Reduction in Left Ventricular Wall Stress and Improvement in Function in Failing Hearts using Algisyl-LVR
In This Article
Summary
This article describes procedures for implanting a novel hydrogel in failing hearts and quantifying its effect on left ventricular wall stress and function. These procedures have been successfully applied in dogs and humans.
Abstract
Injection of Algisyl-LVR, a treatment under clinical development, is intended to treat patients with dilated cardiomyopathy. This treatment was recently used for the first time in patients who had symptomatic heart failure. In all patients, cardiac function of the left ventricle (LV) improved significantly, as manifested by consistent reduction of the LV volume and wall stress. Here we describe this novel treatment procedure and the methods used to quantify its effects on LV wall stress and function.
Algisyl-LVR is a biopolymer gel consisting of Na+-Alginate and Ca2+-Alginate. The treatment procedure was carried out by mixing these two components and then combining them into one syringe for intramyocardial injections. This mixture was injected at 10 to 19 locations mid-way between the base and apex of the LV free wall in patients.
Magnetic resonance imaging (MRI), together with mathematical modeling, was used to quantify the effects of this treatment in patients before treatment and at various time points during recovery. The epicardial and endocardial surfaces were first digitized from the MR images to reconstruct the LV geometry at end-systole and at end-diastole. Left ventricular cavity volumes were then measured from these reconstructed surfaces.
Mathematical models of the LV were created from these MRI-reconstructed surfaces to calculate regional myofiber stress. Each LV model was constructed so that 1) it deforms according to a previously validated stress-strain relationship of the myocardium, and 2) the predicted LV cavity volume from these models matches the corresponding MRI-measured volume at end-diastole and end-systole. Diastolic filling was simulated by loading the LV endocardial surface with a prescribed end-diastolic pressure. Systolic contraction was simulated by concurrently loading the endocardial surface with a prescribed end-systolic pressure and adding active contraction in the myofiber direction. Regional myofiber stress at end-diastole and end-systole was computed from the deformed LV based on the stress-strain relationship.
Introduction
Reduction of ventricular wall stress is considered a cornerstone in the treatment of heart failure1. In its simplest form given by Laplace's law, ventricular wall stress is directly proportional to the diameter of the ventricle and the ventricular pressure, and is inversely proportional to the wall thickness of the ventricle. It is widely believed that increased ventricular wall stress is responsible for the adverse remodeling process in which the ventricles progressively become dilated, eventually leading to heart failure2. Clinical and animal studies have shown that increased wall stress induces changes in proteins, contractile element synthesis and gene expression that support the remodeling process3,4,5. Increased wall stress has also been shown to be an independent predictor of subsequent LV remodeling6,7.
Many new surgical treatments and devices have been developed with a central aim of reducing ventricular wall stress in a bid to prevent and reverse the progression of heart failure in patients8,9,10. Though these treatments share the same aim, they achieve it differently. For example, the surgical ventricular reduction procedure10 seeks to reduce ventricular wall stress by surgically reducing the size of a dilated left ventricle, but its result is a subject of controversy11,12.
Recently, the injection of a biocompatible material, Algisyl-LVR, into the left ventricle as a treatment for dilated cardiomyopathy has garnered considerable attention in the medical community. This treatment has been shown to be effective in preventing or even reversing the progression of heart failure in animal studies13,14 and, most recently, in a human clinical trial15. Contrary to other devices, this treatment seeks to reduce the ventricular wall stress by injecting material into the left ventricular wall to thicken it.
Detailed knowledge of ventricular wall stress, particularly in humans, however, remains elusive. This lack of knowledge is primarily because forces or stresses cannot be measured directly in the intact ventricles16. Though closed-form analytical equations such as Laplace's law can estimate left ventricular wall stress, they were developed based on restrictive assumptions which include axis-symmetry of the LV, material isotropy and homogeneity within the LV. Due to these factors, the prediction of ventricular wall stress in actual LV using Laplace's law is inaccurate17. To remove these restrictions and to obtain more accurate prediction of the ventricular wall stress, mathematical modeling using the finite element (FE) method with patient-specific ventricular geometry should be used in place of the simplified Laplace's law17.
The FE method is a numerical technique that is frequently used to solve a set of partial differential equations (PDE) describing a boundary value problem. This method is particularly useful when a closed form solution is difficult or cannot be obtained analytically. In the context of a mathematical LV model used to quantify ventricular wall stress, the set of PDEs are the governing equations of mechanical equilibrium (balance of linear momentum) that describe the LV motion when pressure or load is applied at the LV's endocardial surface. When the FE method is used, the LV wall is split into inter-connected sub-domains or elements (usually hexahedron with 8 corner nodes) that deform according to a prescribed stress-strain relationship of the myocardium.
Stress-strain relationships describing large deformation of the LV during passive filling in diastole and during active contraction in systole have been previously validated in large animal studies. The LV is modeled to be approximately three times stiffer in the myofiber direction than in directions perpendicular to the myofiber direction during diastole18. Active contraction during systole is modeled by increasing the stiffness of the LV along the myofiber direction. This increase in stiffness is a function of time and depends on experimentally determined variables such as the intracellular calcium concentration and the sarcomere length19.
Using this prescribed stress-strain relationship of the myocardium, the FE method calculates the new nodal positions based on load(s) applied to the LV. Once the new nodal positions are calculated, the resultant strain (a measure of deformation) and stress can be determined in each element to produce the strain and stress distribution within the LV.
Here, we outline the steps required to implant Algisyl-LVR in patients and to create the corresponding patient-specific LV mathematical models before and after treatment to quantify the LV wall stress.
Protocol
1. Algisyl-LVR Implant Procedure (Refer to video)
- Algisyl-LVR (LoneStar Heart, Inc. Laguna Hills, CA) is a Calcium-Alginate hydrogel consisting of two components. The Na+-Alginate component is a sterile aqueous solution with 4.6% mannitol and the Ca2+-Alginate component consists of water insoluble particles suspended in a sterile 4.6% mannitol solution (00:27).
- The procedure for implant placement can be performed using either a standard sternotomy or a small limited anterior thoracotomy on the beating heart. Cardiopulmonary bypass is not needed for the procedure.
- Right before use, mix the Na+-Alginate component and the Ca+-Alginate component by combining these two components into one syringe for intramyocardial injections (00:33).
- After 2 minutes, the two different alginate types will cross-link and form a gel that is ready for implant and placement in the myocardium via an injection.
- Identify the free wall of the LV at the mid-ventricular level halfway between the LV apex and base (00:44)
- Starting from the antero-septal groove at the mid-ventricular level, insert the needle at an approximately 45-degree angle and inject 0.3cc of Algisyl-LVR slowly (0.1 ml per second) in one uninterrupted motion (01:15).
- Repeat the injection (step 6) at 10 to 19 sites in a single line (circumferentially) along the mid-ventricular level, starting from the antero-septal groove and ending at the postero-septal groove. The number of implants is dictated by the size of the ventricle, spacing the implants approximately 1 cm apart.
2. Quantification of Left Ventricular Stress Using Mathematical Modeling
- Before using mathematical modeling to quantify LV wall stress, one must have already obtained the short axis and long axis view of the magnetic resonance images (MRI) containing the LV of the patient. Acquisition of these images can be performed using standard MRI protocol (e.g. Zhang et al.20).
- Digitize the endocardial surface and epicardial surface of the LV from the MR images containing the short-axis (SA) view of the LV. This can be done using the Contour Segmentation Object (CSO) library found in the freely-available software MevisLab.
- In our laboratory, we have created a program based on the modules found in the CSO library of MevisLab so that one simply "contours" the endocardial and epicardial boundary found in the SA view of the MR images containing the LV. Points of the epicardium and the endocardium in real three-dimensional (3D) space are then automatically generated from these contours.
- Import the 3D points from step 2 into a commercial software, Rapidform (INUS Technology, Inc, Sunnyvale, CA), to create surfaces of the LV epicardium and the endocardium in the Initial Graphics Exchange Specification (IGES) format. The steps used in creating these IGES surfaces in Rapidform are:
- Insert/Import.
- Create polygon mesh.
- Output IGES surfaces.
- Import the IGES surfaces into the commercial software TrueGrid to create an FE mesh of the LV.
- Fill the space between the endocardial and epicardial surface with the eight-node trilinear brick element. In general, a mesh containing about 3,000 elements with 3 elements through the wall thickness is sufficient to model the LV21.
- Once this is completed, export the mesh as an input deck for the FE solver LS-DYNA (LSTC, Livermore, CA). More details of this process can be found in Guccione et al.23.
- Assign the myofiber directions using our in-house software "Closer" that modifies the input deck exported from TrueGrid. Closer assigns the myofiber direction in each element as a vector that is parallel to the local epicardial tangent plane. This vector is oriented at an angle measured with respect to the local circumferential direction. In human LV, this angle is set to vary linearly across the wall thickness from -60 ° at the epicardium to 60 ° at the endocardium23.
- Write the boundary conditions and assign the myocardial material model to the elements in the input deck from step 5.
- Impose nodal displacements at the LV base with the keyword "SPC" in LS-DYNA. The nodes in the epicardial-basal ring are fixed and the rest of the nodes at the LV base are constrained to move only on the basal plane.
- Assign a constitutive law or stress-strain relationship described earlier (see "Introduction") to all the elements using the keyword "MAT" with material identity 128 in LS-DYNA.
- Define the elemental surfaces making up the endocardium and impose pressure boundary conditions with the keyword "LOAD_SEGMENTS".
- Define a pressure-time load curve using the keyword "DEFINE_CURVE".
- To simulate end of diastole, prescribe a pressure that increases rapidly with time to a prescribed end-diastolic pressure (EDP) of 20mmHg. The pressure is kept constant at EDP and sufficient time is then allowed for the LV to reach steady-state.
- To simulate end of systole, prescribe a pressure that increases rapidly with time from the end-diastolic state until a prescribed end-systolic pressure (ESP) of 125mmHg is achieved. The pressure is kept constant at ESP and sufficient time is then allowed for the LV to reach steady-state.
- Import the completed input deck into the commercial FE solver LS-DYNA to compute the ventricular wall stresses and the LV cavity volume at end of diastole and at end of systole.
- Adjust the material parameters reflecting the passive stiffness and the contractility of the myocardium until the computed LV cavity volume matches the MRI-measured volume at end of diastole and end of systole.
Representative Results
Injection of Algisyl-LVR into the LV free wall thickens it and continuously reduces the size of the LV over time. The thickening of the LV wall and the reduction in size of the LV is evident in the MRI of the LV in a patient at end of systole, before and 6 months after receiving Algisyl-LVR (Figure 1).
Figure 2 shows the outcome of each step involved in the quantification of the left ventricular wall stress. In Figure 2a, the epicardial and the endocardial edges were identified from a short-axis view of the LV in the MRI and contoured using MevisLab. The resultant IGES endocardial surface (blue) and the epicardial surface (red) created from Rapidform using the contoured points are shown in Figure 2b. Following this, the space between the endocardial and epicardial surfaces was filled with 8-noded trilinear brick element using TrueGrid (Figure 2c). Vectors defining the myofiber direction in each element were then computed with Closer. These vectors are shown as black arrows in Figure 2d and a slice of the LV wall (blue) shows the variation of myofiber direction across the LV wall as described earlier. In Figure 2e, the boundary conditions, namely, the pressure and the nodal displacement imposed on the LV are shown. The applied pressure is shown as arrows pointing towards the endocardial wall. Nodes at the epicardial-basal ring (shown as spheres) were constrained from moving in all directions whereas the rest of the basal nodes (shown as cubes) were constrained to move only in the basal plane (Figure 2e). Lastly, Figure 2f shows the computed ventricular wall stress in the myofiber direction at end of diastole from a patient before treatment. It is obvious from the figure that elevated stresses are found at the endocardium and at regions where the LV wall is thin.
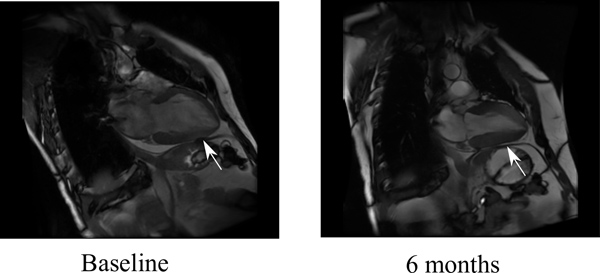
Figure 1. Effects of Algisyl on a patient's LV (indicated by arrow) after 6 months, as seen on magnetic resonance images. The images show that the LV has shrunk and the wall thickness has increased after 6 months.
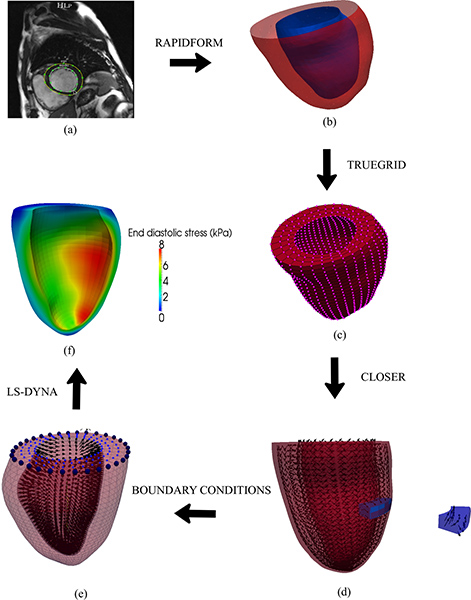
Figure 2. Steps involved in the quantification of left ventricular wall stress. (a) Digitizing the MR images. (b) Creating IGES surfaces. (c) Creating the FE mesh. (d) Assigning the myofiber orientation. (e) Imposing the boundary conditions. (f) Calculating the ventricular wall stress (shown here at end of diastole). Refer to text for explanation.
Discussion
Algisyl-LVR injection therapy
Injection of material into the LV free wall to reduce ventricular wall stress is a novel treatment designed for patients with dilated cardiomyopathy. This treatment has shown great promise in both preclinical and clinical studies 15. A randomized, controlled study to evaluate this treatment as a method of LV augmentation for patients with severe heart failure (AUGMENT-HF) has been underway since February 2012.
Multiple iterations of the product are being developed that target different physician segments and clinical needs. In the surgical product version for cardiothoracic surgeons, the alginate is delivered in a standard syringe and custom needle system designed for surgeons to perform the injections through a small surgical incision in the chest (minimal thoracotomy). The implant procedure is performed on a beating heart. The physical properties of the alginate hydrogel upon injection into the myocardium are similar to that of the diastolic myocardium, and become a permanent implant. The duration of the total operative procedure is expected to be less than 60 minutes in most cases, limiting exposure of the patient to a minimal anesthesia time. A second product version may offer patients with heart failure a procedure that can be performed by interventional cardiologists and, in some isolated cases, other specialists in a noninvasive or hybrid cardiology laboratory. It also would allow acute effects to be studied.
Quantification of left ventricular stress using mathematical modeling
The method of using mathematical modeling with the FE method is currently the only way to precisely quantify in-vivo regional wall stress in the ventricles. Combining mathematical modeling with medical imaging such as MRI enables one to compute in-vivo regional wall stress in patient-specific ventricles so as to help understand the functional state of these ventricles and quantify the mechanical effects of the injection treatment in patients.
Though we have treated the LV as a homogeneous material here, this method can be (and has been) extended to quantify in-vivo ventricular wall stress in non-homogeneous ventricles, particularly, when myocardial infarction is present. In such cases, the boundaries of the infarct and its adjacent borderzone have to be identified from MRI using gadolinium as a contrast agent. These boundaries are imported into TrueGrid to create elements that purely reside within each distinct region, namely, the infarct, the borderzone and the remote region. Material parameters reflecting pathological changes in each region can be assigned through the respective elements in LS-DYNA. These parameters have been found in a patient with a myocardial infarction using in-vivo myocardial strain measured from tagged MRI 21. Patients who require surgical revascularization will often experience atrial fibrillation during the post-operative period, which is associated with very poor quality tagged MRI data. Such patients also require a few days to recuperate from surgery. Thus, 3D echocardiography and speckle-tracking may be a more appropriate imaging modality and myocardial strain measurement technique than tagged MRI to study acute effects of surgical procedures.
Lastly, we used the commercial software Rapidform, Truegrid and LS-DYNA in the process of generating patient-specific mathematical models of the ventricles because we have found them to be generally efficient in accomplishing their respective tasks. However, other software is available, such as Cubit (for generating FE mesh) and Abaqus (a FE solver) that may also be suitable for creating mathematical models of the ventricles.
Disclosures
Mr. Hinson is an employee of LoneStar Heart, Inc.
Acknowledgements
This study was supported by National Heart, Lung, and Blood Institute Grants R01-HL-77921 and -86400 (to J. M. Guccione).
Materials
| Name | Company | Catalog Number | Comments |
| REGENTS | |||
| Na+-Alginate | LoneStar Heart, Inc | ||
| Ca2+-Alginate | LoneStar Heart, Inc | ||
| EQUIPMENT | |||
| MevisLab | Mevis Medical Solution | ||
| TrueGrid | XYZ Scientific Application, Inc | ||
| Rapidform | Inus Technology, Inc | ||
| LS-Dyna | Livermore Software Technology Corporation |
References
- Yin, F. C. Ventricular wall stress. Circ. Res. 49 (4), 829-842 (1981).
- Grossman, W. Cardiac hypertrophy: useful adaptation or pathologic process. Am. J. Med. 69 (4), 576-584 (1980).
- Swynghedauw, B. Development and functional adaptation of contractile proteins in cardiac and skeletal muscles. Physiol. Rev. 66 (3), 710-771 (1986).
- Komuro, I., Kurabayashi, M., Takaku, F., Yazaki, Y. Expression ofcellular oncogenes in the myocardium during the developmental stage and pressure-overloaded hypertrophy of the rat heart. Circ. Res. 62 (6), 1075-1079 (1988).
- Schunkert, H., Dzau, V. J., Tang, S. S., Hirsch, A. T., Apstein, C. S., Lorell, B. H. Increased rat cardiac angiotensin converting enzyme activity and mRNA expression in pressure overload left ventricular hypertrophy: effect on coronary resistance, contractility, and relaxation. J. Clin. Invest. 86 (6), 913-920 (1990).
- Aikawa, Y., Rohde, L., Plehn, J., Greaves, S. C., Menapace, F., Arnold, M. O., Rouleau, J. L., Pfeffer, M. A., Lee, R. T., Solomon, S. D. Regional wall stress predicts ventricular remodeling after anteroseptal myocardial infarction in the Healing and Early Afterload Reducing Trial (HEART): an echocardiography-based structural analysis. Am. Heart J. 141 (2), 234-242 (2001).
- Hung, C. L., Verman, A., Uno, H., Shin, S. H., Bourgoun, M., Hassanein, A. H., McMurray, J. J., Velazquez, E. J., Kober, L., Pfeffer, M. A., Solomon, S. D. VALIANT investigators. Longitudinal and Circumferential Strain Rate, Left Ventricular Remodeling, and Prognosis After Myocardial Infarction. J. Am. Coll. Cardiol. 56 (22), 1812-1822 (2010).
- Mann, D. L., Acker, M. A., Jessup, M., Sabbah, H. N., Starling, R. C., Kubo, S. H. Clinical evaluation of the CorCap Cardiac support device in patients with dilated cardiomyopathy. Ann. Thorac. Surg. 84 (4), 1226-1235 (2007).
- Grossi, E. A., Patel, N., Woo, J. Y., Goldberg, J. D., Schwartz, C. F., Subramanian, V., Feldman, T., Bourge, R., Baumgartner, N., Genco, C., et al. for the RESTOR-MV Study Group. Outcomes of the RESTOR-MV trial (randomized evaluation of a surgical treatment for off-pump repair of the mitral valve). J. Am. Coll. Cardiol. 56 (24), 1984-1993 (2010).
- Athanasuleas, C. L., Stanley, A. W. H., Buckberg, G. D. Restoration of contractile function in the enlarged left ventricle by exclusion of remodeled akinetic anterior segment: surgical strategy, myocardial protection and angiographic results. J. Card. Surg. 13 (6), 418-428 (1998).
- Jones, R. H., Velazquez, E. J., Michler, R. E., Sopko, G., Oh, J. K., O'Connor, C. M., Hill, J. A., Menicanti, L., Sadowski, Z., Desvigne-Nickens, P., Rouleau, J. L., Lee, K. L. STICH Hypothesis 2 Investigators. Coronary bypass surgery with or without surgical ventricular reconstruction. N. Engl. J. Med. 360 (17), 1705-1717 (2009).
- Buckberg, G. D., Athanasuleas, C. L., Wechsler, A. S., Beyersdorf, F., Conte, J. V., Strobeck, J. E. The STICH trial unravelled. Eur. J. Heart Fail. 12 (10), 1024-1027 (2010).
- Sabbah, H. N., Wang, M., Jiang, A., Ilsar, I., Sabbah, M. S., Helgerson, S., Peterson, R., Tarazona, N., Lee, R. Circumferential mid-ventricular intramyocardial injections of alginate hydrogel improve left ventricular function and prevent progressive remodeling in dogs with chronic heart failure. Circulation. 120, S912 .
- Yu, J., Christman, K. L., Chin, E., Sievers, R. E., Saeed, M., Lee, R. J. Restoration of left ventricular geometry and improvement of left ventricular function in a rodent model of chronic ischemic cardiomyopathy. J. Thorac. Cardiovasc. Surg. 137 (1), 180-187 (2009).
- Lee, R. J., Hinson, A., Helgerson, S., Bauerschmitt, R., Sabbah, H. N. Polymer-based restoration of left ventricular mechanics. Cell Transplant. 22 (3), 529-533 (2013).
- Huisman, R. M., Elzinga, G., Westerhof, N., Sipkema, P. Measurement of left ventricular wall stress. Cardiovasc. Res. 14 (3), 142-153 (1980).
- Zhang, Z., Tendulkar, A., Sun, K., Saloner, D. A., Wallace, A. W., Ge, L., Guccione, J. M., Ratcliffe, M. B. Comparison of the Young-Laplace law and finite element based calculation of ventricular wall stress: implications for post infarct and surgical ventricular remodeling. Ann. Thorac. Surg. 91 (1), 150-156 (2011).
- Guccione, J. M., McCulloch, A. D., Waldman, L. K. Passive material properties of intact ventricular myocardium determined from a cylindrical model. J. Biomech. Eng. 113 (1), 42-55 (1991).
- Guccione, J. M., Waldman, L. K., McCulloch, A. D. Mechanics of active contraction in cardiac muscle: Part II--Cylindrical models of the systolic left ventricle. J. Biomech. Eng. 115 (1), 82-90 (1993).
- Zhang, P., Guccione, J. M., Nicholas, S. I., Walker, J. C., Crawford, P. C., Shamal, A., Acevedo-Bolton, G., Guttman, M. A., Ozturk, C., McVeigh, E. R., Saloner, D. A., Wallace, A. W., Ratcliffe, M. B. Endoventricular patch plasty for dyskinetic anteroapical left ventricular aneurysm increases systolic circumferential shortening in sheep. J. Thorac. Cardiovasc. Surg. 134 (4), 1017-1024 (2007).
- Wenk, J. F., Klepach, D., Lee, L. C., Zhang, Z., Ge, L., Tseng, E. E., Martin, A., Kozerke, S., Gorman, J. H. 3. r. d., Gorman, R. C., Guccione, J. M. First evidence of depressed contractility in the border zone of a human myocardial infarction. Ann. Thorac. Surg. 93 (4), 1188-1193 (2012).
- Guccione, J. M., Kassab, G. S., Ratcliffe, M. B. . Computational cardiovascular mechanics: modeling and applications in heart failure. , (2010).
- Streeter, D. D., Spotnitz, H. M., Patel, D. P., Ross, J., Sonnenblick, E. H. Fiber Orientation in the Canine Left Ventricle during Diastole and Systole. Circ. Res. 24 (3), 339-347 (1969).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved