A subscription to JoVE is required to view this content. Sign in or start your free trial.
A Renal Denervation Approach to Prevent Inflammation and Fibrogenesis in Chronic Kidney Disease
Not Published
In This Article
Summary
Described here is a protocol for renal denervation that is used to define the role of renal nerve-derived signaling in persistent renal tubular injury, inflammation, and fibrogenesis. It is focused on sympathetic nerve-mediated signaling.
Abstract
Chronic kidney disease (CKD) is affecting increased numbers of people across the world, and there remains no effective treatment strategy. Sympathetic nerve activation has been recognized as an important factor in the development and progression of cardiovascular disease, hypertension, and CKD. Catheter-guided renal denervation is useful to control blood pressure (BP) in patients with resistant hypertension and CKD. Sympathetic nerve-derived norepinephrine (NE) has been implicated in tissue homeostasis and the progression of various diseases, including CKD. The molecular mechanisms and signaling pathways triggered by sympathetic nerve activation, which drive renal inflammation and fibrogenesis in CKD progression, remain undefined. Presented here is the detailed methodology for renal denervation (RDNx) in experimental models of CKD. The results show that this method effectively ablates the renal nerve, as evidenced by the loss of tyrosine hydroxylase immunoreactivity and levels of kidney NE. This results in the suppression of renal tubular injury, inflammation, and fibrogenesis in CKD models. Competence of surgeons performing surgical procedures to denervate the kidney is a requirement to achieve consistent results. RDNx can be utilized to study the roles of renal nerve, nerve-derived neurotransmitters, and factors, as well as unveil their downstream signaling pathways. Defining the molecular mechanisms and underlying functions will lead to the design of novel therapeutic interventions for CKD, regardless of its etiology (e.g., diabetes, hypertension, and cardiovascular complications).
Introduction
CKD, characterized by tubular injury, persistent renal inflammation, and fibrosis, ultimately leads to end stage kidney disease (ESKD)1,2,3. Sympathetic nervous system governs both normal and pathological functions of diverse organ systems, including those in the kidney4. One type of catecholamine, norepinephrine (NE or noradrenaline), originates from sympathetic neurons and is an effector of the sympathetic nervous system5. In both patients and experimental models, increased sympathetic nerve activity and tissue levels of NE are hallmarks of CKD progression6,7,8.
Renal denervation (RDNx) is used as a therapeutic option for the treatment of drug-resistant hypertension9,10,11,12, but the underlying molecular mechanisms are not yet fully understood. Moreover, studies13,14 defining the role of renal nerve in CKD progression using chemical denervation or antagonists/agonists of sympathetic nerve-derived neurotransmitters (or its receptors) are limited, due to systemic effects that are not specific to the kidney. As reported15,16,17, RDNx can overcome this limitation and has been successful in defining (in vivo) the roles of the renal nerve, renal nerve-derived factors, and downstream signaling pathways in eliciting renal inflammation and fibrosis.
Provided here is a detailed methodology and representative results for the use of RDNx to study renal tubular injury, inflammation, and fibrosis in a mouse model of CKD.
Protocol
Mice were cared for prior to and during the experiment in accordance with the policies of the Institutional Animal Care and Use Committee (IACUC) at the University of Nebraska Medical Center (UNMC), and the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. All portions of the protocol received prior approval from the UNMC-IACUC.
1. Renal denervation
- Use male (129S1/SvImJ) mice (8–10 weeks old) from the Jackson Laboratories.
- Anesthetize mice using a cocktail containing ketamine (200 mg/kg body weight) and xylazine (16 mg/kg body weight), injected intraperitoneally16,18.
- Check for the lack of response to the toe pinch. Shave the left side of mouse’s back with an electric shaver. Clean the region with an alcohol prep pad, and then ensure asepsis using an iodine-based prep pad.
- Maintain the body temperature at 36–38 °C on a heating pad with a temperature controller.
NOTE: Use sterilized surgical instruments for all procedures. - Make an incision 1.5 cm long using a surgical blade on the mouse’s back, 5 mm lateral to the spine.
- Under a surgical microscope, expose the left kidney vessels connected to the kidney through left flank incision. Then, separate the artery and vein carefully from the surrounding connective tissue using micro-dissecting forceps.
- For stripping the nerves, paint the kidney vessels with filter paper soaked in 95% ethanol for 2 min15,16.
NOTE: Ensure that the vessels turn white in color under the microscope. - Wash the kidney vessels for 2 min with PBS. Close the muscular layer with absorbable sutures and close the wounded skin using an autoclip.
- Perform a sham operation in the same manner as above, except strip the kidney nerves.
- After surgery, inject 0.1 µg/kg body weight (BW) buprenorphine subcutaneously (SC) 1x/day for 3 days to reduce surgical pain.
2. Renal fibrosis models
- Unilateral ureteral obstruction (UUO) model
- Two days after renal denervation, anesthetize two male mice (129S1/SvImJ) aged 8–10 weeks as described in step 1.2.
- Check for the lack of response to a toe pinch, then shave the left side of the mouse back.
- Clean the region with an alcohol prep pad and ensure that the region is aseptic using an iodine-based prep pad.
- Maintain the body temperature at 36–38 °C on a heating pad with a temperature controller.
- Expose the left kidney using autoclaved cotton swab through a left flank incision using a surgical blade on the mouse’s back, 1 cm lateral to the spine.
- Ligate the left ureter completely near the kidney and pelvis using a 5-0 silk tie15,18.
- Close the muscular layer with absorbable sutures and close the wounded skin using an autoclip.
- On the other mouse, perform a sham operation using the same surgical procedure, except for ligation of the ureter.
- After surgery, inject 0.1 µg/kg buprenorphine SC 1x/day for 3 days to reduce surgical pain.
- Ischemic acute kidney injury (AKI) to CKD transition model
- Two days after the denervation, anesthetize male 129S1/SvImJ mice aged 8–10 weeks as described in step 1.2.
- Check for the lack of response to a toe pinch and shave the left side of the mouse’s back. Clean the region with an alcohol prep pad and ensure that the region is aseptic using an iodine-based prep pad.
- Maintain the body temperature at 36–38 °C on a heating pad with a temperature controller.
- Expose the left kidney using an autoclaved cotton swab through the left flank incision on the back, 1 cm lateral to the spine, using a surgical blade.
- Subject the mice to 30 min of left renal ischemia by clamping the left renal pedicle with both the artery and vein with nontraumatic microaneurysm clamps, as described previously16,19. Close the incisions temporarily during this ischemic period using clamps.
- Once the ischemic period is over, remove the clamps for reperfusion.
NOTE: Visually confirm reperfusion of the kidneys. The color of the kidneys will change from dark red to bright red upon reperfusion. - Close the muscular layer with absorbable sutures and close the wounded skin using an autoclip.
- After the surgery, inject 0.1 µg/kg BW buprenorphine SC 1x/day for 3 days to reduce surgical pain.
- Perform a sham operation with the same surgical procedure except clamping the renal pedicle.
3. Harvesting of kidneys and blood
- At 1 h, 3 h, 6 h, or 12 h (or 1, 2, 3, or 10 days) post-UUO; or at 0.5, 1, 2, 4, 8, or 16 days post-IRI, anesthetize by placing the mouse’s nose to a 50 mL conical tube with gauze containing isoflurane.
- Collect either 1) 100 µL blood samples from the retroocular vein plexus using a heparinized capillary tube or 2) 500 µL of blood from the vena cava through a midline abdominal incision using a syringe (25 G needle) containing heparin.
- For harvesting kidneys, expose the operated kidney by midline abdominal incision. Cut renal vessels and surrounding tissues with surgical blade and then remove the capsules.
NOTE: Ensure that UUO kidneys have urine filled the pelvis to confirm UUO induction. - Collect plasma from the blood by centrifugation at 8,000 x g for 3 min at room temperature (RT) using a heparinized capillary tube. Store at -20 °C until use.
- For biochemical assays, snap-freeze the kidneys using liquid nitrogen immediately after removal. Store at -80 °C deep freezer until use.
4. Analysis of renal function
- Anesthetize mice 15 days post-ischemia/reperfusion injury (IRI) as described above.
- Expose the right kidney using autoclaved cotton swab through a right flank incision on the back, 1 cm lateral to the spine.
- Remove the kidney (i.e., perform a nephrectomy) from the renal vessels and ureter using scissors, after ligation of the renal vessels and ureter16,20.
- Take blood samples from the retroocular vein plexus 6 h prior to placing the mouse in a metabolic cage.
- To obtain urine samples, place the animals in mouse-specific metabolic cages for 18 h for analysis of glomerular filtration rate (GFR) by creatinine clearance16.
- Measure urinary and plasma concentrations of creatinine using a commercially available kit (Table of Materials). In brief, add 30 µL of plasma to a mixture of Reagents A and B (100 µL each). Then, read the optical density both 1) immediately and 2) at 5 min post-incubation at 510 nm and RT using a microplate reader. For urine, use 50 µL of Reagent A, 50 µL of Reagent B, and 100 µL of water per 5 µL of urine.
- Calculate GFR as follows:
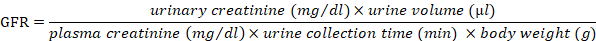
- Alternatively, measure GFR by transcutaneous measurement at 24 h post-Nx, as described previously21. In brief, under anesthesia with isoflurane, place a miniaturized fluorescence detector on the mouse’s back, then inject fluorescein-isothiocyanate (FITC)-conjugated sinistrin (an inulin analog, 0.15 mg/g BW) through the retroocular vein plexus. In conscious and freely moving mice, collect data for the half-life of FITC-sinistrin for 1 h and perform the analysis.
5. Confirmation of renal denervation
- Evaluation of kidney NE level
- Homogenize kidneys frozen at -80 °C in a deep freezer with a glass tissue homogenizer. Use homogenizing solution containing 1 mM EDTA and 4 mM sodium metabisulfite in 400 µL of 0.1 N HCl.
- Centrifuge the homogenates at 13,600 x g and transfer the supernatant to new tube.
- Extract NE from samples by using a cis-diol-specific affinity gel, acylate, then convert enzymatically with a commercially available kit (Table of Materials)15,16.
- Transfer 300 µL of the lysates into the 48 well plate and add 250 µL of distilled water to each well, followed by adding 50 µL of assay buffer and 50 µL of extraction buffer.
- Cover the plate with adhesive foil and incubate for 30 min at RT on a shaker (~600 rpm) then remove the foil, empty, and blot dry by tapping the inverted plate on a hand towel.
- Wash the plate 2x for 5 min at RT using 1 mL of washing buffer by shaking on a platform shaker, followed by blotting dry as done in step 5.1.3.2.
- Add 150 µL of acylation buffer and 25 µL of acylation reagent into the wells, then incubate for 15 min at RT on the shaker.
- Empty the plate and blot dry as done in step 5.1.3.2, then wash 1x for 10 min as done in step 5.1.3.3, followed by blot drying as done in step 5.1.3.2. Add 150 µL of 0.025 M hydrochloric acid into the wells, cover the plate with adhesive foil, and incubate for 10 min at RT on the shaker.
- Add 25 µL of enzyme solution to the pre-coated NE microtiter strips, followed by 20 µL of supernatant (as done in step 5.1.3.5) and standard solutions of NE (0 ng/mL, 5 ng/mL, 20 ng/mL, 75 ng/mL, 250 ng/mL, and 1,000 ng/mL) into each strip. Add 50 μL of the NE antiserum to the strips. Incubate for 2 h at RT on the shaker.
- After discarding the buffer and washing the microtiter strips with 300 μL of washing buffer on the shaker, remove the buffer and blot dry as done in step 5.1.3.2. Incubate for 30 min at RT on the shaker with 100 μL of enzyme conjugate in each well.
- After discarding the buffer and washing the strips, add 100 μL of substrate into each well. Incubate for 20–30 min at RT on a shaker, followed by adding 100 μL of stop solution.
- Read the absorbance of the solution using a microplate reader at 450 nm and calculate the concentration using standard solution as control.
- Immunohistochemistry for tyrosine hydroxylase
- Fix kidneys in 4% paraformaldehyde and process the paraffin embedding with the central part of the fixed kidney with a cross section of 3 – 4 mm thickness.
- Place the paraffin-embedded kidney on a microtome, cut to 3–4 µm thick, and attach the section to the microscope slide.
- Deparaffinize the paraffin-embedded kidney sections with xylene. Rehydrate with 100%, 95%, and 90% ethanol (in this sequence) 2x at each step.
- Permeabilize the kidney sections in 0.1% SDS for 5 min and wash 3x with PBS at RT.
- Autoclave the permeabilized kidney sections in 10 mM sodium citrate at 120 °C for 10 min for antigen retrieval16,22.
- Cool down the autoclaved kidney sections for 30 min at RT and wash 3x with PBS at RT.
- Incubate with 3% BSA in PBS for 30 min at RT to prevent non-specific binding.
- After removal of BSA by suction, incubate the kidney sections with antibody (1:1000) against tyrosine hydroxylase overnight at 4 °C15.
- Next day, wash the kidney sections 3x with PBS at RT and incubate with HRP-conjugated anti-rabbit antibody for 1 h at RT.
- After washing 3x with PBS at RT, apply 3,3 –diaminobenzidine (DAB) to the kidney sections and stain for up to 2 min.
- After washing 3x with PBS at RT, dehydrate the kidney sections with 90%, 95%, and 100% ethanol and xylene for 5 min each. Mount the coverslips with a 1:1 mixture of mounting medium and xylene.
- Western blot analysis for tyrosine hydroxylase
- Isolate whole kidney proteins using lysis buffer with phosphatase inhibitor and protease16,18.
- Run 30 µg of kidney protein on PAGE gel in Tris-glycine-SDS buffer at 80 V for 1 h.
- Transfer the protein to PVDF membrane from gel in Tris-glycine buffer at 80 V for 100 min at 4°C.
- Block the nonspecific proteins on the membrane using 5% skim milk for 30 min at RT.
- Incubate membranes with diluted antibody (1:2,000) against tyrosine hydroxylase in 10 mL of a commercially available blocking solution15,16.
- Use anti-β-actin antibody as loading controls on stripped membranes.
- Expose membranes to the chemi-luminescence substrate solution. Quantify the bands using analysis software.
6. Analysis of tubular injury
- Tubular damage: section paraffin-embedded kidney into 3 µm thick sections using microtome and stain using a periodic acid/Schiff (PAS) stain. First, incubate the sections with 5% PAS stain for 5 min. Then, rinse with distilled water and incubate with Schiff reagent for 15 min.
- Wash PAS-stained kidneys with tap water for 5 min, counterstain with hematoxylin for 1 min, and dehydrate after washing with tap water for 5 min. Cover with mounting medium.
- Select five fields from each PAS-stained kidney randomly in a blinded manner and photograph using light microscopy.
- Score the damaged tubule with flattened lumen according to the loss of brush border, proteinaceous cast, loss of tubular nucleus, and dilation as follows: grade 0 = normal; grade 1 = >10%; grade 2 = 10%–25%; grade 3 = 26%–50%; grade 4 = 51%–75%; grade 5 = <75%15,16,18.
- Apoptotic cell death: analyze apoptotic cell death in paraffin-sectioned kidney samples by performing a TUNEL assay using a commercially available kit (Table of Materials), following the manufacturer’s protocol.
7. Inflammation
- Analysis of neutrophil
- Prepare kidney sections as described in step 5.2 and incubate with antibodies against polymorphonuclear neutrophil overnight at 4 °C, followed by incubation with HRP-conjugated secondary antibody15,16.
- Perform DAB staining and dehydration as described in steps 5.2.10 and 5.2.11.
- Take photographic images from cortical or outer medullary regions in a blinded manner.
- Analysis of macrophage
- Prepare kidney sections as described in section 5.2.
- Incubate the kidneys with antibodies against F4/80 overnight at 4 °C. Incubate with an HRP-conjugated secondary antibody15,16.
- Take images from cortical or outer medullary regions in a blinded manner.
- Analysis of inflammatory cytokine level
- Following section 5.3, prepare kidney samples.
- Incubate the kidney samples with antibodies against ICAM-1, TNF-α, IL-1β, IL-6, and TLR4 overnight at 4 °C. Then, incubate with the corresponding secondary antibodies15.
8. Evaluation of fibrosis
- Sirius red stain
- Following section 5.2, rehydrate the kidney sections.
- Stain the kidney sections with Sirius red solution (0.5 g of Direct Red 80 + 1.3% picric acid in 500 mL of distilled water) for 30 min15,16,18.
- After washing 2x with acidified water (0.5% acetic acid, glacial), physically remove the water from the slides.
- After mounting (section 5.2), take images of cortical or outer medullary regions in a blinded manner.
- Evaluation of fibroblast activation and fibrogenic factors
- Following section 5.3., prepare kidney samples.
- Incubate the kidney samples with antibodies against alpha-SMA, fibronectin, TGF-β, and phospho-Smad3 overnight at 4 °C. Incubate with the corresponding secondary antibodies15.
- Use anti-β-actin antibody as a loading control on stripped membranes.
- Quantify the bands using analysis software.
Results
Removal of renal nerve by renal denervation surgery
Renal denervation (RDNx) was carried out 2 days prior to IRI or UUO to define whether renal nerve contributes to the initiation and development of kidney fibrosis and inflammation. Mice were subjected to either 1) 30 min IRI followed by reperfusion for 1, 2, 4, 8, or 16 days or 2) UUO for 1 h, 3 h, 6 h, or 12 h; 1, 2, 3 or 10 days.
To confirm whether RDNx was successfully applied, tyrosine hydroxylase (TH)-positive symp...
Discussion
This protocol describes the detailed methods for renal nerve ablation in a mouse model. Further, the pivotal role of the renal nerve in triggering inflammatory and fibrotic responses to injury in CKD models is demonstrated. Complete separation of renal artery from connective tissue and veins is a critical step for successful RDNx that allows full exposure of renal nerves and complete nerve ablation. Since there is an overlap of the renal artery over the vein, the portion overlapped is not well-exposed to the alcoho...
Disclosures
The authors declare no competing financial interests.
Acknowledgements
This study is supported by NIH grants DK-116987, DK-120533 and American Heart Association (A.H.A.) Grant in Aid 15GRNT25080031 (B.J.P.), AHA postdoctoral fellowship Grant 15POST25130003 (H.S.J.), and grants (NRF-2016R1C1B2012080 and NRF-2019R1F1A1041410) from the National Research Foundation of Korea (J.K.).
Materials
| Name | Company | Catalog Number | Comments |
| 129S1/SvlmJ | Jax Lab | Stock #000090 | |
| 0.1% SDS | BioRad | 1610416 | |
| 0.5% acetic acid (glacial) | Fisher Chemical | BP1185 | Sirius Red Stain |
| 1mM EDTA | Sigma | E6758 | |
| 1.3% Picric acid | Sigma | P6744 | Sirius Red Stain |
| 10 mM sodium citrate, pH6.0 | Sigma | C9999 | |
| 3% BSA | Sigma | A7906 | |
| 3,3-diaminobenzidine (DAB) | Vector Lab | SK-4100 | |
| 4% Paraformaldehyde | Electron Microscopy Sciences | 15710-S | |
| 4mM Sodium metabisulfite | Sigma | S9000 | |
| 5% skim milk | BioRad | 1706404 | |
| 5-0 Silk | Oasis | MV-682-V | Ureteral obstruction |
| 70% Isopropyl alcohol | Fisher Chemical | A459 | |
| 95% Ethanol | Decon Labs | 2701 | Removal of renal nerve |
| Anti-α-SMA antibody | Sigma | A5228 | |
| Anti-β-actin antibody | Sigma | A-5316 | |
| Anti-F4/80 antibody | Proteintech | 18705-1-AP | |
| Anti-Fibronectin antibody | Cedarlane | CL5495AP | |
| Anti-ICAM-1 antibody | Santa Cruz | SC-1511-R | |
| Anti-IL-1β antibody | Abcam | ab9722 | |
| Anti-IL6 antibody | Abcam | ab83339 | |
| Anti-Phospho-Smad3 antibody | Abcam | ab51451 | |
| Anti-PMN antibody | Accurate | AIAD51140 | |
| Anti-TGF-β antibody | Santa Cruz | SC-7892 | |
| Anti-TLR4 antibody | IMGENEX | IMG-579A | |
| Anti-TNF-α antibody | Abcam | ab9739 | |
| Anti-Tyrosine Hydroxylase antibody | Abcam | ab112 | |
| Autoclave | Tuttnauer | EZ9PLUS | |
| Autoclip | MikRon | 205016 | |
| Bouin’s Fixative | Polysciences | 16045-1 | |
| Coplin Jar | Grainger | 3WEF1 | |
| Cotton tip | Midline | MDS202055 | |
| Creatinine Assay Kit | BioAssay Systems | DICT-500 | |
| DC Temperature Controller | FHC | 40-90-8D | |
| Direct Red 80 | Sigma | 365548 | Sirius Red Stain |
| Filter paper | Whatman | 3030917 | Removal of renal nerve |
| FITC-conjugated sinistrin | MediBeacon | N/A | GFR analysis |
| Heparinized capillary tube | Fisher Scientific | 22-260-950 | |
| Heparinized tube | Terumo Medical Corp. | Capiject | |
| HRP-conjugated anti-rabbit antibody | Vector Lab | PI-1000 | |
| Insulin syringe | Becton Dickinson | 305500 | |
| Ketamine | Par Pharmaceutical | Ketalar | Anesthetic agent |
| Lab Works analysis software | Ultra-Violet Products | N/A | Analysis of Western blot band density |
| Light microscope | Leica | Leica DMR | |
| Metabolic cage | Tecniplast | 3600M021 | GFR analysis |
| Microaneurysm clamp | Roboz | RS-5422 | Ischemia/reperfusion |
| Microdissecting forcep | Roboz | RS-5069 | |
| Microplate reader | Tecan | Infinite 200 PRO | |
| Mounting medium | Fisher Scientific | SP15-100 | |
| Noepinephrine ELISA kit | ALPCO Diagnostics | 17-NORHU-E01.1 | |
| PAGE gel of Any KD | BioRad | 456-9034 | |
| Phosphatase inhibitor | Sigma | P5726 | |
| Povidon-Iodine Prep Pad | Professional Disposables International | C12400 | |
| Protease | Calbiochem | 539134 | |
| Protein lysis buffer | Thermo Scientific | 78510 | |
| PVDF membrane | BioRad | 162-0176 | |
| Scalpel Handle | Roboz | RS-9843 | |
| Scissors | Roboz | RS-5882 | |
| Surgical blade | Bard-Parker | 371110 | |
| Surgical microscope | Nikon | SMZ-745 | |
| Superblock | Thermo Scientific | 37535 | |
| Transcutaneous Measurement System | MediBeacon | N/A | GFR analysis |
| Tris-Glycine buffer | BioRad | 1610771 | |
| Tris-Glycine-SDS buffer | BioRad | 1610744 | |
| TUNEL assay kit | Roche | 11684795910 | |
| Tweezers | Roboz | RS-5137 | |
| Western Lightning Chemiluminescence Substrate solution | PerkinElmer | NEL10400 | |
| Xylazine | Akorn Animal health | 139-236 | Anesthetic agent |
| Xylene | HistoPrep | HC700 |
References
- Grams, M. E., et al. Predicting timing of clinical outcomes in patients with chronic kidney disease and severely decreased glomerular filtration rate. Kidney International. 93 (6), 1442-1451 (2018).
- Coca, S. G., Singanamala, S., Parikh, C. R. Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney International. 81 (5), 442-448 (2012).
- Hsu, R. K., Hsu, C. Y. The Role of Acute Kidney Injury in Chronic Kidney Disease. Seminars in Nephrology. 36 (4), 283-292 (2016).
- Okusa, M. D., Rosin, D. L., Tracey, K. J. Targeting neural reflex circuits in immunity to treat kidney disease. Nature Reviews in Nephrology. 13 (11), 669-680 (2017).
- Kanagy, N. L. Alpha(2)-adrenergic receptor signalling in hypertension. Clinical Science (London). 109 (2), 431-437 (2005).
- Converse, R. L., et al. Sympathetic overactivity in patients with chronic renal failure. New England Journal of Medicine. 327 (27), 1912-1918 (1992).
- Veelken, R., et al. Autonomic renal denervation ameliorates experimental glomerulonephritis. Journal of American Society of Nephrology. 19 (7), 1371-1378 (2008).
- Veelken, R., Schmieder, R. E. Renal denervation--implications for chronic kidney disease. Nature Reviews Nephrology. 10 (6), 305-313 (2014).
- Wei, F. F., Zhang, Z. Y., Huang, Q. F., Staessen, J. A. Diagnosis and management of resistant hypertension: state of the art. Nature Reviews Nephrology. 14 (7), 428-441 (2018).
- Wyatt, C. M., Textor, S. C. Emerging evidence on renal denervation for the treatment of hypertension. Kidney International. 94 (4), 644-646 (2018).
- Bhatt, D. L., et al. A controlled trial of renal denervation for resistant hypertension. New England Journal of Medicine. 370 (15), 1393-1401 (2014).
- Jalil, F., White, W. B. A New Era of Renal Denervation Trials for Patients With Hypertension. American Journal of Kidney Diseases. 71 (5), 615-618 (2018).
- Amann, K., et al. Glomerulosclerosis and progression: effect of subantihypertensive doses of alpha and beta blockers. Kidney International. 60 (4), 1309-1323 (2001).
- Amann, K., et al. Effects of low dose sympathetic inhibition on glomerulosclerosis and albuminuria in subtotally nephrectomized rats. Journal of American Society of Nephrology. 11 (8), 1469-1478 (2000).
- Kim, J., Padanilam, B. J. Renal nerves drive interstitial fibrogenesis in obstructive nephropathy. Journal of American Society of Nephrology. 24 (2), 229-242 (2013).
- Kim, J., Padanilam, B. J. Renal denervation prevents long-term sequelae of ischemic renal injury. Kidney International. 87 (2), 350-358 (2015).
- Eriguchi, M., et al. Renal denervation has blood pressure-independent protective effects on kidney and heart in a rat model of chronic kidney disease. Kidney International. 87 (1), 116-127 (2015).
- Jang, H. S., Padanilam, B. J. Simultaneous deletion of Bax and Bak is required to prevent apoptosis and interstitial fibrosis in obstructive nephropathy. American Journal of Physiology- Renal Physiology. 309 (6), 540-550 (2015).
- Jang, H. S., Kim, J., Park, Y. K., Park, K. M. Infiltrated macrophages contribute to recovery after ischemic injury but not to ischemic preconditioning in kidneys. Transplantation. 85 (3), 447-455 (2008).
- Han, S. J., Jang, H. S., Kim, J. I., Lipschutz, J. H., Park, K. M. Unilateral nephrectomy elongates primary cilia in the remaining kidney via reactive oxygen species. Science Reports. 6, 22281 (2016).
- Scarfe, L., et al. Transdermal Measurement of Glomerular Filtration Rate in Mice. Journal of Visualized Experiments. (140), e58520 (2018).
- Jang, H. S., Kim, J. I., Han, S. J., Park, K. M. Recruitment and subsequent proliferation of bone marrow-derived cells in the postischemic kidney are important to the progression of fibrosis. American Journal of Physiology-Renal Physiology. 306 (12), 1451-1461 (2014).
- Clayton, S. C., Haack, K. K., Zucker, I. H. Renal denervation modulates angiotensin receptor expression in the renal cortex of rabbits with chronic heart failure. American Journal of Physiology-Renal Physiology. 300 (1), 31-39 (2011).
- Mulder, J., Hokfelt, T., Knuepfer, M. M., Kopp, U. C. Renal sensory and sympathetic nerves reinnervate the kidney in a similar time-dependent fashion after renal denervation in rats. Am Journal of Physiology - Regulatory, Integrative and Comparative Physiology. 304 (8), 675-682 (2013).
- Weihe, E., Depboylu, C., Schutz, B., Schafer, M. K., Eiden, L. E. Three types of tyrosine hydroxylase-positive CNS neurons distinguished by dopa decarboxylase and VMAT2 co-expression. Cell and Molecular Neurobiology. 26 (4-6), 659-678 (2006).
- Foss, J. D., Wainford, R. D., Engeland, W. C., Fink, G. D., Osborn, J. W. A novel method of selective ablation of afferent renal nerves by periaxonal application of capsaicin. American Journal of Physiology - Regulatory, Integrative and Comparative Physiology. 308 (2), 112-122 (2015).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved