昇華によってフェロセンの精製
概要
ソース: タマラ ・ m ・力、化学のテキサス A & M 大学
昇華、最初、液体になることがなく気体に固体の直接相転移が起こる温度と圧力より低い化合物の三重点 (図 1) という。昇華のプロセスは、有機と無機の固体を浄化するために利用できます。浄化技術の中には、気相に直接固体を加熱します。すべての非揮発性の不純物は、気化した化合物は、冷たい表面に固体として収集する (蒸着) 取り残されます。昇華を使用してフェロセン、183 ° C の三重点温度と無機固体を浄化するためにここでは、1
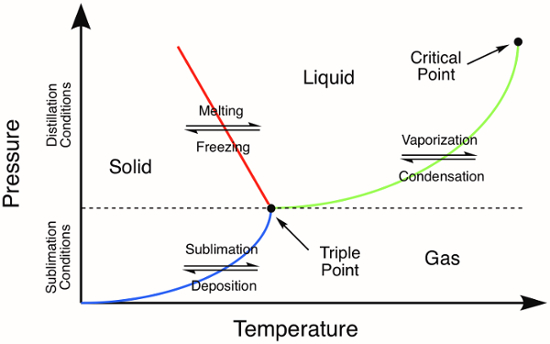
図 1.汎用的な相図。色付きの線は、相転移の圧力と温度の要件を表します。固体の蒸留は、圧力と相図の緑の線で表される、トリプル ポイントの上の温度で発生します。青い線は昇華が発生する温度と圧力の条件を表します。
手順
1. Schlenk ラインのセットアップ
詳細な手順、有機化学の基本シリーズで「Schlenk ライン転送の溶剤」と「液体の脱気」の動画をご覧ください。Schlenk ラインの安全は、この実験を行う前に確認必要があります。ガラスは、使用前に星の亀裂の検査する必要があります。リキッド N2を使用している場合に、Schlenk ライン トラップに O2がない凝縮されて確保するため注意が必要があります。温液体 N2 O2凝縮し、有機溶媒存在下で爆発。O2が凝縮されている、または青い液体がコールド トラップで観測されたことが疑われる場合は動的真空下ではコールド トラップを残します。リキッド N2トラップを削除したり、真空ポンプをオフにしないでください。-ポンプに液体 O2が崇高な時間をかけてこそすべて O2の昇華が一度リキッド N2のトラップを削除しても安全です。
結果
申請書と概要
参考文献
- Kaplan, L., Kester, W. L., Katz, J. J. Some properties of iron biscyclopentadienyl. J Am Chem Soc. 74, 5531-5532 (1952).
タグ
スキップ先...
このコレクションのビデオ:

Now Playing
昇華によってフェロセンの精製
Inorganic Chemistry
54.6K 閲覧数

シュレンク管を用いた Ti(III) メタロセンの合成
Inorganic Chemistry
31.6K 閲覧数

グローブ ボックスと不純物センサー
Inorganic Chemistry
18.6K 閲覧数

エバンス メソッド
Inorganic Chemistry
68.5K 閲覧数

単結晶および粉末 x 線回折
Inorganic Chemistry
104.4K 閲覧数

電子常磁性共鳴 (EPR) 分光法
Inorganic Chemistry
25.4K 閲覧数

メスバウアー分光法
Inorganic Chemistry
22.0K 閲覧数

Ph3P BH3ルイス酸-塩基相互作用
Inorganic Chemistry
38.9K 閲覧数

フェロセンの構造
Inorganic Chemistry
79.5K 閲覧数

群論の赤外分光法への応用
Inorganic Chemistry
45.2K 閲覧数

分子軌道 (MO) 理論
Inorganic Chemistry
35.3K 閲覧数

Quadruply 金属-金属接合外輪
Inorganic Chemistry
15.3K 閲覧数

色素増感太陽電池
Inorganic Chemistry
15.8K 閲覧数

酸素運ぶのコバルト (ii) 錯体の合成
Inorganic Chemistry
51.7K 閲覧数

根本的な重合反応の光化学開始
Inorganic Chemistry
16.7K 閲覧数
Copyright © 2023 MyJoVE Corporation. All rights reserved