A subscription to JoVE is required to view this content. Sign in or start your free trial.
Design and Validation of a Volumetric-extrusion Bioprinter for Bioprinting of Soluble Basement Membrane Extract for Translational Research
In This Article
Summary
Soluble basement membrane extracts are the most widely used biological matrices in cancer research, but their complex rheological behaviour makes their bioprinting difficult with commercially available bioprinting systems. This work presents a customized bioprinting strategy to produce pure matrix constructs with good shape fidelity in both single and multiple layers.
Abstract
In translational research, 3D in vitro models are essential for advancing scientific understanding and therapeutic development. To this aim, Soluble Basement Membrane Extracts (SBMes), derived from mouse sarcoma cells, play the key role of replicating mechanical and biochemical features of in vivo microenvironment. They are particularly valuable in a variety of research fields, from tissue engineering to drug testing. In particular, they have become fundamental in cancer research as traditional 2D cell cultures, widely used in the field, can provide misleading information since they do not capture the three-dimensional (3D) structure and tumor microenvironment.
In cancer research, effective 3D in vitro models are crucial for better understanding cancer evolution and anticipating challenges, such as drug resistance mechanisms. Organoids have emerged as promising 3D in vitro models, offering a more accurate representation of tumor biology. They grow in SBMes as they create an environment where cells can grow and self-organize into 3D structures. However, SBMe presents significant challenges, including low mechanical properties and complex rheological behavior, preventing its use with commercially available pneumatic-extrusion bioprinting systems.
To address these limitations, we developed a low-cost, volumetric-controlled bioprinting system and a specific protocol for printing structures with SBMe. This system is based on a volumetric extruder and overcomes the limitations of traditional pneumatic-driven approaches. Once assembled and programmed with a specific g-code, the system can bioprint both pure and diluted SBMe to obtain both single and multi-layer constructs. This approach offers a more reliable and scalable method for producing 3D cell culture models, paving the way for more effective research into novel treatments and drug testing.
Introduction
In translational research, creating 3D in vitro models that closely replicate the in vivo environment is essential for advancing scientific understanding and therapeutic development1. A key component in building them is the use of extracellular matrix (ECM) analogs, such as Soluble Basement Membrane Extracts (SBMes), which better reflect the complexities of natural tissues. SBMes are particularly valuable in a variety of research fields, from tissue engineering to drug testing2,3. In particular, they have become fundamental in cancer research, as they replicate mechanical and biochemical features of the tumor microenvironment, enabling the development of models that better reflect tumor growth, metastasis, and treatment responses.
Indeed, despite considerable advancements over the past few decades, many therapies continue to fail, leading to frequent treatment failure in clinical settings. This failure is often attributed to drug resistance mechanisms caused by high levels of cell heterogeneity4,5. Effective in vitro models, capable of accurately recapitulating the complexities of human tumors, are crucial to better understanding cancer evolution and anticipating this phenomenon.
Traditional 2D cell cultures have been foundational in cancer research but can lead to misleading information about drug efficacy and tumor behavior, as they fail to capture the 3D structure and microenvironment of actual tumors6,7,8. In contrast, 3D in vitro models have emerged as promising alternatives, offering a more accurate representation of tumor microenvironment and biological behavior by mimicking 3D complexity9,10.
In cancer research, organoids are widely used as 3D models for studying disease and drug efficacy, since they provide more accurate and relevant models of human tissues and organs. The use of SBMe, derived from Engelbreth-Holm-Swarm mouse sarcoma cells, has been pivotal in advancing cancer research11,12. It provides a gel-like substrate in which cells can grow, proliferate, and self-organize into organoid structures, ensuring proper development and growth13,14.
However, SBMe poses a number of challenges due to its unique and complex characteristics. As a non-Newtonian fluid, SBMe exhibits shear-thinning properties, meaning its viscosity decreases with increasing shear stress, making its handling and application inconsistent under variable forces. Additionally, its thixotropic behavior adds to the complexity, as it can recover its viscosity over time when shear stress is removed15,16. These properties, together with low mechanical strength (with storage modulus values ranging from 10 Pa to 5,000 Pa depending on protein concentration) and thermosensitive behavior, worsen these challenges, requiring meticulous control during preparation and use as well as bioprinting it pure.
Standard pneumatic-driven bioprinting systems fail to properly control SBMe dispensing as the application of pressure causes uncontrollable behavior after its ejection from the syringe. It is affected by a phenomenon similar to the "spurt" effect" described for polymeric melts, in which the flow rate abruptly increases above a certain critical pressure value17,18,19. As reported in our previous work16, the volumetric-control dispensing strategy allows uncoupling SBMe extrusion from the pressure generated in the dispenser, which is dependent on the rheological properties of the matrix, working conditions, and nozzle geometry. In recent years, several attempts have been made in the market to exploit the same principle, with Corning's Matribot leading the way. It allows the handling and deposition of SBMe and other temperature-sensitive bioinks thanks to controlled temperature conditions. However, it might be expensive, limiting accessibility for smaller labs.
In our prior paper, we have presented a low-cost custom alternative-a volumetric-driven bioprinting system16. This version was based on a modified entry-level 3D printer with a custom 3D printed extruder. Despite its effectiveness in bioprinting, it presented some limitations in printing repeatability. Being derived from an entry-level 3D printer, it had some problems with the Z-movements and auto-homing. To address these needs, we developed an advanced version of our custom low-cost volumetric dispensing system and a specific protocol for bioprinting structures using SBMe.
Here, we provide protocols for the construction and use of this system. It is composed of a custom volumetric-controlled extruder, based on our previous study16, a bioprinter case, and a control system. Production and assembly are made for each module. When assembled and programmed with a specific gcode file, following the protocol description for matrix handling, the system is capable of bioprinting pure or diluted constructs using the SBMe with good shape fidelity in both single and multiple layers.
Protocol
1. 3D printing and assembly of volumetric bioprinter
- Prepare and 3D-print the bioprinter components.
- Download the *.STL design files from Supplemental File 1.
- Load *.STL file in the proprietary slicing software to define the orientation of the pieces on the printing plate and all the options required for the 3D printer. Print the pieces using polylactic acid (PLA) with medium-quality options (0.4 mm nozzle size, 1.75 mm filament diameter, 220 °C extruder temperature, 50 °C print bed plate temperature, 0.2 mm layer height, 300 mm/s printing speed).
NOTE: Medium-quality options might differ according to the printer used for printing the pieces. - Detach the printed pieces from the 3D printer bed. Remove the printed supporting structures, if present, and polish surfaces where required.
- Assemble the extruder (Figure 1).
- Insert two linear ball bearings (LM4UU) in the vertical holes of the pusher block. Insert also a leadscrew nut T8 in the back central hole and secure it with four screws (M3x10).
- Attach a rigid shaft coupling to the stepper motor and then fasten it to the extruder motor housing using four screws (M3x6).
NOTE: Orient the stepper motor according to the groove in the extruder motor housing to allow proper positioning of the jack for cables. - Fasten the stepper motor, assembled in step 1.2.2, to a T8 leadscrew (150 mm) using the rigid shaft coupling previously inserted. Ensure every block is properly secured with the set screws.
- Attach one Self-Aligning Flange Bearing (8 mm inner diameter) at the bottom of the extruder base with two screws (M4x8).
- Insert two ball bearings (4 x 9 x 4 mm) in the specific holes on top of the extruder base and the other two in the holes in the lower section.
- Insert the assembled part (step 1.2.1) into the extruder base through the cavity in the upper part of the structure. Connect the four ball bearings (step 1.2.5) with two 4 mm rods, one for each side. Let the rods pass through the two linear ball bearings in the assembled part in step 1.2.1.
NOTE: It is also possible to first insert the bars on the upper two ball bearings and then put them into the extruder base until they reach the bottom ball bearing and everything is secured in the defined slots. - Insert the assembled part in step 1.2.3 in the central hole of the extruder base. Rotate the leadscrew to pass the leadscrew nut and reach the self-aligning flange bearing. Use the set screws in the component to lock the leadscrew and two screws (M3x8) to secure the extruder motor housing on the top of the extruder base.
- Use two other screws (M3x40) and M3 nuts to attach the syringe barrel flange retainer to the bottom part of the extruder base, to lock the syringe barrel flange (insert direction from top to bottom). Use two other screws (M3x30) and M3 nuts to attach the syringe piston retainer to the upper holes of the pusher block (insert direction from bottom to top) to lock the plunger flange.
- Insert two screws (M4x80) into two springs and let them pass through the holes present in the bottom part of the extruder base and the syringe block. Screw the nuts leaving the space for the syringe to avoid slipping of the syringe block.
NOTE: Screws must go from back to front.
- Assemble the X-axis (Figure 2).
- Insert four linear ball bearings (LM8UU x 24 mm) into the X_Left_Block and X_Right_Block parts in the upper and bottom holes, two for each. Attach also two belt blocks on top of either X right and X left blocks with screws (M2x10) leaving space for the belt.
- Insert two ball bearings (5 x 16 x 5 mm) in the central holes of X_Right_Block, one on each side. Place a pulley (Bore 5 mm) in the inner cavity of X_Right_Block and connect the two ball bearings with a 5 mm rod (35 mm length), letting it pass through the pulley.
NOTE: Use a longer rod for the assembly to have better handling. After positioning the pieces in place, cut the rod of the desired length. - Attach a limit switch to the X_motor_housing with two screws (M3x8). Insert NEMA 17 motor into the motor housing and use four screws (M3x8) to fix the X_block_1 cover to the motor housing and the motor.
- Attach a pulley (Bore 5 mm) to the motor and lock it with set screws and then screw together the assembled part in step 1.3.3 to X_block_1 with four screws (M5x8). Attach also a limit switch at the bottom of X_Left_Block with two screws (M3x8).
NOTE: The outer diameter of the pulleys in steps 1.3.2 and 1.3.4 must be the same. - Place two linear ball bearings (LM8UU x 45 mm) into the holes at the top and the bottom of the X_slider.
- Connect the assembled parts in steps 1.3.2 and 1.3.4 with two 8 mm rods (200 mm length) and let them pass through the assembled part in step 1.3.5.
NOTE: Cut the rods of the desired length before assembling. - Lock one belt end to the X_slider_belt_connection using the belt block and two screws (M2x8). Run the belt around the pulley in the assembled part 1.3.4, then around the pulley in the assembled part 1.3.2 and fix the other belt end to the X_slider_belt connection using the second belt block and two other screws (M2x8). Ensure the belt is properly tight to obtain the correct movement.
NOTE: During this step, the X_slider must be moved towards one end of the X-axis structure to avoid interference. - Screw the X_slider_belt_connection to the X_slider with one screw (M6x20).
- Connect the extruder to the X_slider and fix it with four screws (M5x10).
- Assemble the Y-axis (Figure 3).
- Place the fixing bracket to the up rear bioprinter part using four screws (M3x10) and four nuts.
NOTE: The position of the fixing bracket can be adapted to obtain the proper tension of the belt and allow the insertion of the stepper motor. - Insert the stepper motor into the fixing bracket and screw it with four screws (M3x8). Next, place a pulley (bore 5 mm) on the crankshaft and fix it with set screws.
NOTE: The pulley could be attached to the crankshaft before putting the motor into the fixing bracket to avoid some interference between components. - Locate two pulleys (8 mm inner diameter) on the front 8 mm rod and lock them on the two rods (2.5 cm far from the end). Repeat this operation for the rear rod and add another pulley, 5 cm far from the right end.
- Attach 12 Self-Aligning Flange Bearings (8 mm diameter) to the inner upper parts of the bioprinter.
- Connect the two up-front bioprinter parts and the two up-rear bioprinter parts (direction left/right) using the assembled rods in step 1.4.3 and lock them to the Self-Aligning Flange Bearings (8 mm diameter). Secure the two assembled parts at all junctions with screws (M3x8).
NOTE: When positioning the rear rod, place a closed belt that connects the pulley on the stepper motor with the inner pulley on the rod.
NOTE: Secure the stepper motor tightening the nuts to ensure no movement of the motor and proper tension of the belt. - Connect the two assembled parts in step 1.4.5 using four 8 mm rods. Let the rods pass through the linear ball bearings present in the assembled part 1.3 and lock them to the Self-Aligning Flange Bearings (8 mm diameter). Secure the two assembled parts at all junctions with screws (M3x8).
- Fix an open belt end on the upper part of the X_block_1 using the belt block and repeat this operation for the X_block_2. Run the belt around the rear pulleys on the left, then around the front pulley, and fix the other belt end again to the X_block_1 with another belt block. Repeat this operation for the second belt.
NOTE: The belts must be properly tightened to obtain the correct movement and of the same length to avoid bending or interference in Y movements.
- Place the fixing bracket to the up rear bioprinter part using four screws (M3x10) and four nuts.
- Assemble the Z-axis (Figure 4).
- Screw together the bottom bioprinter case parts (M4x8).
- Attach one Self-Aligning Flange Bearing (8 mm diameter) to the floor of the bottom of the assembled part in step 1.5.2.
- Fix two flanged linear ball bearings (LMH8UU) to the external holes present in the Z_slider and one leadscrew nut in the central hole with screws (M3x8).
- Fix a limit switch to the Z_plane left side with two screws (M3x8).
- Fasten the stepper motor to a rigid shaft coupling. Attach a stepper motor to the Z_block with four screws (M3x20) and a threaded rod to the rigid shaft coupling. Ensure every block is properly secured with the set screws.
- Connect the assembled part in step 1.5.5 to the bioprinter base using two 8 mm rods. Let them pass through the assembled part in step 1.5.3.
- Fix the Z_block to the back panel using two screws (M5x8).
- Connect Z_plane to Z_slider using four screws (M3x25). Insert them into the Z_plane holes from top to bottom, pass the spring between Z_plane and Z_slider, and secure them with nuts. Adjust spring tension to level the plane.
- Attach four adjustable feet under the bioprinter structure in the specific cavity. Level the bioprinter by adjusting the feet.
- Connect the assembled part to the assembled part in step 1.4.6 with eight screws (M4x12).
- Attach two hinges on the front of the structure in the specific locations and one magnet on the right part in the relative spot with screws (M2x8). Screw the overall structure to 2 mm transparent plexiglass sheets to close the openings.
NOTE: Screw the front plexiglass panel to the hinges and put another screw in correspondence with the magnet to obtain a front door.
- Display the assembly (Figure 5A).
- Let the wires, connected to the display, pass through the bottom hole and attach the display to the display housing with four screws (M3x3).
NOTE: This passage needs to be done without the LCD knob cover. It could be located on the preinstalled knob after the attachment to the display housing. - Close the housing with the cover using four screws (M2x6).
- Insert the SD card into the left slot.
- Let the wires pass through the hole on the top of the structure. Screw the assembled part at the top of the bioprinter with two screws (M3x8).
NOTE: All wires (motors, limit switches, and display) will run close to the wall and exit from the bottom left hole. They are fixed with cable clips to avoid interference with the mechanical parts.
- Let the wires, connected to the display, pass through the bottom hole and attach the display to the display housing with four screws (M3x3).
- Electronics assembly (Figure 5B)
- Fix the socket to the outer part of the electronic case with two screws (M3x10).
- Attach the motherboard to the inner part of the electronic case with four screws (M3x10).
- Attach the bottom part of the power supply to the inner part of the electronic case with four screws (M3x8).
- Pass the wires through the circular hole on the cover and connect them to the motherboard. Close the electronic case with its cover using four screws (M3x10).
NOTE: It is possible to connect the electronics case to the bioprinter structure or to leave it on the desk in case of small working spaces.
Figure 6 shows all electronic cable connections. They can be slightly different according to the 3D printer motherboard in use.
2. Software setting and gcode generation process
- Preparation of slicing software settings
- Open the slicing software.
NOTE: Instructions may differ depending on the software used or the version. - Sign in to the platform.
- Go to the Settings options. Select the printer and then Add a non-networked printer. Select custom FFF printer and in the box Printer name, type the name for the printer. Click on Add.
NOTE: If it is the first software installation, the Add printer panel will automatically open after signing in. - In the Machine Settings window, change the default settings x, y, z to 176, 120, 45 mm.
- Deselect the Heated bed option if it is not already deselected.
- Insert into the Starting g-code and Ending g-code panels the text lines reported in the files titled 'JoVE_Starting_G-code.txt' and 'JoVE_Ending_G-code.txt' in Supplemental File 2.
- Modify the value 150 in line 'M92 E150' in the starting g-code file according to the flow rate to be applied. Estimate the proper value using the following formula:
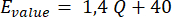
Where Q is the desired flow rate [µL/min].
NOTE: This formula was experimentally obtained with the described system. It might be different if there are changes to the mechanical or electrical components. A possible alternative could be to fix the value according to the system in use and create a custom MATLAB code to generate the desired gcode file.
- Modify the value 150 in line 'M92 E150' in the starting g-code file according to the flow rate to be applied. Estimate the proper value using the following formula:
- Change the Compatible material diameter value in the Extruder 1 panel to 1,75 and then, close.
- Go to Marketplace, select the Z Offset Setting plugin and install it. At the end of the process, restart the software to finalize. Once it reopens, select Settings | Printer | Manage Printers on the toolbar. In the Preferences window, click on Settings and activate all options by selecting Check all option and close it.
- Configure the setup by clicking on the Print Settings (third button) in the stage menu, which contains the configuration panel. Enter the infill percentage in the current print profile in the configuration panel. Select the Custom button to modify the current print settings and the profile Fine in the dropdown menu. Set Print Speed to 10 mm/s.
- Open the slicing software.
- gcode preparation
- Download the *.STL design file, titled 'JoVE_Square', from Supplemental File 2 of this paper.
- Load .STL file into the slicing software.
- Locate the piece on the flat surface and move to coordinates 65, 42.5, 0 (X, Y, Z) mm. Create two copies by clicking on the .STL file name in the object list and selecting the multiply selected option. Move them to the following coordinates: 16, 42.5, 0 mm and -33, 42.5, 0 mm (X, Y, Z).
NOTE: It is not possible to automatically define the printing order unless manually modified directly in the final gcode file. Coordinates are defined for a 6-multiwell plate. - Go to the Print Settings in the stage menu. In the Walls section, put 0 as Wall line count. Put 0 as Top/Bottom thickness value in the Top/Bottom section. In the Infill panel, insert the following values: Infill Pattern: Grid, Infill line distance: 5 mm, Infill line directions: [0].
NOTE: It is possible to reduce Travel Speed in the Print speed section to improve the X axis accuracy. - Disable retraction in the Travel section and print cooling in the Cooling section.
- In the Build Plate Adhesion section, select None as the Build Plate Adhesion Type, while in the Special Modes section, select One at a Time option as the Print Sequence.
- Click on the Slice button to generate the g-code.
- Open the g-code file with Notepad application and remove lines "M104 S200", "M105", and "M109 S200" in line 12 after the phrase ";Generated with Cura_SteamEngine 4.9.0".
- Add "G1 Z25" before the following line ";MESH: JoVE_Square_rev.STL(6)" and split the next one into two: (1) G0 F3600 X142.46 Y7.499 and (2) G1 Z0.3.
NOTE: X and Y coordinates are just examples. - Modify the Z value to Z25 in every line present after ";TIME_ELAPSED" comment every last layer instruction of each piece.
NOTE: For printing just a single piece, step 2.3.10 is not required.
3. Swelling/degradation test
- Preparatory steps
- Take a sterile 1 mL glass syringe (TLL terminal) and a sterile 25 G conical nozzle. Assemble them and put the assembled syringe in the refrigerator.
- Put pipette tips and the SBMe matrix vial in the refrigerator overnight the day before the experiment.
- Print three PLA grids (Φ = 20 mm) with 1 mm x 1 mm circular pore size and a side vertical pillar to grasp them.
- Print constructs
- Take the assembled syringe and the SBMe matrix vial from the refrigerator. Aspirate 500 µL of the SBMe matrix and put it in the incubator for 15 min to allow gelation.
- Take the syringe from the incubator and place it in the bioprinter printhead. Ensure the syringe plunger is properly in contact with the pusher block.
- Secure the syringe fixing plunger and syringe barrel flange with the syringe piston retainers, and syringe barrel with syringe clamp.
- Calibrate the system using the Auto-home setting in the bioprinter menu to automatically detect the origin of axes.
- Move the extruder using the command Move Axes until the needle tip touches the PLA grid to manually define Z0.
- Start printing by selecting the g-code file to print in the bioprinter menu.
- Put the multiwell plate in the incubator for 2 min to allow the SBMe to settle down after extrusion.
- Take the multiwell plate and put 2 mL of cell culture medium into the three wells containing the bioprinted grids. Put the multiwell plate in the incubator.
- At each time-point (t0, 1 h, 2 h, 3 h, 4 h, 1, 3, 7, 10, and 14 days), take each grid from the plate and dry it with clean paper to remove the excess of medium and weight on a balance. Put the grid back into the plate and then in the incubator to continue the experiment. Estimate the swelling ratio and degradation rate using the following formulas:
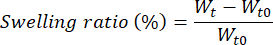
Where Wt is the weight of the constructs at each time point and Wt0 the weight of the dried constructs.
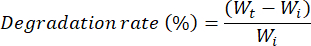
Where Wi is the weight at the end of the swelling test.
4. Bioprinting of SBMe constructs
- Preparatory steps
- Take a sterile 1 mL glass syringe (TLL terminal) and a sterile 25 G conical nozzle and assemble them under the biosafety cabinet. Put them into a sterile box in the refrigerator.
- Put the pipette tips and the SBMe matrix vial in the refrigerator overnight (4 °C on-ice) the day before the experiment.
- Add 20 µL of trypan blue to a 1.5 mL centrifuge tube.
- Set the centrifuge at 345 × g, 4 °C and let it to cool down (~3-4 h).
- Take 5 mL of water into a 15 mL centrifuge tube as a balance for the centrifuge.
- Warm Phosphate Buffer Solution (PBS) and Media in a water bath.
NOTE: Cell culture medium is made of complete Dulbecco modified Eagle's medium (DMEM) supplemented with 10% Fetal Bovine Serum (FBS), Penicillin-Streptomycin, and L-glutamine. - Place the bioprinter under the biological safety cabinet. Sterilize under UV light for 20 min.
NOTE: To ensure the proper functioning of the bioprinter, execute the Auto-home function and verify the proper movements of each axis and working of limit switches.
- Bioink preparation
- Take the flask in the incubator containing commercial murine prostate cancer cells and put it under the biosafety cabinet.
- Remove the cell culture medium from the flask.
NOTE: Volumes reported in this protocol apply to T25 Cell Culture flasks. - Wash cells once with 2 mL of PBS and then aspirate it.
- Add 500 µL of trypsin and put the flask in the incubator for 5 min to allow cell detachment from the plate.
- Check cells for trypsinization and if necessary, gently tap the flask bottom.
- Add 4.5 mL of medium to the flask. Carefully wash the plate to ensure all the detached cells are floating in the cell culture medium.
- Aspirate the medium and add it to a 15 mL centrifuge tube.
- Take a 20 µL aliquot and mix it with the trypan blue aliquot. Then, put 10 µL of this mixture into the cell counting chamber to count the live cells using the following formula:
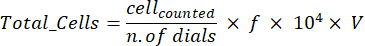
Where f is the dilution factor for trypan blue suspension (1:1 ratio); V is cell suspension volume [mL]. - Centrifuge the remaining cell suspension for 5 min (345 × g, 4 °C).
- Remove the supernatant and resuspend into the cold SBMe matrix at the desired cell density (106 cells/mL)
- Aspirate 800 µL of the matrix suspension containing cells with the cold syringe.
- Remove the air bubbles that might be trapped in the syringe and put it in a sterile box. Then, place it in the incubator for 15 min to allow gelation.
- Bioprinting process
- Take the syringe from the incubator and put it in the bioprinter printhead. Ensure the syringe plunger is properly in contact with the pusher block.
NOTE: The model and the volume must be the same as the syringe used for the experiment. - Secure the syringe fixing plunger and syringe barrel flange with the syringe piston retainers, and syringe barrel with syringe clamp.
- Use M4 nuts at the bottom part of the extruder to adjust the spring pressure and lock the syringe in place.
NOTE: Steps 4.3.1 to 4.3.4 are shown in Figure 7. - Calibrate the system using the Auto-home setting in the bioprinter menu to automatically detect the origin of axes.
NOTE: This passage always ensures the proper initial settings independently from the type of support we use for printing. - Place a 6-multiwell plate on the printing plane.
- Move the extruder using the command Move Axes until the needle tip touches the bottom of the well to manually define Z0.
- Select the gcode file in the menu option of the bioprinter menu and start printing. A square six-layer grid (2.6 cm side) will be printed in each well.
- After the bioprinting process, use an inverted microscope to ensure that the printing process was successful and put the multiwell plate in the incubator for 2 min to allow the SBMe to settle down after extrusion.
- Put 2 mL of cell culture medium into each well.
NOTE: The volume must fully cover the constructs.
- Take the syringe from the incubator and put it in the bioprinter printhead. Ensure the syringe plunger is properly in contact with the pusher block.
Results
In this article, we present a protocol for bioprinting constructs made of SBMe using a low-cost, custom-made, volumetric bioprinter. We provide a detailed description and 3D printing *.STL files to build the system and we demonstrate its potential use in cancer research by printing single and multi-layer structures. Each 3D printed component has been properly cleaned from support structure or unwanted printing residues before assembling. Figure 8 shows different views of the overall assembly...
Discussion
In translational research, SBMes are particularly valuable in a variety of research fields, from tissue engineering to drug testing2,3. In particular, they have become fundamental in cancer research, as they replicate mechanical and biochemical features of the tumor microenvironment, enabling the development of models that better reflect tumor growth, metastasis, and treatment responses. SBMe is the most widely used biological matrix, especially for 3D organoids<...
Disclosures
The authors have no conflicts of interest to declare.
Acknowledgements
This work was partially funded by the Accelerator Award n° A26815 entitled: "Single-cell cancer evolution in the clinic" funded through a partnership between Cancer Research UK and Fondazione AIRC (n° 22790) and by the European Union - Next Generation EU, Mission 4, Component 1, CUP D53D23003310006.
Materials
| Name | Company | Catalog Number | Comments |
| 12-multiwell plate | Sigma-Aldrich | CLS351143 | |
| 3D FDM printer | Flashforge | Adventurer 5M Pro | |
| 3D printer Motherboard | GEETECH | GT2560 REV A+ | |
| 6-multiwell plate | Sigma-Aldrich | M8687 | |
| AC socket | HiLetgo | https://www.amazon.it/HiLetgo-Terminal-Socket -Holder-Switch/dp/B0814PT3YG/ref=sr_1_1?__ mk_it_IT=%C3%85M%C3%85%C5%BD%C3% 95%C3%91&crid=2TRIBP34VJ61A &dib=eyJ2IjoiMSJ9.zW6_VmeiudR I4yMpjJqJm7OdeoctTKW2mrEzml BAyJVa1hkM4PiJT3pLY0mXdxm1 AZQxa2_f5J4q4d5v3vUeqLkjJ6BM kWHAoeEfbEQJNuZaQ3NjmUlqWy _1AfQpnRp4VqJ2m3bkWsChztCa Ok-ZdQJlobGxJaMKg7WbV352 Hyo.0tEw2GYoPQJ4SdNYXO1XM Jrl2yXMvRey55Hj7GhISkk&dib_ tag=se&keywords=AC+socket+HiLetgo&nsdOpt OutParam=true&qid=1734702097&sprefix=ac+ socket+hiletgo%2Caps%2C173&sr=8-1 | 15 A 250 V |
| Adjustable feet | EXIN DEHCEN | https://www.amazon.it/gp/product/ B0CPLTSNQB/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | Height 27-40 mm |
| Ball bearing 4 mm | QUARKZMAN | 684ZZ | Dimensions 4 x 9 x 4 mm |
| Ball bearing 5 mm | XiKe | 625-2RS | Dimensions 5 x 16 x 5 mm |
| Belt closed | Turmberg3D | https://www.amazon.it/gp/product/ B09B2FJQBV/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | Width 6 mm |
| Belt open | Fajoeda | https://www.amazon.it/gp/product/ B09QCTVPTH/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&th=1 | Width 6 mm |
| Bioprinting nozzle | Fisher Scientific | 17780789 | Corning Standard Conical Bioprinting Nozzles |
| Bürker Cell Counting chamber | GLASWARENFABRIK KARL HECHT | 40443001 | |
| Centrifuge | Eppendorf | 5702 R | |
| Centrifuge tube | Sigma-Aldrich | CLS431470 | 15 mL |
| Centrifuge tube | Sigma-Aldrich | EP0030120086 | 1.5 mL |
| Display | Sigma-Aldrich | BR718905 | |
| DMEM | Gibco Thermofisher | 11965092 | |
| FBS | Sigma-Aldrich | F7524 | |
| Flanged linear ball bearing | Sourcing map | https://www.amazon.it/gp/product/ B07S6LZHM7/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | LMF8UU |
| Glass syringe | Fisher Scientific | 11520062 | Hamilton GasTight 1 mL (TLL terminal) |
| Hinges | YASQZ | https://www.amazon.it/gp/product/ B09DNXGDGG/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | |
| Incubator | Fisher Scientific | 15481374 | Eppendorf Galaxy 48 R CO2 Incubator, 48 L, Stainless Steel |
| Inverted microscope | Nikon | Eclipse Ti2-U | |
| Jumper wires | Iverntech | https://www.amazon.it/Iverntech-XH2 -54-Terminale-Stampante-Stepper/dp/ B07Q12B6K5/ref=pd_ci_mcx_mh_ mcx_views_0_image?pd_rd_w=cb2r J&content-id=amzn1.sym.57a351cf- ba07-47e8-b302-aa2aa1a3f209%3 Aamzn1.symc.ca948091-a64d-450e- 86d7-c161ca33337b&pf_rd_p=57a3 51cf-ba07-47e8-b302-aa2aa1a3f209 &pf_rd_r=YHY9RFYZJK7PYAY2RXX D&pd_rd_wg=7WkXB&pd_rd_r=8239 66cb-4977-48ce-b095-0dcedf8c372f& pd_rd_i=B07Q12B6K5&th=1 | |
| LCD unit | Paradisetronic.com | https://www.amazon.it/Display-controller -conduttori-adattatore-stampante/dp/B01 DUR4064/ref=sr_1_9?__mk_it_IT=%C3 %85M%C3%85%C5%BD%C3%95%C3 %91&crid=1OAHVIIRA16X3&dib=eyJ2Ij oiMSJ9.07V2Jf7RzJjTUlIuFobUJFjUzG4 8633KkdcMXSEfnWLRX41d5vAF7xPBJ maniVxDoXecfihZzzOXlP3v-e29cUjoGLf GDyv5DoDVwp7ndohNTYvYzoi3gWkF- Hsd2YbT7tjb1Dra1afqSn26CbFdEvJ84y qyX7Gi3-7DHh8yMwdd6_7quiuW_mjsm w1nu0YQG68GZ0XM7unOsvyddQ__R_ 3dndxb3mOjgGjklEgu_KEbZJPOZDIdSo oU3nrrKHj56hCfk9ACwYpB80QGCbTPK 0p-fE0g9h02WPmtHV38UU3m5AdZsTQ HqvnVlJg703qGT-8ze8fQItw2rtbgl_J86H RMReJOhVLQBkqNwxh57CQ.yNbeFmZ ZBEUhadz2dQa8NxpZD9g4P6ighRU_97 yxgnw&dib_tag=se&keywords=display% 2B3D%2Bprinter%2Bprusa%2Bi3&nsdO ptOutParam=true&qid=1734714093&s= industrial&sprefix=display%2B3d%2B printer%2Bprusa%2Bi3%2Cindustrial% 2C143&sr=1-9&th=1 | Old Prusa i3 LCD unit. Link to commercially available alternative |
| Leadscrew M8 | RS PRO | 280-408 | |
| Leadscrew nut M8 | Comioke | https://www.amazon.it/dp/B0C7QB13C4? ref=ppx_yo2ov_dt_b_fed_asin_title&th=1 | Nut for M8 screws |
| Leadscrew nut T8 | Aipaide | https://www.amazon.it/gp/product/B086QJCX1M/ref =ppx_yo_dt_b_search_asin_title?ie=UTF8&psc=1 | Nut for T8 screws |
| Leadscrew T8 | VBESTLIFE | https://www.amazon.it/gp/product/B07CXRB52Y/ref =ppx_yo_dt_b_search_asin_title?ie=UTF8&th=1 | Length 15 mm |
| L-Glutammine | Sigma-Aldrich | G7513 | |
| Limit switch | CESFONJER | https://www.amazon.it/gp/product/B07SPX492J/ref =ppx_yo_dt_b_search_asin_title?ie=UTF8&psc=1 | |
| Linear ball bearing (LM4UU) | A ABSOPRO | https://www.amazon.it/gp/product/B0CB3M5GHJ/ref =ppx_yo_dt_b_search_asin_title?ie=UTF8&psc=1 | Dimensions 4 x 8 x 12 mm |
| Linear ball bearing (LM8LUU) | Fdit | https://www.amazon.it/LM8LUU-Linear -Motion-cuscinetti-stampante/dp/B07N 58W3GZ/ref=sr_1_1_sspa?__mk_it_IT =%C3%85M%C3%85%C5%BD%C3% 95%C3%91&crid=33VNCCCSIFJRP& dib=eyJ2IjoiMSJ9._JJ2doXI33C2JagX 8x6E2Bz2ILo10852tBi1erWqGn363ku zbcvknbMtsK6wm5jdR1P5XwC6Ovui at3kGIJPpYFOG_FiIBYT8xLkkbiphhR 4ncR6R9tPPyeN--_3RxFgKMh5tMO_ jesAcu7Anp87Qb91ruM85CKqduV-y6 HQJtAvVSXmfG4yE4N7d4_dDVKsR7 UkEmDIznJE0HIhvvI2R6vX2tMN5yMc _ZrUbUB0gPF6SgXHVJmKRizCRqL7J 64WRD-XnMSpVHY_LUVJ7gn9kJQ35 _zQw8vLD-_kRyvxwvE.4IVMdKzF1Ax mQHQhWEi6LH12UI4CmMgi288_g3zG CPY&dib_tag=se&keywords=LM8LUU+ Fdit&nsdOptOutParam=true&qid=17347 04494&s=industrial&sprefix=lm8luu+fdit+ %2Cindustrial%2C136&sr=1-1-spons&sp _csd=d2lkZ2V0TmFtZT1zcF9hdGY&psc=1 | Dimensions 8 x 15 x 45 mm |
| Linear ball bearing (LM8UU) | ARCELI | https://www.amazon.it/gp/product/ B07BV3YBP2/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | Dimensions 8 x 15 x 24 mm |
| Magnets | OCEUMAOA | https://www.amazon.it/gp/product/ B0CB6C2RF4/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&th=1 | 8 x 3 mm |
| Matrigel | Gibco Thermofisher | LOT 0202003 | SBMe |
| Motor driver | Longruner | https://www.amazon.it/gp/product/ B071P41ZBW/ref=ppx_yo_dt_b_search _asin_title?ie=UTF8&psc=1 | Driver A4988 |
| PBS | MicroGem, VWR | TL1006-500ML | |
| Penicillin-Streptomycin | Gibco Thermofisher | 15070063 | |
| Pipette tips | Sigma-Aldrich | Z740058 | BRAND filter tips, racked, TipBox (volume 50-1000 μL) |
| Plexiglass sheets | MVQPER | https://www.amazon.it/MVQPER-trasparenti- plexiglass-trasparente-decorazione/dp/B0CF Q4BHQZ/ref=sr_1_14?dib=eyJ2IjoiMSJ9.bf7 Mp7I5m2qAR1Fp1V6DoYF2erUlNQUK_NuF ZLz9A7uJKGa_RNeni6NiO4YQfAjVHBtZgdzI 356XVc3BIuKVpV8Pfg0rImqNXgxG-kfklAYrb rXv4tV6RL1ShFwgLzfQh8R0sEoFJPcLF2f2E EcGMlvEwOuCv6qRnS1MQ-VaA0sLPnM0ts wdL4sLVxsonWcfSrHAUrgWh4fpVNMA4Kwh 0UaeQhfexFY0dNWhPYftXvvz9LLj1uMID2E Gi_ScmYqY5LahbYOmKVVPPiPIgCVdLjaM8 yqNp2ZJGV9Fge2xa6OAHxmMeaQUqGkoQ 88x9GkX7EWuOwYO0dEUYSzHCdGONWo eZLfSJOR3C6-knjzKrOQ0ziouJfo-ktbak7v3A H53BtIOiXbT57cKqh-uc9wI_j_lHN-8okAe2yO 6upc61MoydZujuxnfLG7hwytqHQG2.ApKq8 MVs8Xihn_zcA39nML-2lpjnagGhXIx0clcJN_ E&dib_tag=se&keywords=lastra+plexiglass+ 2+mm&nsdOptOutParam=true&qid=1734702823&sr=8-14 | 2 mm thickness, transparent sheet, 21 x 32.5 cm |
| Plexiglass sheets | MVQPER | https://www.amazon.it/MVQPER-trasparenti -plexiglass-trasparente-decorazione/dp/B0C FQ4BHQZ/ref=sr_1_14?dib=eyJ2IjoiMSJ9.b f7Mp7I5m2qAR1Fp1V6DoYF2erUlNQUK_N uFZLz9A7uJKGa_RNeni6NiO4YQfAjVHBtZ gdzI356XVc3BIuKVpV8Pfg0rImqNXgxG-kfk lAYrbrXv4tV6RL1ShFwgLzfQh8R0sEoFJPc LF2f2EEcGMlvEwOuCv6qRnS1MQ-VaA0s LPnM0tswdL4sLVxsonWcfSrHAUrgWh4fpV NMA4Kwh0UaeQhfexFY0dNWhPYftXvvz9 LLj1uMID2EGi_ScmYqY5LahbYOmKVVPP iPIgCVdLjaM8yqNp2ZJGV9Fge2xa6OAHx mMeaQUqGkoQ88x9GkX7EWuOwYO0dE UYSzHCdGONWoeZLfSJOR3C6-knjzKrO Q0ziouJfo-ktbak7v3AH53BtIOiXbT57cKqh- uc9wI_j_lHN-8okAe2yO6upc61MoydZujux nfLG7hwytqHQG2.ApKq8MVs8Xihn_zcA3 9nML-2lpjnagGhXIx0clcJN_E&dib_tag=se &keywords=lastra+plexiglass+2+mm&nsd OptOutParam=true&qid=1734702823&sr=8-15 | 2 transparent sheets, 2 mm thickness, 20 x 20 cm |
| Plexiglass sheets | MVQPER | https://www.amazon.it/MVQPER-trasparenti -plexiglass-trasparente-decorazione/dp/B0C FQ4BHQZ/ref=sr_1_14?dib=eyJ2IjoiMSJ9. bf7Mp7I5m2qAR1Fp1V6DoYF2erUlNQUK_ NuFZLz9A7uJKGa_RNeni6NiO4YQfAjVHB tZgdzI356XVc3BIuKVpV8Pfg0rImqNXgxG- kfklAYrbrXv4tV6RL1ShFwgLzfQh8R0sEoFJ PcLF2f2EEcGMlvEwOuCv6qRnS1MQ-VaA 0sLPnM0tswdL4sLVxsonWcfSrHAUrgWh 4fpVNMA4Kwh0UaeQhfexFY0dNWhPYft Xvvz9LLj1uMID2EGi_ScmYqY5LahbYO mKVVPPiPIgCVdLjaM8yqNp2ZJGV9Fge 2xa6OAHxmMeaQUqGkoQ88x9GkX7E WuOwYO0dEUYSzHCdGONWoeZLfSJO R3C6-knjzKrOQ0ziouJfo-ktbak7v3AH53B tIOiXbT57cKqh-uc9wI_j_lHN-8okAe2yO6 upc61MoydZujuxnfLG7hwytqHQG2.ApKq 8MVs8Xihn_zcA39nML-2lpjnagGhXIx0clcJ N_E&dib_tag=se&keywords=lastra+plexigl ass+2+mm&nsdOptOutParam=true&qid=1 734702823&sr=8-16 | 2 transparent sheets, 2 mm thickness, 20 x 30 cm |
| Polylactic acid (PLA) filament | Flashforge | Black 100102 | |
| Power supply | SQUADO | https://www.amazon.it/SQUADO-Alimentatore -stabilizzato-dissipatore-efficienza /dp/B0DMTG6SJD/ref=sr_1_6?dib =eyJ2IjoiMSJ9.1vVze6Zt6fhKCvFb eAOSiRe57N6a1mlBnkLdHiEym4 ugHzueF6_FdOodb3sftCXb5raTg eerMMUbL5KcaI_SDa_2j7s2XohI B01QMJDyq0xQ5Gp3KLIZozDHV fGBdSrvwgyOl-BXSDy_ocZlj2TL5 e21p3K6qpvSaT5yo8WA5tngS93 Eqe8s8D6mjpnnPFrb9RpQpj299Y nNRYgr_YF3NCt3mH0r0FocKcCa LNpKtfGLCbnz_yB-360tp5SAi7A7 cgBf5I4NzQ5iQTTXAG727x0Hml mG-eaFBSJv6M1LPooZiT2aT7dx o_7tzyU6MQ9vAEozXkLTaTTMX RA_svPp96sxp93NRLReI64xpFd S3Zh1y_JPQijbMAIuvuYflhh6JlPB W6-fpW6QXl9v5wNEs_YwjzP9Sf RKxRmchzZhgsK-34XJeaEmjbRq H3piM0Zx.MfPYUNvCEhDBqLDh C-iqBEu-vjIVgsrQmOjeLuGo9YQ& dib_tag=se&keywords=power+sup ply+AC+INPUT%3A+110-220V%2 C+DC+OUTPUT%3A+12V+15A&n sdOptOutParam=true&qid=173471 4722&sr=8-6 | Old Prusa i3 power supply (AC INPUT: 110-220 V, DC OUTPUT: 12 V 15 A). Link to commercially available alternative |
| Pulley 5 mm | VooGenzek | https://www.amazon.it/gp/product/ B0B8H16KWX/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | Bore 5 mm, Width belt space 6 mm |
| Pulley 8 mm | Yxtaii | https://www.amazon.it/gp/product/ B07X852RFN/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&th=1 | Bore 8 mm, Width belt space 6 mm |
| Ribbon cables | QUARKZMAN | https://www.amazon.it/sspa/click?ie =UTF8&spc=MTo0NDgyNTkwNzU wNjM5NzM5OjE3MzQ3MTQ1OTE6 c3BfbXRmOjMwMDEyMjk4NTMzN TYzMjo6MDo6&url=%2FQUARKZM AN-connettore-Lunghezza-Comput er-confezione%2Fdp%2FB0CQRM DQNG%2Fref%3Dsr_1_15_sspa% 3F__mk_it_IT%3D%25C3%2585M %25C3%2585%25C5%25BD%25 C3%2595%25C3%2591%26crid%3 D3BZ8Q05YA50TX%26dib%3Dey J2IjoiMSJ9.zoYnL2ViiarlDS_7165D ldxu-TTbVbWUVAP3pCybFB-bjGl wSgrpnkMhhsJj3NRo8Mvht8ev5H vGQ-UUpPom12OcOcwyRVXLAR UvMyTm8mpAdZSaOritwHRgwu2 JuW9UoS4jlCbA-gzP2FEAAH47K fFOWN6FEhc5r0YGBcSIsA6_QV6 _LA1k_ifj3c2BFm2Fj8YjW-auz7Yiy ULfZusvGX28S1K5kJdTa_YOwJY YC0Y__s-CCchHKiZgDhFjtFogUz0 Tonob4HQZiO_hkrnm3ixpNj9rgE- hvVq1FK1uD9M.kkP89x1SJ0SLxZ sv3YomYEfmkhpvP4WQf2TtC76K YAE%26dib_tag%3Dse%26keywords %3DCavo%2Bflessibile%2Ba%2Bna stro%2Bpiatto%2Bper%2Bstampant e%2B3D%2Ba%2B10%2Bpin%26n sdOptOutParam%3Dtrue%26qid% 3D1734714591%26s%3Dindustrial %26sprefix%3Dcavo%2Bflessibile %2Ba%2Bnastro%2Bpiatto%2Bper %2Bstampante%2B3d%2Ba%2B10 %2Bpin%252Cindustrial%252C170 %26sr%3D1-15-spons%26sp_csd% 3Dd2lkZ2V0TmFtZT1zcF9tdGY%26psc%3D1 | ribbon cable 2651 28AWG 10 pins |
| Rigid shaft coupling | Gwolf | https://www.amazon.it/gp/product/ B088R8CW5Z/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | Coupling from 5 to 8 mm |
| Rods 4 mm | RS-components | 682-810 | Diameter 4 mm |
| Rods 5 mm | Sourcingmap | a14010200ux0214 | Diameter 5 mm |
| Rods 8 mm | RS-components | 682-078 | Diameter 8 mm |
| Screws | Bnnrjia | 281-984 | M2x6 - Flat head |
| Screws | INCREWAY | https://www.amazon.it/INCREWAY- assortite-portatili-accessori- riparazione/dp/B07KN37M5J/ ref=sr_1_20?__mk_it_IT=%C 3%85M%C3%85%C5%BD% C3%95%C3%91&crid=12BH MAWD8SK4Z&dib=eyJ2IjoiM SJ9.SjXKoxV0NYqENPoUeS4 2-rj1Af398uiCLXDxFs0C9HfS 8vwEPmsfAMPwIMfllkAfWNY TIEU4KXhJRgRHInNI3U_aow 0EPyfufQKlA4wHz1eOJBvYO ND-5obbXfRPn7VXQ8kzBXM VfUHLAIFlJhTO8AXclkwgjB-z pRvQ6oE5Fr_1sO7lFnzURtD1 YBf65O58M3hrDXf5r_8MIz1c Asct-2P8AyWWBZAB1buYoyP gnnOiBL5EBS7twJWqcDU86m h1ywyufiUitm6kIwNuMx32D0h 0jkHna481aaYP_Q7kHgDxxrR _fFXj5gV2XqEesqGkxPCmHfF WVMqvB_YBdEAuTeL08EP_p TY50uT4UeWgdJuT58kB7Gv- Y5fPMPRS_JEQdO-oJ9DlO9n 0OszkmYwrPh-AivwVN6xh4hN 2pseXannS_on9aiNv1lh3x-f2PZ 0R.jCyNdxQiJSAsGVm7ZPhDK eFxQGBAu-M_42ZLTSSBiDg&d ib_tag=se&keywords=viti+M3x3& nsdOptOutParam=true&qid=1734 711519&sprefix=viti+m3x3%2Cap s%2C169&sr=8-20 | M2x8 - Flat head |
| Screws | INCREWAY | https://www.amazon.it/INCREWAY -assortite-portatili-accessori-ripara zione/dp/B07KN37M5J/ref=sr_1_2 0?__mk_it_IT=%C3%85M%C3%85 %C5%BD%C3%95%C3%91&crid= 12BHMAWD8SK4Z&dib=eyJ2IjoiM SJ9.SjXKoxV0NYqENPoUeS42-rj1 Af398uiCLXDxFs0C9HfS8vwEPms fAMPwIMfllkAfWNYTIEU4KXhJRg RHInNI3U_aow0EPyfufQKlA4wHz 1eOJBvYOND-5obbXfRPn7VXQ8 kzBXMVfUHLAIFlJhTO8AXclkwgjB -zpRvQ6oE5Fr_1sO7lFnzURtD1YB f65O58M3hrDXf5r_8MIz1cAsct-2P8 AyWWBZAB1buYoyPgnnOiBL5EBS 7twJWqcDU86mh1ywyufiUitm6kIwN uMx32D0h0jkHna481aaYP_Q7kHg DxxrR_fFXj5gV2XqEesqGkxPCmHf FWVMqvB_YBdEAuTeL08EP_pTY50 uT4UeWgdJuT58kB7Gv-Y5fPMPRS _JEQdO-oJ9DlO9n0OszkmYwrPh- AivwVN6xh4hN2pseXannS_on9aiN v1lh3x-f2PZ0R.jCyNdxQiJSAsGVm 7ZPhDKeFxQGBAu-M_42ZLTSSB iDg&dib_tag=se&keywords=viti+M3 x3&nsdOptOutParam=true&qid=17 34711519&sprefix=viti+m3x3%2Ca ps%2C169&sr=8-20 | M2x10 - Flat head |
| Screws | INCREWAY | https://www.amazon.it/INCREWAY- assortite-portatili-accessori-riparazi one/dp/B07KN37M5J/ref=sr_1_20? __mk_it_IT=%C3%85M%C3%85% C5%BD%C3%95%C3%91&crid=12 BHMAWD8SK4Z&dib=eyJ2IjoiMSJ 9.SjXKoxV0NYqENPoUeS42-rj1Af3 98uiCLXDxFs0C9HfS8vwEPmsfAM PwIMfllkAfWNYTIEU4KXhJRgRHIn NI3U_aow0EPyfufQKlA4wHz1eOJB vYOND-5obbXfRPn7VXQ8kzBXMV fUHLAIFlJhTO8AXclkwgjB-zpRvQ6 oE5Fr_1sO7lFnzURtD1YBf65O58 M3hrDXf5r_8MIz1cAsct-2P8AyWW BZAB1buYoyPgnnOiBL5EBS7twJ WqcDU86mh1ywyufiUitm6kIwNuM x32D0h0jkHna481aaYP_Q7kHgDx xrR_fFXj5gV2XqEesqGkxPCmHfF WVMqvB_YBdEAuTeL08EP_pTY5 0uT4UeWgdJuT58kB7Gv-Y5fPMPRS _JEQdO-oJ9DlO9n0OszkmYwrPh-Ai vwVN6xh4hN2pseXannS_on9aiNv1l h3x-f2PZ0R.jCyNdxQiJSAsGVm7Z PhDKeFxQGBAu-M_42ZLTSSBiDg &dib_tag=se&keywords=viti+M3x3&n sdOptOutParam=true&qid=1734711 519&sprefix=viti+m3x3%2Caps%2 C169&sr=8-20 | M3x3 - Flat head |
| Screws | RS-components | 281-978 | M3x6 - Flat head |
| Screws | RS-components | 281-978 | M3x10 - Flat head |
| Screws | RS-components | 281-978 | M3x20 - Flat head |
| Screws | Faankiton | https://www.amazon.it/Faankiton-600- M3-Incassato-Cilindrica/dp/B0CFY6V 3HV/ref=sr_1_3_sspa?crid=29NMQZ B1QM7EW&dib=eyJ2IjoiMSJ9.aiGS- zo819G6hZmghT7TFDH1TL5lzRsjT R5PBvqoHbKOBLipyX-kuevmqqPpW m_7Ngng-Wa6aLoDuXMtoqexgeNpr Qn2cISsCatqzhc8fxhJ0yIN4s3YEpD KzIroWWVNzswptkWthnGkLXwHck m4wqLr1sf0qyK9oYYlZdKo_Ypa9RQ s3aYPNd_f-DWpT5rdsG1GL3rd3b9c fLSqXxMd8QnnK3hDcg10Uz9otPwo zJC2QAQFce__CkNwDfBp5Vvgmy1 DLX4a7PolnNePrJiObgjHMJLlfwizGx fie28RAYQ.03Epc_RrVWlBUqUbHU TPDcoDUxaKBHep1CuTOhFj-fI&dib _tag=se&keywords=m3x25%2Bviti&n sdOptOutParam=true&qid=17347118 59&s=industrial&sprefix=M3x25%2Ci ndustrial%2C185&sr=1-3-spons&sp_ csd=d2lkZ2V0TmFtZT1zcF9hdGY&th=1 | M3x25 - Flat head |
| Screws | Faankiton | https://www.amazon.it/Faankiton-600 -M3-Incassato-Cilindrica/dp/B0CFY6 V3HV/ref=sr_1_3_sspa?crid=29NMQ ZB1QM7EW&dib=eyJ2IjoiMSJ9.aiG S-zo819G6hZmghT7TFDH1TL5lzRsj TR5PBvqoHbKOBLipyX-kuevmqqPp Wm_7Ngng-Wa6aLoDuXMtoqexgeN prQn2cISsCatqzhc8fxhJ0yIN4s3YEp DKzIroWWVNzswptkWthnGkLXwHc km4wqLr1sf0qyK9oYYlZdKo_Ypa9R Qs3aYPNd_f-DWpT5rdsG1GL3rd3b9 cfLSqXxMd8QnnK3hDcg10Uz9otPwo zJC2QAQFce__CkNwDfBp5Vvgmy1D LX4a7PolnNePrJiObgjHMJLlfwizGxfie 28RAYQ.03Epc_RrVWlBUqUbHUTPD coDUxaKBHep1CuTOhFj-fI&dib_tag=s e&keywords=m3x25%2Bviti&nsdOptO utParam=true&qid=1734711859&s=ind ustrial&sprefix=M3x25%2Cindustrial%2 C185&sr=1-3-spons&sp_csd=d2lkZ2V 0TmFtZT1zcF9hdGY&th=1 | M3x30 - Flat head |
| Screws | AERZETIX | https://www.amazon.it/AERZETIX- carbonio-fissaggio-cacciavite-mont aggio/dp/B0BDYZZ8LQ/ref=sr_1_4 ?__mk_it_IT=%C3%85M%C3%85 %C5%BD%C3%95%C3%91&crid= 217010XNB7ZXP&dib=eyJ2IjoiMSJ 9.e5QjA-q9hEvlleIzwY3ssA3x7vIBF Hpcg73tKkBJu9QbDwMwlC5L9-9u 7my7ZG5hZij0WyaijakqAoX4rIZ3rv EgvlPtN6bwKTBgqAK-3uu3GBy6K EPQ2iIEUWx_U73nonJy5IzmTDH Xijn-kRg1iCOM99S5upSWbmmazg 8lctsyXlg_tmXHjHn63WsIo7WcBuL rtVpPW8oMsZCndwh33pNQXBnk1 iStT81spopzrw3gScK1Wdz6RttH6j YL611rNBdkd-PEP2CYhDKeSKHE AqmABjHGLyG6p40k20npHqkg8g- 9Msl3fZZiS4rRxNFOHBjEdVTLBrn PjRjxTspBfZSEKQnqsGm1WPlz8cS khCs.ujWHAL4y-RNahay-1AJl34uN hUj-f6mZ7X1_KZaACvU&dib_tag=s e&keywords=m3x50%2Bviti&nsdOp tOutParam=true&qid=1734711964& s=industrial&sprefix=m3x50%2Bviti %2Cindustrial%2C136&sr=1-4&th=1 | M3x50 - Flat head |
| Screws | JOCAHULFX | https://www.amazon.it/Incassato- Esagonale-Rondelle-Esagonali- Inossidabile/dp/B0DF7P5CC2/ref=sr_ 1_6?__mk_it_IT=%C3%85M%C3%8 5%C5%BD%C3%95%C3%91&crid=2 7B231M7SRJ3R&dib=eyJ2IjoiMSJ9.3 bij8nHLQAebjY8LHQS6V1GsAE07H cFGUpHAh7jA1oQIxjoTJmxjNuwg0y H14oz-YUvzD0eqVRASqAZTK2spiQ saX0pug7rWgGfENeGhztno5lMcJDT XQfe9H6ocLZANpAFv_p0dRbO6ggA vZN0TfyiuR4focFMAjr6VFGVbprEQO EaS1KRs8loF3FgL0rsmP81K806Gv5 zeSurK_sp8GEelqtm1G3J059zUdPaJ z7zJQEFPSH0TXv3lml5u9lnvnAbM0j ClF03nG5ZjB-6WUPfS41Os5DgzTpS zUTR-YZc1HnRUxy6l5grDPFmDiNZn 0Ie9MrlkcPI2A8dubQZm3d8pYVpg6Y 2OqzifmZzNsQM.Zuh5Ko4bf20Bz46c NFRHJvcyPfJyglpcvN1uM16-yOI&dib _tag=se&keywords=m4+viti+testa+sv asata&nsdOptOutParam=true&qid=17 34712054&s=industrial&sprefix=m4+v iti+testa+svasata%2Cindustrial%2C12 8&sr=1-6 | M4x8 - Flat head |
| Screws | JOCAHULFX | https://www.amazon.it/Incassato-Esagonale -Rondelle-Esagonali-Inossidabile/dp/B0DF 7P5CC2/ref=sr_1_6?__mk_it_IT=%C3%85 M%C3%85%C5%BD%C3%95%C3%91&c rid=27B231M7SRJ3R&dib=eyJ2IjoiMSJ9. 3bij8nHLQAebjY8LHQS6V1GsAE07HcFG UpHAh7jA1oQIxjoTJmxjNuwg0yH14oz-YU vzD0eqVRASqAZTK2spiQsaX0pug7rWgG fENeGhztno5lMcJDTXQfe9H6ocLZANpAF v_p0dRbO6ggAvZN0TfyiuR4focFMAjr6VF GVbprEQOEaS1KRs8loF3FgL0rsmP81K8 06Gv5zeSurK_sp8GEelqtm1G3J059zUdP aJz7zJQEFPSH0TXv3lml5u9lnvnAbM0jCl F03nG5ZjB-6WUPfS41Os5DgzTpSzUTR -YZc1HnRUxy6l5grDPFmDiNZn0Ie9Mrlkc PI2A8dubQZm3d8pYVpg6Y2OqzifmZzNs QM.Zuh5Ko4bf20Bz46cNFRHJvcyPfJyglp cvN1uM16-yOI&dib_tag=se&keywords=m4 +viti+testa+svasata&nsdOptOutParam=tru e&qid=1734712054&s=industrial&sprefix= m4+viti+testa+svasata%2Cindustrial%2C1 28&sr=1-6 | M4x12 - Flat head |
| Screws | Globstar | https://www.amazon.it/testa-svasata- impronta-Phillips-acciaio/dp/B083RH 5JYL/ref=sr_1_9?__mk_it_IT=%C3% 85M%C3%85%C5%BD%C3%95%C 3%91&crid=XEVGIE9R1K2F&dib=ey J2IjoiMSJ9.sYNfljBDPU1N2px8nueT zi6D61Z46HD98MSuIwqXNKrfkSOM GYgywNTC8-dsYqNXiZEMS42kGB4 QdBtz_TVFLvgLCFQVl9W5n7Bo4fQS _VKHh9VLmA_RjdF75wPsSbRPIdSA nidi0pehfkRoBIrEtYVn-cUq4TXH0P1D pGLc_4J88tU02ieLVee-YarMxZLKeLYt kK6B2CmM6d_AE0K7UJofwnOCOLp 3R72KpMTltjkJutf0WxBFsNQhYG8Wz 0q1o_3uRs_aNM0wJtGeSohS3fABN8C z-kYxdfOikF1zi4nE1wn8YUw8lfsCxeUI ioUp4Lz0GaXhHkkXxC5jssOsuOyW_6 CeDYrx0AOAy1URHBY.4xc_PNzUrZe FFz0FV9hV7uu_-Es6f3jhi6koImPVcgw &dib_tag=se&keywords=m4x70%2Bvi ti%2Btesta%2Bsvasata&nsdOptOutPa ram=true&qid=1734712182&s=industri al&sprefix=m4x70%2Bviti%2Btesta%2 Bsvasata%2Cindustrial%2C134&sr=1-9&th=1 | M4x70 - Flat head |
| Screws | Aerzetix | https://www.amazon.it/AERZETIX-cilin drica-impronta-intaglio-montaggio/dp/B 0BLNJ9PTX/ref=sr_1_8?__mk_it_IT=% C3%85M%C3%85%C5%BD%C3%95 %C3%91&crid=3AESCF6VKG6B6&di b=eyJ2IjoiMSJ9.3HlweLtFpKk8huuM4 tfcAbDSl2biH6iWGO7PXpXKxzQnVM cKVrvCI0CkZanzuaduqvaeCs8DLxlFs c_s4-qvSDCE8dvXDhuO4nHPwIhPAl BnL5G6tLMycq0ZS0ZN83GAj_VOUc GVLq4fdcfeurabYdPzrWf3PpwM6_-y OlEOLVvlzzBKgQWO7fiMSCFuGA6 X4j8xVlNzobgrSfsnDNqZpEgH289rp AslvAA9D2YEmvJSm0CRqWecsKPJ iB8yNhelz01McT-qy3s6utg4_Thec18s iTwIY2bFdQ5XYhkTzBxAF86lr6VMXZ 3Ym26ZiedQE1_7ceB7jYcyQ5mOMR fx0d8pYVpg6Y2OqzifmZzNsQM._rEh uS8xCOyhS7xqQQe9H3kwn-ppl22Rf VmzFpFpyW0&dib_tag=se&keyword s=m4x80%2Bviti%2Btesta%2Bsvasat a&nsdOptOutParam=true&qid=17347 12214&s=industrial&sprefix=m4x80% 2Bviti%2Btesta%2Bsvasata%2Cindu strial%2C141&sr=1-8&th=1 | M4x80 - Flat head |
| Screws | Globstar | https://www.amazon.it/testa-svasata- impronta-Phillips-acciaio/dp/B083RJ S9NT/ref=sr_1_30?__mk_it_IT=%C3 %85M%C3%85%C5%BD%C3%95% C3%91&crid=15MBBIPYCF06S&dib =eyJ2IjoiMSJ9.wHpVBrZH8GsdSIko N0T0-2663KN2Tr-H4orSaQtmYf64T nTN15zshBEbfwlrPNyJaD4F1cXZvO Evle2B5qQb_Un-tR8WzVwAxQFWe SMPaYKjM5ZgpbhFaaS6Zj0BQntEIA hyTnvnLg4IBEE9y-4jQ66ZIkmXcWFo zXK-eTFnle6uFI4nXhbCXTWjM5MP_ sNolDP0O4HS7Bnxk_wanuwC9zEuH ZeVNsejl6gwYR0IlJK5iu0V9Y-bgFcr1- iTBhrARDKG7-0Nc5188TSaN9snT7K 6yO2J3ctfC24PTGnaZAFYKMvrNqz OAihVBKIW8xOhIx0ZRipPa9ggsMs _pe1VgY2dCXtUrE_bo1dd4oGlUuw .v5iiMZ2rMQlZlZs2WDyqM_mo1utvu PD71Jyb-O7BY1Y&dib_tag=se&key words=m5x8+viti+testa+svasata&ns dOptOutParam=true&qid=17347123 79&s=industrial&sprefix=m5x8+viti+ testa+svasata%2Cindustrial%2C201 &sr=1-30 | M5x8 - Flat head |
| Screws | Globstar | https://www.amazon.it/testa-svasata- impronta-Phillips-acciaio/dp/B083RH M4BY/ref=sr_1_30?__mk_it_IT=%C3 %85M%C3%85%C5%BD%C3%95% C3%91&crid=15MBBIPYCF06S&dib= eyJ2IjoiMSJ9.wHpVBrZH8GsdSIkoN 0T0-2663KN2Tr-H4orSaQtmYf64Tn TN15zshBEbfwlrPNyJaD4F1cXZvO Evle2B5qQb_Un-tR8WzVwAxQFW eSMPaYKjM5ZgpbhFaaS6Zj0BQnt EIAhyTnvnLg4IBEE9y-4jQ66ZIkmX cWFozXK-eTFnle6uFI4nXhbCXTW jM5MP_sNolDP0O4HS7Bnxk_wan uwC9zEuHZeVNsejl6gwYR0IlJK5iu 0V9Y-bgFcr1-iTBhrARDKG7-0Nc5 188TSaN9snT7K6yO2J3ctfC24PTG naZAFYKMvrNqzOAihVBKIW8xOhI x0ZRipPa9ggsMs_pe1VgY2dCXtUr E_bo1dd4oGlUuw.v5iiMZ2rMQlZlZs 2WDyqM_mo1utvuPD71Jyb-O7BY 1Y&dib_tag=se&keywords=m5x8% 2Bviti%2Btesta%2Bsvasata&nsdOpt OutParam=true&qid=1734712379&s =industrial&sprefix=m5x8%2Bviti%2 Btesta%2Bsvasata%2Cindustrial%2 C201&sr=1-30&th=1 | M5x10 - Flat head |
| Screws | Globstar | https://www.amazon.it/testa-svasata- impronta-Phillips-acciaio/dp/B083RH 9SYZ/ref=sr_1_30?__mk_it_IT=%C3 %85M%C3%85%C5%BD%C3%95% C3%91&crid=15MBBIPYCF06S&dib =eyJ2IjoiMSJ9.wHpVBrZH8GsdSIko N0T0-2663KN2Tr-H4orSaQtmYf64T nTN15zshBEbfwlrPNyJaD4F1cXZv OEvle2B5qQb_Un-tR8WzVwAxQF WeSMPaYKjM5ZgpbhFaaS6Zj0BQ ntEIAhyTnvnLg4IBEE9y-4jQ66ZIkm XcWFozXK-eTFnle6uFI4nXhbCXT WjM5MP_sNolDP0O4HS7Bnxk_w anuwC9zEuHZeVNsejl6gwYR0IlJK5 iu0V9Y-bgFcr1-iTBhrARDKG7-0Nc5 188TSaN9snT7K6yO2J3ctfC24PTG naZAFYKMvrNqzOAihVBKIW8xOhIx 0ZRipPa9ggsMs_pe1VgY2dCXtUrE _bo1dd4oGlUuw.v5iiMZ2rMQlZlZs2 WDyqM_mo1utvuPD71Jyb-O7BY1Y &dib_tag=se&keywords=m5x8%2Bvi ti%2Btesta%2Bsvasata&nsdOptOutP aram=true&qid=1734712379&s=indus trial&sprefix=m5x8%2Bviti%2Btesta% 2Bsvasata%2Cindustrial%2C201&sr= 1-30&th=1 | M6x20 - Flat head |
| SD card | SanDisk | https://www.amazon.it/SanDisk-Ultra- Scheda-memoria-classe/dp/B08GYG6 T12/ref=sr_1_8?crid=3ETH6HJL8VKD A&dib=eyJ2IjoiMSJ9.5omqicizrizfHo8r pPQ6sWJ_C0sfwUqPQv6VrziwK-SCX 4-aSgFGois1pfvTwB0SuDxdNjByo2zU fBonWwUKAf3clazXEIGNl0GhojQmvj Bt_6RS31QcAM5Y50BpAdndMXIHy9 iGTswgYEwnbt9uj56G_soC6OdPiTvr W3M8jwtTxQDUl8UcHZeRMPq0T8sd v4hskHGmipFItAzE1lyDiy0otrqUhz-ph OggXBAM6gMyGf0qBtU1AFwfj81Shl HS2qojCiRq_JLKyBkc_Y4P_V1YtsGc MxnoG6YzYowwEYbX7uTFWJB-Wvc B_vYw-D5s0B-HDSYRSl-C60yhtqhrUw grg9w6PN5U5s01vos4bauIgIN6lOez1 tz2OHwL5YL3QCHKxNnJV0IM42cH0 du8fIEIbUwYC5jRLqowx1UyaQKQ4W WaGozdZ9l5qUoeQ7KU.iOPo2BYLK STjqjB8BevaVgPa5QD7BuP9CieN6u 4_Qeo&dib_tag=se&keywords=sd% 2Bcard%2Bsandisk&nsdOptOutParam =true&qid=1734704871&sprefix=SD% 2Bcard%2Bsan%2Caps%2C141&sr=8-8&th=1 | SanDisk 32 GB Extreme PRO SD card, UHS-I |
| Self-aligning Flange bearing | Brateuanoii | KFL08 | 8 mm inner diameter |
| Slicing software | Ultimaker Cura | https://ultimaker.com/it/software/ ultimaker-cura/?gad_source=1& gclid=CjwKCAiAyJS7BhBiEiwAy S9uNUjnbkNIYNUco6YaZYHcFT 2tqBac23MU4Qx3FaMXjl_UifJ_ k8as3BoCeVkQAvD_BwE | Ultimaker Cura 4.9.0 |
| Springs | YIXISI | https://www.amazon.it/sspa/click?ie =UTF8&spc=MTo4NDAzNDQ1MzM 0OTUyODQzOjE3MzQ3MDQ4MD c6c3BfYXRmOjMwMDM5MDQzMz kzODAzMjo6MDo6&url=%2FYIXIS I-Assortimento-Estensione-Compre ssione-Dispositivi%2Fdp%2FB0DD T3KTBF%2Fref%3Dsr_1_1_sspa% 3F__mk_it_IT%3D%25C3%2585M %25C3%2585%25C5%25BD%25 C3%2595%25C3%2591%26crid% 3D1NT7CA1YJGWK4%26dib%3D eyJ2IjoiMSJ9.IhFmevq0fXTGGwE cGvfUxxLOudQqQcPjSGXlWklXO HY2Ahr2XxWSg6MaNBgZ71N1pQ h9Wkp5c2ary4Hm2xVi4dTH8Ny4 4-OhlcrTn0G8emvX66Zu7U0LGm xGYKyKCGxzlhwYC4EnlmTSH_O JnA5oG-BQzuxFBdCrce0txErCEm S40xONaaYp2R5yMiPDw2PqsPP jLmoSCYs4XctxEvAwn_5rp71t0U QsZNku6-jDiiuh5klpC9DsnjipLFP unRCbITWnw6wslENLdJtPDOGF 8vbO5WFf7PnUy4BZ7tUPt7I.B6j eUKwx1_t0sPaxmSHYVbRd1MG w-kosEYkHuKlPtpQ%26dib_tag% 3Dse%26keywords%3Dmolle%2B YIXISI%26nsdOptOutParam%3Dtr ue%26qid%3D1734704807%26spr efix%3Dmolle%2Byixisi%252Caps %252C137%26sr%3D8-1-spons%2 6sp_csd%3Dd2lkZ2V0TmFtZT1zcF 9hdGY%26psc%3D1 | |
| Stepper motor | Twotrees 1234 | 17HS4401 | Stepper motor bipolar NEMA 17 42 Ncm 1.5 A |
| Stepper motor | STEPPERONLINE | UL1007AWG6 | Stepper motor bipolar NEMA 17 42 Ncm 1.5 A |
| Stepper motor fixing bracket | YOTINO | https://www.amazon.it/gp/product/ B07D7XH97T/ref=ppx_yo_dt_b_ search_asin_title?ie=UTF8&psc=1 | |
| Threaded nut M2 | RS-components | 281-984 | |
| Threaded nut M3 | RS-components | 281-984 | |
| Threaded nut M4 | RS-components | 281-984 | |
| Threaded nut M5 | RS-components | 281-984 | |
| Threaded nut M6 | RS-components | 281-984 | |
| TRAMP - C1 cell line | ATCC | CRL-2730 | Epithelial cell line isolated from the prostate of an adult male transgenic mouse with adenocarcinoma |
| Trypan blue | Gibco Thermofisher | 15250061 | |
| Trypsin | Gibco Thermofisher | 15400054 | Sterile |
References
- Patel, T. et al. Multicellular tumor spheroids: A convenient in vitro model for translational cancer research. Life Sci. 358, 123184 (2024).
- Tanaka, M. et al. Emerging translational research in neurological and psychiatric diseases: From in vitro to in vivo models. Int J Mol Sci. 24 (21), 15739 (2023).
- De Maria Marchiano, R. et al. Translational research in the era of precision medicine: Where we are and where we Will Go. J Pers Med. 11 (3), 216 (2021).
- Knowlton, S. et al. Bioprinting for cancer research. Trends Biotechnol. 33 (9), 504-513 (2015).
- McGranahan, N. et al. Biological and therapeutic impact of intratumor heterogeneity in cancer evolution. Cancer Cell. 27 (1), 15-26 (2015).
- Van Zundert, I. et al. From 2D to 3D cancer cell models-The enigmas of drug delivery research. Nanomaterials. 10 (11), 2236 (2020).
- Hoarau-Véchot, J. et al. Halfway between 2D and animal models: Are 3D cultures the ideal tool to study cancer-microenvironment interactions? Int J Mol Sci. 19 (1), 181 (2018).
- Yamada, K. M. et al. Modeling tissue morphogenesis and cancer in 3D. Cell. 130 (4), 601-610 (2007).
- Manduca, N. et al. 3D cancer models: One step closer to in vitro human studies. Front Immunol. 14, 1175503 (2023).
- Wang, L. et al. The 3D revolution in cancer discovery. Cancer Discov. 14 (4), 625-629 (2024).
- Benton, G. et al. Multiple uses of basement membrane-like matrix (BME/Matrigel) in vitro and in vivo with cancer cells. Int J Cancer. 128 (8), 1751-1757 (2011).
- Benton, G. et al. Matrigel: From discovery and ECM mimicry to assays and models for cancer research. Adv Drug Deliv Rev. 79-80, 3-18 (2014).
- Rawal, P. et al., Prospects for 3D bioprinting of organoids. Bio-Design Manuf. 4 (3), 627-640 (2021).
- Hou, S. et al. Advanced development of primary pancreatic organoid tumor models for high-throughput phenotypic drug screening. SLAS Discov Adv Sci Drug Discov. 23 (6), 574-584 (2018).
- Kane, K. I. W. et al. Determination of the rheological properties of Matrigel for optimum seeding conditions in microfluidic cell cultures. AIP Adv. 8 (12), 125332 (2018).
- De Stefano, P. et al. Bioprinting of Matrigel scaffolds for cancer research. Polymers. 13 (12), 2026 (2021).
- Hatzikiriakos, S. G. et al. Role of slip and fracture in the oscillating flow of HDPE in a capillary. J Rheol. 36 (5), 845-884 (1992).
- Kalika, D. S. et al. Wall slip and extrudate distortion in linear low-density polyethylene. J Rheol. 31 (8), 815-834 (1987).
- Molenaar, J. et al. Modeling polymer melt-flow instabilities. J Rheol. 38 (1), 99-109 (1994).
- Valot, L. et al. Chemical insights into bioinks for 3D printing. Chem Soc Rev. 48 (15), 4049-4086 (2019).
- Sundaramurthi, D. et al. 3D bioprinting technology for regenerative medicine applications. Int J Bioprinting. 2 (2), 9-26 (2016).
- Maloney, E. et al. Immersion bioprinting of tumor organoids in multi-well plates for increasing chemotherapy screening throughput. Micromachines. 11 (2), 208 (2020).
- Snyder, J. E. et al. Bioprinting cell-laden Matrigel for radioprotection study of liver by pro-drug conversion in a dual-tissue microfluidic chip. Biofabrication. 3 (3), 034112 (2011).
- Patil, L. S. et al. Toward measuring the mechanical stresses exerted by branching embryonic airway epithelial explants in 3D matrices of Matrigel. Ann Biomed Eng. 50 (9), 1143-1157 (2022).
- Lai, V. K. et al. Swelling of collagen-hyaluronic acid co-gels: An in vitro residual stress model. Ann Biomed Eng. 44 (10), 2984-2993 (2016).
- Lam, N. T. et al. Fabrication of a Matrigel-collagen semi-interpenetrating scaffold for use in dynamic valve interstitial cell culture. Biomed Mater. 12 (4), 045013 (2017).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved