14.3 : Homogeneous Equilibria for Gaseous Reactions
Homogeneous Equilibria for Gaseous Reactions
For gas-phase reactions, the equilibrium constant may be expressed in terms of either the molar concentrations (Kc) or partial pressures (Kp) of the reactants and products. A relation between these two K values may be simply derived from the ideal gas equation and the definition of molarity. According to the ideal gas equation:
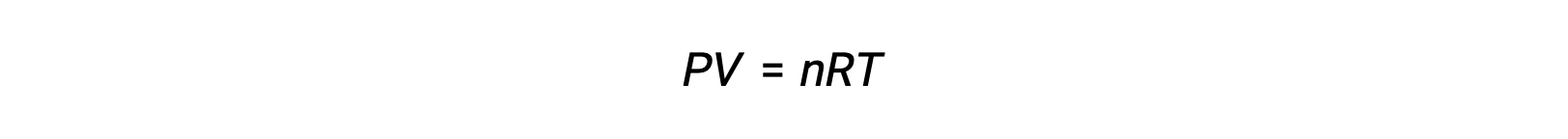
Molar concentration or molarity is given by number of moles divided by the volume:
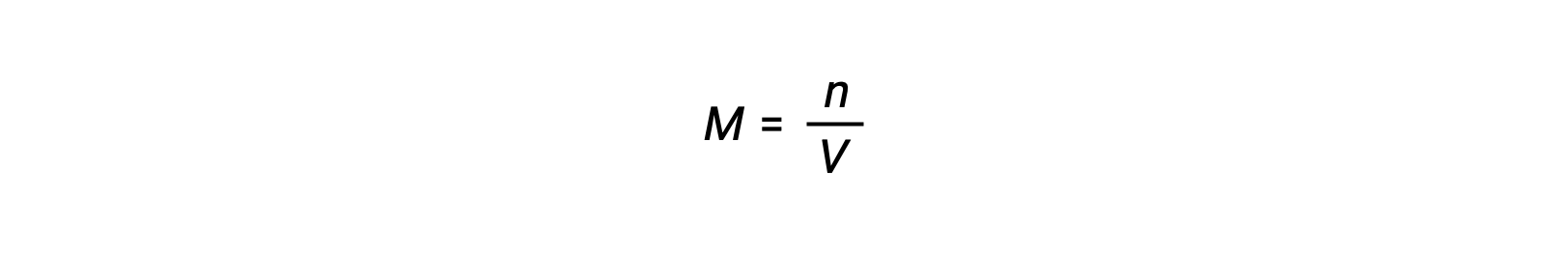
Thus,
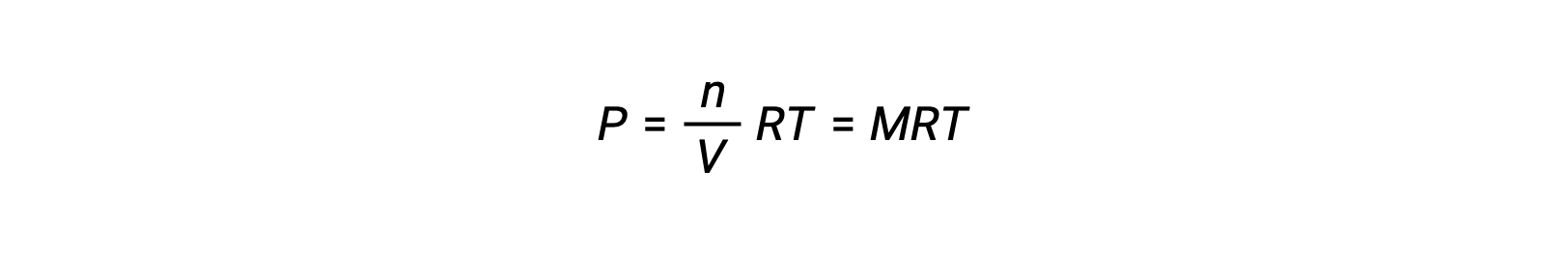
where P is partial pressure, V is volume, n is number of moles, R is the gas constant, T is temperature, and M is molar concentration.
For the gas-phase reaction: m A + n B ⇌ x C + y D
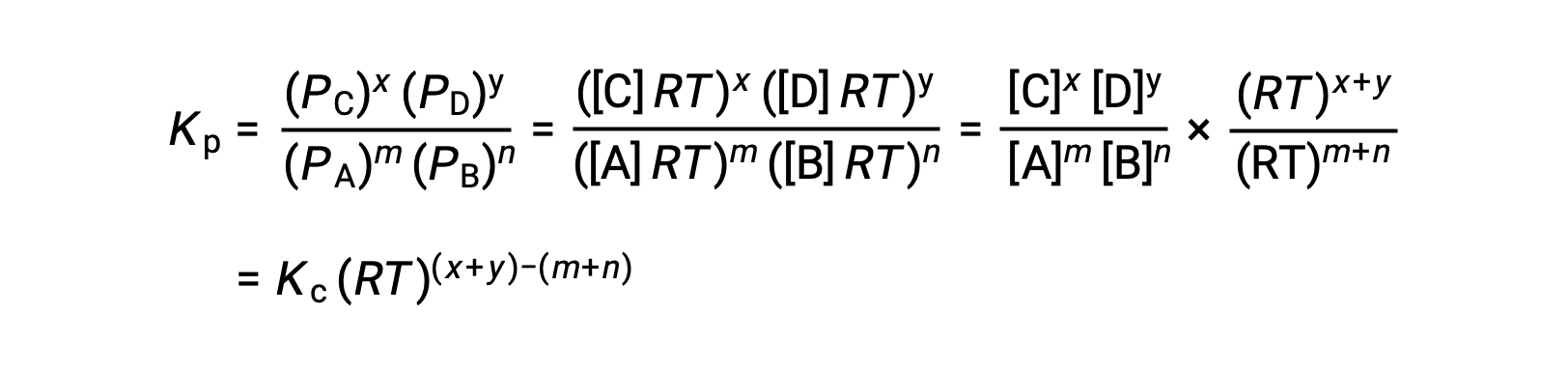
And so, the relationship between Kc and KP is
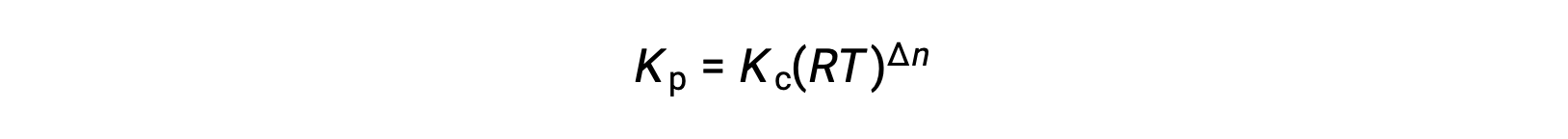
where Δn is the difference in the molar amounts of product and reactant gases, in this case:
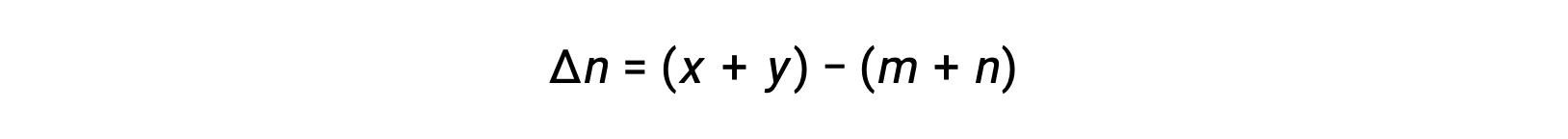
This text has been adapted from Openstax, Chemistry 2e, Section 13.2 Equilibrium Constants.
From Chapter 14:

Now Playing
14.3 : Homogeneous Equilibria for Gaseous Reactions
Chemical Equilibrium
21.8K Views

14.1 : Dynamic Equilibrium
Chemical Equilibrium
44.3K Views

14.2 : The Equilibrium Constant
Chemical Equilibrium
41.7K Views

14.4 : Calculating the Equilibrium Constant
Chemical Equilibrium
26.8K Views

14.5 : Reaction Quotient
Chemical Equilibrium
45.1K Views

14.6 : Calculating Equilibrium Concentrations
Chemical Equilibrium
42.3K Views

14.7 : Le Chatelier's Principle: Changing Concentration
Chemical Equilibrium
52.3K Views

14.8 : Le Chatelier's Principle: Changing Volume (Pressure)
Chemical Equilibrium
31.0K Views

14.9 : Le Chatelier's Principle: Changing Temperature
Chemical Equilibrium
26.5K Views

14.10 : The Small x Assumption
Chemical Equilibrium
44.0K Views
ABOUT JoVE
Copyright © 2025 MyJoVE Corporation. All rights reserved