A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Analysis of Spinal Cord Blood Supply Combining Vascular Corrosion Casting and Fluorescence Microsphere Technique: A Feasibility Study in an Aortic Surgical Large Animal Model
Not Published
In This Article
Summary
This study combines fluorescence microsphere technique and vascular corrosion casting to simultaneously investigate spinal cord blood flow and visualize spinal cord feeding arteries in a large animal model. This model can be employed to investigate morphological vascular alterations and hemodynamic parameters in the same tissue.
Abstract
Spinal cord ischemia after cardiovascular interventions continues to be a devastating problem in modern surgery. The role of intraspinal vascular networks and anterior radiculomedullary arteries (ARMA) in preventing spinal cord ischemia is poorly understood. We are the first to combine the fluorescence microsphere technique and vascular corrosion cast to investigate spinal cord blood supply in a large animal model. Landrace pigs (n= 30, 35.1 ± 3.9 kg) underwent a lateral thoracotomy. Fluorescent microspheres were injected into the left atrium and a reference sample was aspirated from the descending aorta. Repeated measurements of spinal cord and renal cortical blood flow from the left and right kidneys with three different microsphere colors in 5 pigs were taken to validate reproducibility. Spinal cord blood flow to the upper thoracic (T1-T4), mid-thoracic (T5-T8), lower thoracic (T9-T13), and lumbar (L1-L3) levels were determined. After euthanasia, we carried out selective vascular corrosion cast and counted the left and right ARMAs from levels T1-T13. We observed no alterations in blood flow analysis and fluorescence background noise when using vascular corrosion casting in the same tissue. Repeated measurements of cortical renal blood flow and spinal cord blood flow were reproducible. Blood flow analysis of the left and right kidneys revealed a strong correlation (r = 0.94, p<0.001). We detected more left than right ARMAs, with the highest prevalence at T4 (p<0.05). The mean number of ARMAs was 8 ± 2. Their number in the upper thoracic region ranged from 2 to 7 (mean of 5 ± 1), while in the lower thoracic region they ranged from 0 to 5 (mean of 3 ± 1 (p<0.001)). This study shows that combining fluorescence microsphere technique and vascular corrosion cast is well suited for assessing the blood flow and visualizing the arteries at the same time.
Introduction
Microspheres
Since its introduction by Rudolph and Heymann in 1967, microsphere technique has undergone continuous development1. Today it is the gold standard for measuring regional organ perfusion. Microspheres for blood flow studies are typically 15-µm-diameter particles labeled with colored, radioactive, or fluorescent substances. When injected into the left atrium, they mix into the central circulation and trigger microembolization in small capillaries ("trapping"). Blood flow is proportional to the number of microspheres in the region of interest. Following introduction of the reference sample method, it became possible to calculate absolute blood flow in mL/min/g by comparing the number of microspheres in the reference sample, aspirated at a predefined rate downstream to the injection site, with the number of microspheres in the region of interest2. However, the radioactive microspheres that were first introduced were hazardous for both humans and animals because of the radiation burden. Their expense, especially due to their high disposal costs and large animal experimental models, led to new methods3. Fluorescent microspheres have the advantage of great accuracy, very good spectral separation, high reliability, and low cost compared to radioactive microspheres4,5.
Vascular Corrosion Casting and Spinal Cord Anatomy
Vascular corrosion casting has a long history in describing the morphology of vessels and visualizing small vessels that remain otherwise undetectable by the human eye. With the invention of low viscosity resin in 1970, it became possible to study the microvasculature and distribution of small vessels6,7,8. Modifications in resin's viscosity helped to obtain highly detailed vascular castings. In combination with scanning electron microscopy, this method can provide a precise image of the endothelial surface of the vessels9.
In contrast to these advantages, there are certain sources of error that can affect the casts' reliability. Although modern polymers have improved the quality of casts, there is still some shrinkage. For example, the average shrinking of the polyurethane-based resin (the same one that we used in our experiments) is reported to be 6.8% after one week10. Furthermore, extravasation and changes in the surface and surrounding tissue have been mentioned11. However, these observations have not been made with the resin we used10. Even though combining the fluorescence microsphere technique and vascular corrosion cast is a useful method to describe the anatomy of the vasculature and determine the tissue perfusion in the same model, no simultaneous usage has been reported to our knowledge.
We are the first to combine fluorescence microsphere technique and vascular corrosion casting in an experimental porcine model to determine spinal cord perfusion and visualize ARMA. The ARMAs are branches from segmental intercostal arteries supplying the anterior spinal artery, and vary in number and distribution. 31 somites are formed during embryological development and receive blood from the corresponding segmental arteries through ARMAs, most of which degenerate, and only 4 to 8 of them remain feeding the anterior spinal artery12. They are therefore crucial for supplying adequate blood flow to the anterior two-thirds of the spinal cord and thus motor functions. Our group's recent investigations suggest that ARMAs play a key role in preventing ischemia after cardiovascular surgery interventions13. The Collateral Network Concept introduced by Griepp describes intraspinal vascular networks that can prevent acute ischemic conditions if segmental arteries become occluded14. ARMAs in this case connect the intraspinal collateral system and extraspinal vessels with the anterior spinal artery, thus their number and the maximum distance between them could be an important preoperative risk predictor in aortic surgery15,16. Figure 1 illustrates the blood supply's schematic to the spinal cord.
Protocol
This study was conducted at the University Medical Center Freiburg, Freiburg, Germany. Institutional Review Board (IRB) approval was obtained before beginning any experiment. The study animals received humane care in compliance with the Guide for the Care and Use of Laboratory Animals and in compliance with the guidelines established by the local German government (Protocol number G 14/39). An experienced veterinarian carried out anesthesia, pain control, perioperative monitoring, and euthanasia.
1. Preparation for Surgery
- Allow pigs to house in ventilated rooms and fast them 18 h before surgery. Provide water ad libitum.
- Premedicate the pigs with an intramuscular injection of ketamine (20 mg/kg) and 0.5 mg/kg of body weight (BW) midazolam.
- Insert an 18 G intravenous cannula into an ear and deepen anesthesia with propofol (2-4 mg/kg BW) intravenously (i.v.). Carry out orotracheal intubation with a 6.5 Fr tracheal tube. Ensure adequate ventilation and oxygenation by ventilation with a positive end-expiratory pressure (PEEP) of 5 cm H2O, respiratory frequency of 12-14 min-1, and a tidal volume of 8 mL/kg BW.
- Maintain anesthesia with isoflurane 1.5-2% in O2/Room Air (FiO2 = 0.6) in combination with fentanyl (5-10 µg/kg/h) and vecuronium (0.2-0.4 mg/kg/h).
- Perform electrocardiogram, pulse oximetry, and temperature monitoring. Use vet ointment on eyes to prevent dryness under anesthesia. Carry out adequate pain control with fentanyl (5-10 µg/kg/h) i.v. and monitor heart rate and pain reactions.
- Under sterile conditions, dissect free the common carotid artery and external jugular vein carefully using scissors, and cannulate with a 3-French-catheter using the Seldinger technique17. This step is taken to monitor central venous (CVP) and mean arterial pressure (MAP) via pressure transducer and amplifier.
2. Microsphere Injection
- While maintaining sterile conditions, carry out a left posterolateral thoracotomy by an incision in the 5/6 intercostal space using a #10 scalpel blade for the initial incision. Open the situs with scissors and fingers. Open the parietal pleura by an incision and anesthetize the intercostal nerves by injecting 1-2 mL mepivacaine (2%, 20-40 mg). Open the situs by introducing a rib spreader.
- Dissect the thoracic aorta free using scissors, tweezers, and fingers, and introduce a 3-French-catheter into the aorta to withdraw microsphere reference samples. Connect a three-way-stop-cock for blood sampling.
- Open the pericardium using a scissor and insert a 14-G-cannula into the left atrium through the left atrial appendage. Secure the cannula with a 4-0-prolene suture for microsphere injections. Rinse with approximately 10 mL saline to maintain patency for microsphere injection.
- Calculate the minimum number of microspheres to be injected using the formula:
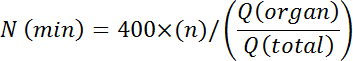
Where, N (min)= minimum number of microspheres required for the injection, n = total number of organ pieces, Q (organ)/Q (total) = fraction of total cardiac output supplying the organ of interest.
NOTE: There should be a minimum number of microspheres in the region of interest to ensure highly accurate measurements18. Here, 2.5 million microspheres were used for the injection. - Store the vials containing the fluorescent microspheres in a refrigerator at 2-8 °C and protect them from light. Vortex the fluorescence microsphere vials containing 10 mL solution (one million microspheres per mL) for 20 s and place them in a cold ultrasonic water bath for 5 min.
NOTE: Because the heat generated might damage the microsphere particles, do not leave them in the ultrasonic bath for too long. - Dilute 2.5 mL (2.5 million) of microspheres with 7.5 mL sodium chloride in a 10-mL plastic syringe.
NOTE: Carry out injection immediately after aspiration into the syringe. Aggregation of microspheres leads to inaccurate measurements. - Inject the microsphere solution into the previously introduced left atrial cannula at a steady injection rate lasting 60 s. Inject only in hemodynamically stable pigs to ensure good microsphere distribution in the cardiovascular system and accurate measurements.
- Aspirate the reference blood sample through the aortic catheter with a withdrawal pump at a predefined aspiration rate of 4.55 mL/min into a 20-mL syringe.
- Start aspiration 15 s before the microsphere injection, and continue for an additional 180 s for a total of 195 s. This step guarantees that all microspheres are "trapped" in the tissue and reference sample, and can be used to calculate blood flow.
- Transfer the blood samples into tubes. Rinse the syringe with 5 mL 2% Tween 80 solution and add it to the tube.
- Repeat steps 2.5-2.8 with fluorescent microspheres of different colors that to do not exhibit spectral overlap of excitation and emission wavelengths to obtain blood flow at different time points.
NOTE: The time points can be chosen according to the experimental setup. Up to 7 different microsphere colors can be used in the same model without performing correction calculations19. Adequate postsurgical treatment, including treatment of postsurgical pain and recovery conditions are obligatory for survival strategies and long-term experiments. Do not leave an animal unattended until it has regained sufficient consciousness to maintain sternal recumbency. Do not return an animal that has undergone surgery to the company of other animals until it has fully recovered. - Perform euthanasia using thiopental (20 mg/kg BW i.v.), potassium (5 mmol/kg BW i.v.), and exsanguination by cutting the inferior vena cava under isoflurane anesthesia.
3. Vascular Corrosion Casting
- Immediately after sacrifice, place the animals in supine position.
- Place a 14-French-catheter in the descending aorta, fix it with a 4-0-prolene suture and flush with 500 mL saline with heparin.
- Mix 50 mg blue vascular casting pigment in 50 g casting resin until a dark blue solution is obtained. Mix the dilution solution (74.1 mL ethanol, 10 mL 2-propanol, and distilled water in 100 mL solution) and 5 g hardener into the resin. Adjust the amount of casting material and solvent needed for injection depending on the experimental setup.
NOTE: Ethylmethylketone or dichloromethane can also be used as a dilution solution. A dilution of up to 40% is recommended. - Draw the prepared casting material into a 50-mL plastic syringe and immediately inject it manually with high pressure into the aortic catheter.
NOTE: The use of an injection apparatus enables pressure monitoring. A physiological pressure of 90-120 mmHg is recommended.- During injection, maintain high pressure by closing the catheter manually. Perform adequate suction of casting material through the inferior vena cava vein. After the injection, adjust the operating table for a better distribution into the organ of interest.
4. Autopsy
NOTE: Autopsy is performed the day after the vascular corrosion casting procedure. Store the pigs in a freezer overnight at approximately −10 °C.
- Place the animal in prone position and make a longitudinal incision above the dorsal spine processes. Carryout a midline incision from the cervical region to the sacrum using a #10 scalpel blade.
- Dissect the paraspinal muscles off the vertebral column and expose the spinal cord via laminectomies using a bone Rongeur.
- After removing fatty tissue in the spinal canal using an anatomical tweezer, count the anterior radiculomedullary arteries from segments T1-T13. Identify the segments through the origins of the spinal nerves.
- Dissect the spinal cord at each segment for blood flow analysis using a disposable microtome blade, and put the tissue in 15-mL polypropylene tubes. Perform the dissection in the middle of two consecutive spinal nerves. Do not use polyethylene tubes because the digesting solution used in the tissue processing will also digest the tubes.
- Expose the left and right kidneys taking a posterior surgical approach between segment T12 and the iliac crest using a scalpel and scissors. Deepen the incision through the latissimus dorsi muscle, and remove fat and parts of the lumbodorsal facia with a tweezer until reaching the renal fossa.
- After the incision into the renal fascia with a scissor, dissect the kidneys free using a scissor and fingers, and remove. Perform this step after removing the paraspinal muscles (step 4.2). Dissect the outer renal cortical part of the left and right kidneys using a #11 scalpel, put them into the tubes for blood flow analysis, and store them in the dark at room temperature.
NOTE: Perform this step to validate microsphere distribution and reproducibility of blood flow analysis due to the simultaneous usage of casting material in the same tissue.
5. Tissue Processing
NOTE: Blood and tissue samples are processed via a modified sedimentation technique for lipid-rich tissues20.
- Allow the samples to rest for 2 weeks in the dark at room temperature (18-22 °C) for autolysis to occur. The samples need not be stored in solution.
- After 2 weeks, place 7 mL of 2.3 M KOH with 0.5% Tween 80 into each tube. Vortex for 20 s, and place in a 50 °C water bath for 48 h.
- After 48 h, centrifuge at 2,000 x g for 20 min at 20 °C; the microspheres are pelleted at this step. Remove the supernatant until there is a volume of 1 mL.
- Next, add 7 mL of Triton X-100 and vortex again. Centrifuge at 2,000 x g for 20 min at 20 °C and discard the supernatant until a volume of approximately 1 mL is obtained.
- To neutralize KOH, add 7 mL of dilute buffer (5.88 g K2HPO4 in 200 mL distilled water and 22.9 g K2HPO4 in 800 mL distilled water; combine the solutions) and vortex the tubes.
- Following the next centrifugation at 2,000 x g for 20 min at 20 °C, remove all but approximately 150 µL of the supernatant.
NOTE: This step is crucial. Execute this step very carefully to minimize microsphere loss. - Finally, add exactly 3 mL of 2-ethoxyethylacetate and allow the tubes to rest for 5 days in the dark at room temperature. This step will release the fluorescent dyes from the microsphere particles.
NOTE: An exact volume of 2-ethoxyethylacetate is crucial because the fluorescence-intensity measurements depend on the concentration. The tube should be placed in the dark, because the fluorescent dyes are no longer bound to the particles and the fluorescence intensity will weaken due to light exposure ("quenching"). - After the last centrifugation step at 2,000 x g for 20 min at 20 °C, take out the supernatant and measure the fluorescence intensity in the spectrometer.
NOTE: A less time-consuming wavelength program can be used to take repeated measurements at different excitation and emission wavelengths according to the microsphere colors used in the experiment. Excitation (Ex) and Emission (Em) wavelength of red, green, and yellow fluorescent microspheres: Red (Ex/Em), green (Ex/Em), yellow (Ex/Em) = 568/595, 455/482, 508/538, respectively. - Calculate the regional blood flow (mL/min/g) using the following formula:
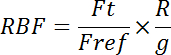
With RBF: regional blood flow; Ft: fluorescence intensity of tissue sample; Fref: fluorescence intensity of reference sample; R: withdrawal rate of pump; g: tissue weight in grams.
Results
Blood Flow Analysis:
We observed no alterations in the blood-flow analysis results and background fluorescence noise when using corrosion-casting material in the same tissue. To prevent background fluorescence with the combined technique, the microsphere colors and inherent background fluorescence of the casting pigment must not exhibit spectral overlap of their excitation and emission wavelengths. Furthermore, spectral overlap wit...
Discussion
We are the first to have combined the fluorescence microsphere technique and vascular corrosion cast in this feasibility study to visualize spinal cord vasculature and determine spinal cord blood flow at the same time.
The spinal cord's thoracic region has some important features that must be considered in cardiovascular surgery. The anterior spinal artery's supply of blood through the anterior radiculomedullary arteries leaves watershed areas with decreased blood flow next to the regi...
Disclosures
The authors have no financial or personal relationship to disclose that would create a conflict of interest or bias.
Acknowledgements
This study was funded by the Deutsche Forschungsgemeinschaft, German Research Foundation (DFG) Grant Nr. KA 3872/2-1. We thank Dr. Amir Alkalak for providing Figure 1.
Materials
| Name | Company | Catalog Number | Comments |
| Fluospheres Green | Molecular Probes | F21010 | |
| Fluospheres Red | Molecular Probes | F8842 | |
| Fluospheres Yellow | Molecular Probes | F21011 | |
| Biofuge Primo R | Thermo Scientific | ||
| Water Bath GFL Type 1083 | GFL | ||
| Weighting Scale | SCALTEC | ||
| Fluorescence spektrometer LS55 | Perkin Elmer | ||
| PU4ii resin, hardener, pigment | vasQtec | blue pigment was used | |
| 15-ml-conicial-tubes | TPP | 91015 | |
| Cellosolve acetate | Sigma-Aldrich | 109967 | |
| potassium hydroxide >85% | Carl Roth | P747.1 | |
| Ethanol >99.5% | Carl Roth | 5054.1 | |
| Triton X-100 | Sigma-Aldrich | X100 | |
| Withdrawal pump 11 | Harvard Apparatus | ||
| 3-way stopcocks | B. Braun | 16496 | |
| Perfusor lines PVC 150 cm/2.6 ml | B. Braun | 8722960 | |
| 4-0 prolene suture | Ethicon | EH7411H | |
| Syringes | B. Braun | 10ml, 20ml and 50ml | |
| Vasofix Braun 18G | B.Braun | 4268130B | |
| Dormicum Midazolam 5mg/ml | Roche Pharma | 3085793 | |
| Ketamin 10% | Medistar GmbH | 3048734 | |
| Kaliumchlorid | B.Braun | 3140598 | |
| tracheal tubus 6.5mm | Rüsch | 112482 | |
| Fentanyl | Janssen-Cilag | ||
| Mepivacain Scandicain 2% | AstraZeneca | 910860 | |
| Microtome blade S35 | Feather | 207500000 | |
| disposable blade #11, #10 | B.Braun | 8080168 | |
| French-14-catheter | B.Braun | 4476142 | |
| French-3-catheter | Vygon | 115,094 |
References
- Rudolph, A. M., Heymann, M. A. The Circulation of the Fetus in Utero. Circ Res. 21 (2), 163 (1967).
- Malik, A. B., Kaplan, J. E., Saba, T. M. Reference sample method for cardiac output and regional blood flow determinations in the rat. J Appl Physiol. 40 (3), 472 (1976).
- Prinzen, F. W., Bassingthwaighte, J. B. Blood flow distributions by microsphere deposition methods. Cardiovasc Res. 45 (1), 13-21 (2000).
- Van Oosterhout, M. F., Willigers, H. M., Reneman, R. S., Prinzen, F. W. Fluorescent microspheres to measure organ perfusion: validation of a simplified sample processing technique. Am J Physiol - Heart Circ Physiol. 269 (2), H725 (1995).
- Glenny, R. W., Bernard, S., Brinkley, M. Validation of fluorescent-labeled microspheres for measurement of regional organ perfusion. J Appl Physiol. 74 (5), 2585 (1993).
- Fujita, T., Murakami, T. Microcirculation of monkey pancreas with special reference to the insulo-acinar portal system. A scanning electron microscope study of vascular casts. Arch Histol Jpn Nihon Soshikigaku Kiroku. 35 (4), 255-263 (1973).
- Bielke, S. R., Nagle, R. B., Trump, B. F., Bulger, R. E. Scanning electron microscopy of renal vascular casts. J Microsc. 108 (1), 89-96 (1976).
- Dollinger, R. K., Armstrong, P. B. Scanning electron microscopy of injection replicas of the chick embryo circulatory system. J Microsc. 102 (2), 179-186 (1974).
- Murakami, T. Application of the scanning electron microscope to the study of the fine distribution of the blood vessels. Arch Histol Jpn Nihon Soshikigaku Kiroku. 32 (5), 445-454 (1971).
- Krucker, T., Lang, A., Meyer, E. P. New polyurethane-based material for vascular corrosion casting with improved physical and imaging characteristics. Microsc Res Tech. 69 (2), 138-147 (2006).
- Aharinejad, S., Franz, P., Böck, P., Lametschwandtner, A., Breiteneder, H., Firbas, W. Sphincterlike structures in corrosion casts. Scanning. 12 (5), 280-289 (1990).
- Bosmia, A. N., Hogan, E., Loukas, M., Tubbs, R. S., Cohen-Gadol, A. A. Blood supply to the human spinal cord: part I. Anatomy and hemodynamics. Clin Anat. 28 (1), 52-64 (2015).
- Kari, F. A., et al. New Insights into Spinal Cord Ischemia after Thoracic Aortic Procedures: The Importance of Anterior Radiculo-Medullary Artery Anatomy for Surgical Outcome. Thorac Cardiovasc Surg. 65, S1-S110 (2017).
- Griepp, E. B., Griepp, R. B. The Collateral Network Concept: Minimizing Paraplegia Secondary to Thoracoabdominal Aortic Aneurysm Resection. Tex Heart Inst J. 37 (6), 672-674 (2010).
- Kari, F. A., et al. Immediate Spinal Cord Collateral Blood Flow During Thoracic Aortic Procedures: The Role of Epidural Arcades. Semin Thorac Cardiovasc Surg. 28 (2), 378-387 (2016).
- Kari, F. A., et al. Immediate Spinal Cord Backup Flow during Frozen Elephant Trunk Procedure: Intraspinal or Paraspinal System?. Thorac Cardiovasc Surg. 64, (2016).
- Seldinger, S. I. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta Radiol. 39 (5), 368-376 (1953).
- Buckberg, G. D., Luck, J. C., Payne, D. B., Hoffman, J. I., Archie, J. P., Fixler, D. E. Some sources of error in measuring regional blood flow with radioactive microspheres. J Appl Physiol. 31 (4), 598-604 (1971).
- Schimmel, C., Frazer, D., Glenny, R. W. Extending fluorescent microsphere methods for regional organ blood flow to 13 simultaneous colors. Am J Physiol - Heart Circ Physiol. 280 (6), H2496 (2001).
- Power, K., Schimmel, C., Glenny, R., Bernards, C. Cerebral blood flow determinations using fluorescent microspheres: variations on the sedimentation method validated. J Neurosci Methods. 87 (2), 159-165 (1999).
- Meffert, P., Bischoff, M. S., Brenner, R., Siepe, M., Beyersdorf, F., Kari, F. A. Significance and function of different spinal collateral compartments following thoracic aortic surgery: immediate versus long-term flow compensation. Eur J Cardiothorac Surg. 45 (5), 799-804 (2014).
- Zülch, K. J. Deficient circulation in the border zone of the two vascular regions as a cause of hitherto unexplained injuries of the spinal cord. Dtsch Z Nervenheilkd. 172 (1), 81-101 (1954).
- Zülch, K. J. Pathogenetic and clinical observations in spinovascular insufficiency. Zentralbl Neurochir. 37 (1), 1-13 (1976).
- Cheshire, W. P., Santos, C. C., Massey, E. W., Howard, J. F. Spinal cord infarction: etiology and outcome. Neurology. 47 (2), 321-330 (1996).
- Shamji, M. F., Maziak, D. E., Shamji, F. M., Ginsberg, R. J., Pon, R. Circulation of the spinal cord: an important consideration for thoracic surgeons. Ann Thorac Surg. 76 (1), 315-321 (2003).
- Hickey, R., Albin, M. S., Bunegin, L., Gelineau, J. Autoregulation of spinal cord blood flow: is the cord a microcosm of the brain?. Stroke. 17 (6), 1183-1189 (1986).
- Gillilan, L. A. The arterial blood supply of the human spinal cord. J Comp Neurol. 110 (1), 75-103 (1958).
- Aminoff, M. J. . Neurology and General Medicine. , (2008).
- Adamkiewicz, A. . Die Blutgefässe des menschlichen Rückenmarkes. I. Theil. Die Gefässe der Rückenmarkssubstanz. Sitzungsberichten der Kaiserlichen Akademie der Wissenschaften, Mathematisch-naturwissenschaftliche Classe. 84, 469-502 (1881).
- Gailloud, P. The artery of von Haller: a constant anterior radiculomedullary artery at the upper thoracic level. Neurosurgery. 73 (6), 1034-1043 (2013).
- Henson, R. A., Parsons, M. Ischaemic lesions of the spinal cord: an illustrated review. QJM: Int J Med. 36 (2), 205-222 (1967).
- Perk, J. Spinal Cord Diseases. Encyclopedia of the Neurological Sciences. , 264-268 (2014).
- Dommisse, G. F. The blood supply of the spinal cord. A critical vascular zone in spinal surgery. J Bone Joint Surg Br. 56 (2), 225-235 (1974).
- Suh, T., Alexander, L. Vascular system of the human spinal cord. Arch Neurol Psychiatry. 41, 659-677 (1939).
- Etz, C. D., et al. Spinal cord blood flow and ischemic injury after experimental sacrifice of thoracic and abdominal segmental arteries. Eur J Cardio-Thorac Surg. 33 (6), 1030-1038 (2008).
- Kieffer, E., Fukui, S., Chiras, J., Koskas, F., Bahnini, A., Cormier, E. Spinal cord arteriography: A safe adjunct before descending thoracic or thoracoabdominal aortic aneurysmectomy. J Vasc Surg. 35 (2), 262-268 (2002).
- Williams, G. M., Roseborough, G. S., Webb, T. H., Perler, B. A., Krosnick, T. Preoperative selective intercostal angiography in patients undergoing thoracoabdominal aneurysm repair. J Vasc Surg. 39 (2), 314-321 (2003).
- Kieffer, E., Fukui, S., Chiras, J., Koskas, F., Bahnini, A., Cormier, E. Spinal cord arteriography: A safe adjunct before descending thoracic or thoracoabdominal aortic aneurysmectomy. J Vasc Surg. 35 (2), 262-268 (2002).
- Nijenhuis, R. J., Krings, T., Mull, M., Thron, A., Wilmink, J. T., Backes, W. H. Non-invasive Spinal Cord Angiography for Imaging Vascular Spinal Cord Malformations. Neuroradiol J. 22 (Suppl 1), 107-114 (2009).
- Kawaharada, N., et al. Magnetic resonance angiographic localization of the artery of Adamkiewicz for spinal cord blood supply. Ann Thorac Surg. 78 (3), 851-852 (2004).
- Hyodoh, H., et al. Usefulness of Preoperative Detection of Artery of Adamkiewicz with Dynamic Contrast-enhanced MR Angiography. Radiology. 236 (3), 1004-1009 (2005).
- Backes, W. H., Nijenhuis, R. J., Mess, W. H., Wilmink, F. A., Schurink, G. W. H., Jacobs, M. J. Magnetic resonance angiography of collateral blood supply to spinal cord in thoracic and thoracoabdominal aortic aneurysm patients. J Vasc Surg. 48 (2), 261-271 (2008).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved