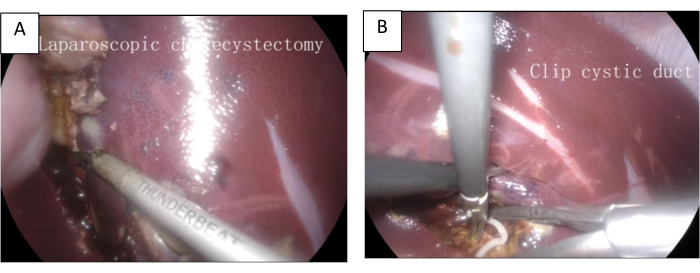
Figure 2: The procedure of LC. (A) The gallbladder is separated from behind the liver. (B) Clip the cystic duct. Please click here to view a larger version of this figure.
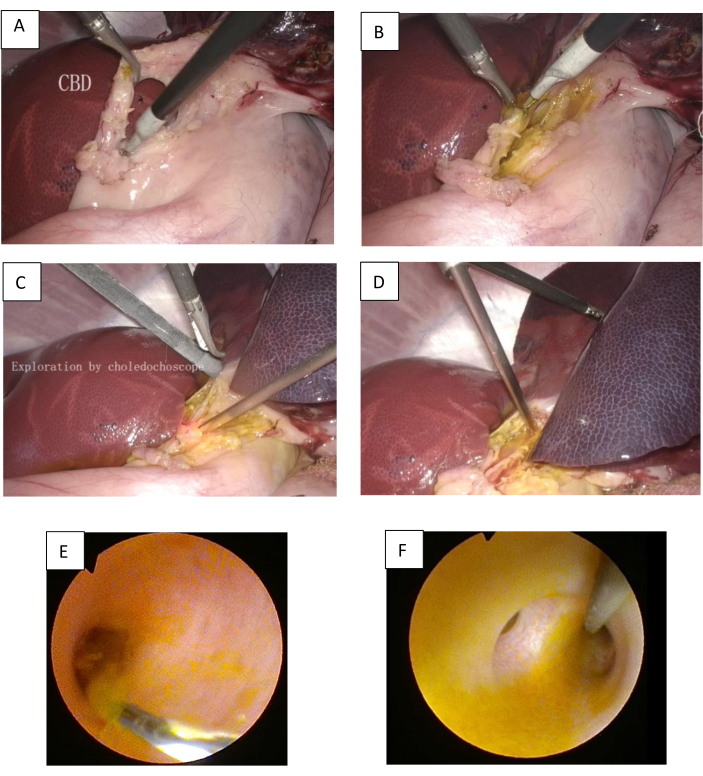
Figure 3: The Procedure of LCBDE: (A) Separate the CBD. (B) The CBD was dissected longitudinally to expose the guidewire. (C) A choledochoscope was used to explore the lower part of the CBD and duodenal papilla downward. (D) A choledochoscope was used to explore the common hepatic duct and intrahepatic bile duct upward. (E) The lower part of the CBD. (F) The hilar bile duct. Please click here to view a larger version of this figure.
| Description | Number of trainees | Procedure | |
| Successful ERCP procedure completion in 30 min | 4 | ERCP | |
| Non experienced trainees with difficulties in the initial 30 min | ERCP | ||
| Non experienced with difficulties after 2-3 trials with assistance from tutors in the subsequent 30 min | 4 | ERCP | |
| Non experienced with successful completion after 2-3 trials with assistance from tutors in the subsequent 30 min | 4 | ERCP | |
| Successful LC procedure completion in 30 min | 4 | LC | |
| Successful LCBDE procedure completion in 30 min | 4 | LCBDE | |
| Total number of trainees | 20 | ||
Table 1: Representative data from the training program.
Discussion
Cholelithiasis is a common and recurrent disease that presents challenges in management. With the advancement of technology, minimally invasive techniques, especially the multi-scopy combined technique, have been increasingly utilized in the treatment of cholelithiasis. According to the size and distribution of stones, the stone processing patterns can be classified as follows: (1) For CBDS smaller than 1 cm, with or without gallbladder stones, it is suggested to remove the stone using ERCP with slow balloon expansion to preserve nipple function, with or without LC. (2) For CBDS larger than 1 cm, with or without gallbladder stones, it is recommended to remove stones using electrohydraulic lithotripsy or a lithotripsy basket after slow balloon dilation under nipply-preserving ERCP, with or without LC. Alternatively, stones can be removed through LCBDE with or without LC. Biliary stent placement under ERCP combined with LC and LCBDE without T-tube is also a viable option. (3) In cases of intrahepatic bile duct stones, the treatment approach may involve laparoscopic hepatectomy or percutaneous transhepatic cholangioscopy, depending on the situation and distribution of calculi. A single treatment using either laparoscopy or duodenoscopy often fails to achieve optimal efficacy for patients with gallbladder and CBD stones. The combined application of multi-scopy has brought revolutionary changes in the minimally invasive treatment of cholelithiasis10. Lv et al.11 demonstrated that the tri-endoscopic approach for concomitant cholecystolithiasis and choledocholithiasis is both safe and effective, resulting in shorter hospital stays and reduced costs. It is evident that proficient skills in duodenoscopy, laparoscopy, and choledochoscopy techniques play a crucial role in the treatment of cholelithiasis. Because lack of experience could lead to a higher risk of adverse events, training and teaching seem to have gained importance. Traditionally, ERCP techniques have been trained using computerized simulators12 or ex vivo13 training models, while laparoscopic skills have been developed through dry box14 or ex vivo box15 methods. However, these training models often fail to integrate the advantages of both ERCP and laparoscopic techniques effectively. Therefore, there is an urgent need for a novel training model that facilitates combined techniques for multi-scopy training.
We developed a live pig model for training in combined multi-scopy techniques. The representative results indicate that this model is effective (Table 1). Techniques such as ERCP, laparoscopic surgery, and choledochoscopy can all be effectively trained using this model. This model presents several unique advantages. Firstly, it is beneficial for simulating human physiological and anatomical structures. For instance, it provides a realistic simulation of the physiological digestive tract for training in duodenoscopy control, as well as opportunities to practice bleeding management and biliary tract suturing using laparoscopic techniques16. The pig model offers a valuable training environment for novice physicians. Secondly, this pig model facilitates training in hybrid techniques. In instances where duodenoscopy proves unsuccessful, the gastroscope can be inserted into the duodenum instead. A laparoscopic guidewire may then be introduced into the common bile duct via the cystic duct and subsequently advanced into the duodenum through the papilla. Following observation under gastroscopic guidance, forceps can be employed to grasp and maneuver the guidewire out of the mouth before reverting to a duodenoscope to navigate the wire along its intended path into the duodenum. Furthermore, this model emphasizes teamwork during training sessions by necessitating close collaboration among team members. This approach fosters a spirit of teamwork and enhances communication skills among young doctors. Lastly, this training model embodies characteristics of minimally invasive surgery by prioritizing reduced trauma. It aims to enhance young doctors' understanding and proficiency in minimally invasive surgical practices.
For ethical considerations, we selected a live pig as the training model due to its size and anatomical structures that closely resemble those of humans17. Its substantial size allows for the use of all equipment and apparatus designed for human applications. Furthermore, its anatomical features provide valuable opportunities for young doctors to practice duodenoscopy control. Among existing animal models, pigs are uniquely suitable for training in multi-scopy techniques. During the operation, no pain management strategies were employed; consequently, the pig exhibited non-responsiveness while both heart rate and respiration remained stable throughout the procedure. The euthanasia process was conducted swiftly and effectively using an injection of 20 mL of potassium chloride under anesthesia.
However, this pig model has several limitations. Firstly, the associated costs are high. The primary expenses of this model arise from the procurement of pigs, as well as the necessary equipment and apparatus, which may hinder its widespread adoption. Secondly, the pig is intended for single use only. Due to in vivo experiments conducted under physiological conditions and the operational damage inflicted on the biliary system, it is challenging to utilize pigs for further studies. Consequently, this also contributes to increased experimental costs. Furthermore, this pig model does not accurately represent a cholelithiasis model. Locating a pig with naturally occurring cholelithiasis poses significant difficulties; additionally, creating an artificial cholelithiasis pig model may not be permissible according to animal care committee regulations. Nevertheless, it remains the ideal model for training in multi-scopy for cholelithiasis due to its appropriate size and similarity to human physiological and anatomical structures. Even in the absence of stones in the bile duct, the training treatment procedure for cholelithiasis can still be performed effectively.
The model may be improved in the future by utilizing whole abdominal organs sourced from slaughterhouses, supported by artificial circulation systems within a wet box to simulate physiological and anatomical conditions. This approach is anticipated to be more cost-effective. With advancements in technology and artificial intelligence18, it is possible to create a multi-endoscopy training artificial simulator using virtual or augmented reality19,20. This development fundamentally transforms the issue of non-reusability associated with the current model.
The model for multi-scopy combined techniques training in the treatment of cholelithiasis is an innovative and practical teaching method. It is helpful for improving young doctors' proficiency and application ability of multi-scopy combined technology. This method provides valuable hands-on experience for medical professionals to enhance their skills in treating cholelithiasis.
Disclosures
The authors have no conflicts of interest or financial ties to disclose.
Acknowledgements
We thank the OLYMPUS China Technical Education Center (Guangzhou) for helping with the study.
Materials
| Name | Company | Catalog Number | Comments |
| Anesthesia machine | Mindray | WATO EX-20 | |
| Choledochoscope | OLMPLUS | CHF-V2 | |
| Duodenoscopy | OLMPLUS | TJF-260V | |
| Electric hook | OLMPLUS | A6292 | |
| Electrocardiogram monitor | Mindray | iMEC12 | |
| Electrocoagulation system | OLMPLUS | ESG-400/USG-400 | |
| Guidewire | OLMPLUS | G-240-3527A | |
| Laparoscopic gastric forceps | OLMPLUS | WA64130L | |
| Laparoscopic needle holder | OLMPLUS | WA64700A | |
| Laparoscopic separation forceps | OLMPLUS | WA64320A | |
| Laparoscopy | OLMPLUS | OTV-S400 | |
| Sphincterotome | OLMPLUS | KD-V431M-0720 | |
| Trocar (5.5 mm, 12 mm) | OLMPLUS | 5.5-A5809/12-A5859 | |
| Ultrasonic scalpel and equipment | OLMPLUS | TB-0520FCS/TD-TB400 | |
| Veress needle | OLMPLUS | SD-301 | |
| Vicryl Plus 5-0 | ETHICON | VCP433H | |
| Video recording system | OLMPLUS | IMH-20 |
References
- Lammert, F. et al. Gallstones. Nat Rev Dis Primers. 2, 16024 (2016).
- Lammert, F. Wittenburg, H. Gallstones: Prevention, diagnosis and treatment. Semin Liver Dis. 44 (3), 394-404 (2024).
- Collins, C., Maguire, D., Ireland, A., Fitzgerald, E., O'sullivan, G. C. A prospective study of common bile duct calculi in patients undergoing laparoscopic cholecystectomy: Natural history of choledocholithiasis revisited. Ann Surg. 239 (1), 28-33 (2004).
- Caddy, G. R. Tham, T. C. Gallstone disease: Symptoms, diagnosis and endoscopic management of common bile duct stones. Best Pract Res Clin Gastroenterol. 20 (6), 1085-1101 (2006).
- Fujita, N. et al. Evidence-based clinical practice guidelines for cholelithiasis 2021. J Gastroenterol. 58 (9), 801-833 (2023).
- Dahiya, D. S. et al. Understanding evidence-based clinical practice guidelines for cholelithiasis 2021. Hepatobiliary Surg Nutr. 13 (2), 352-355 (2024).
- Fried, G. M. Lessons from the surgical experience with simulators: Incorporation into training and utilization in determining competency. Gastrointest Endosc Clin N Am. 16 (3), 425-434 (2006).
- Kachaamy, T. A. Faigel, D. O. Improving ERCP quality and decreasing risk to patients and providers. Expert Rev Gastroenterol Hepatol. 7 (6), 531-540 (2013).
- Loganathan, P. et al. Trainee involvement and ERCP complications: A systematic review and meta-analysis. Dig Dis Sci. 69 (7), 2363-2369 (2024).
- Zhang, Z. et al. Strategies of minimally invasive treatment for intrahepatic and extrahepatic bile duct stones. Front Med. 11 (4), 576-589 (2017).
- Lv, F. et al. Single-stage management with combined tri-endoscopic approach for concomitant cholecystolithiasis and choledocholithiasis. Surg Endosc. 30 (12), 5615-5620 (2016).
- Sahakian, A. B. et al. Can a computerized simulator assess skill level and improvement in performance of ERCP? Dig Dis Sci. 61 (3), 722-730 (2016).
- Velazquez-Avina, J., Sobrino-Cossio, S., Chavez-Vargas, C., Sulbaran, M., Monkemuller, K. Development of a novel and simple ex vivo biologic ERCP training model. Gastrointest Endosc. 80 (6), 1161-1167 (2014).
- Deie, K. et al. Evaluation of minimally invasive surgical skills training: Comparing a neonatal esophageal atresia/tracheoesophageal fistula model with a dry box. Surg Endosc. 36 (8), 6035-6048 (2022).
- Zhang, H. et al. Development of a continuously perfused ex vivo kidney training model for laparoscopic partial nephrectomy: Validity and efficiency. Int J Surg. 109 (12), 3919-3928 (2023).
- Phillips, M. S. Marks, J. M. Overview of methods for flexible endoscopic training and description of a simple explant model. Asian J Endosc Surg. 4 (2), 45-52 (2011).
- Jeong, S., Park, J. S., Lee, D. H. Large animal models in pancreas and biliary disease. Korean J Gastroenterol. 77 (3), 99-103 (2021).
- Ryder, C. Y. et al. Using artificial intelligence to gauge competency on a novel laparoscopic training system. J Surg Educ. 81 (2), 267-274 (2024).
- Bogar, P. Z. et al. Validation of a novel, low-fidelity virtual reality simulator and an artificial intelligence assessment approach for peg transfer laparoscopic training. Sci Rep. 14 (1), 16702 (2024).
- Koch, K. et al. Development and evaluation of artificial organ models for ERCP training in patients with surgically altered anatomies. Sci Rep. 13 (1), 22920 (2023).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved