Abstract
Immunology and Infection
Aktivering og konjugering av løselige polysakkarider med 1-cyano-4-dimetylaminopyridin tetrafluoroborate (CDAP)
ERRATUM NOTICE
Important: There has been an erratum issued for this article. Read more …Konjugatvaksiner er bemerkelsesverdige fremskritt innen vaksinering. For fremstilling av polysakkaridkonjugatvaksiner kan polysakkaridene enkelt funksjonaliseres og knyttes til vaksinebærerproteiner ved hjelp av 1-cyano-4-dimetylaminopyridintetrafluoroborate (CDAP), et cyanylerende reagens som er lett å håndtere. CDAP aktiverer polysakkarider ved å reagere med karbohydrathydroksylgrupper ved pH 7-9. Stabiliteten og reaktiviteten til CDAP er svært pH-avhengig. pH-en til reaksjonen reduseres også under aktivering på grunn av hydrolysen til CDAP, noe som gjør at god pH kontrollerer nøkkelen til reproduserbar aktivering. Den opprinnelige CDAP-aktiveringsprotokollen ble utført ved romtemperatur i ubuffered pH 9-løsninger.
På grunn av den raske reaksjonen under denne tilstanden (<3 min) og det medfølgende raske pH-fallet fra den raske CDAP-hydrolysen, var det utfordrende å raskt justere og opprettholde målreaksjonen pH på kort tid. Den forbedrede protokollen som er beskrevet her, utføres ved 0 °C, noe som bremser CDAP-hydrolysen og forlenger aktiveringstiden fra 3 min til ~15 min. Dimetylaminopyridin (DMAP) ble også brukt som en buffer for å forhåndsjustere polysakkaridløsningen til målaktiverings-pH før du legger til CDAP-reagenset. Den lengre reaksjonstiden, kombinert med den langsommere CDAP-hydrolysen og bruken av DMAP-buffer, gjør det enklere å opprettholde aktiverings-pH for hele aktiveringsprosessens varighet. Den forbedrede protokollen gjør aktiveringsprosessen mindre frenetisk, mer reproduserbar og mer egnet til å skalere opp.
Erratum
Erratum: Activation and Conjugation of Soluble Polysaccharides using 1-Cyano-4-Dimethylaminopyridine Tetrafluoroborate (CDAP)An erratum was issued for: Activation and Conjugation of Soluble Polysaccharides using 1-Cyano-4-Dimethylaminopyridine Tetrafluoroborate (CDAP). A figure was updated.
Figure 4 was updated from:
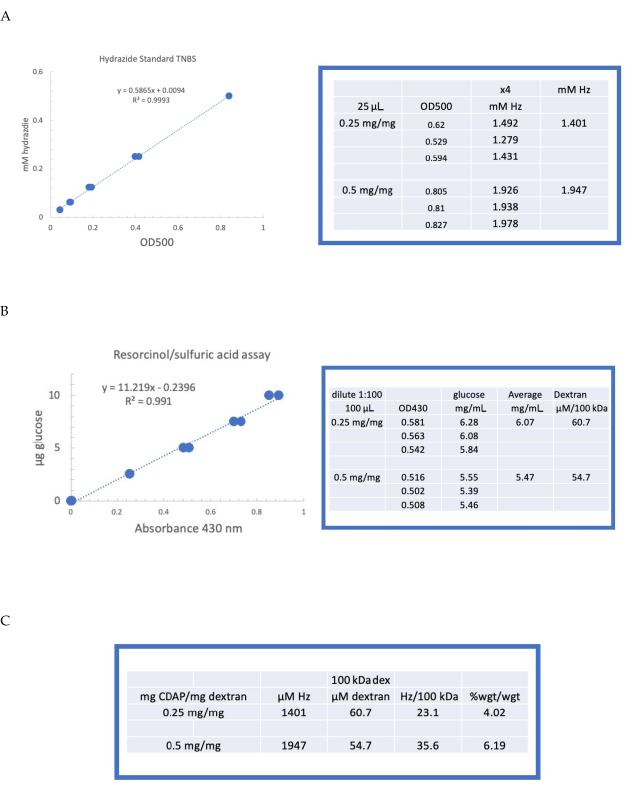
Figure 4: Representative results for CDAP activation of dextran. Typical standard curves for the (A) resorcinol/sulfuric acid and (B) TNBS assays. The assay results for dextran activated with 0.25 and 0.5 mg CDAP/mg dextran are shown. Glucose was used as the standard for the resorcinol assay. Dextran, in mg/mL, is divided by 100 kDa to give a molar concentration. The hydrazide concentration is determined using ADH as the standard and the results expressed as µM Hz. (C) Calculation of hydrazide: dextran ratios.The level of derivatization was calculated as hydrazides per 100 kDa of dextran to facilitate the comparison between polymers of different average molecular weights. The % weight ratio of g ADH/g dextran was calculated using a MW of 174 g/mole for ADH. Please click here to view a larger version of this figure.
to:
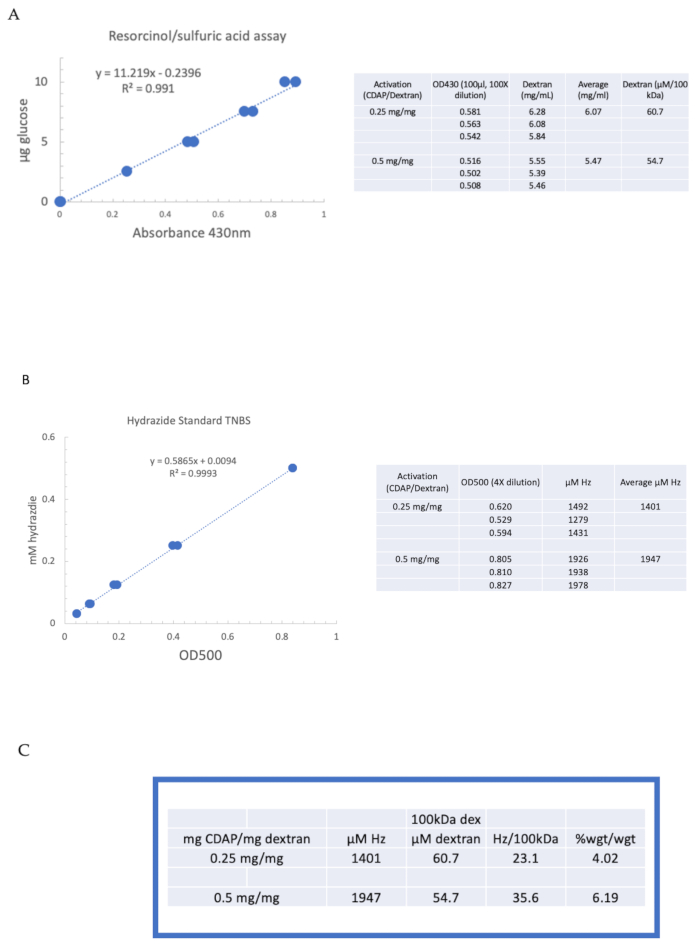
Figure 4: Representative results for CDAP activation of dextran. Typical standard curves for the (A) resorcinol/sulfuric acid and (B) TNBS assays. The assay results for dextran activated with 0.25 and 0.5 mg CDAP/mg dextran are shown. Glucose was used as the standard for the resorcinol assay. Dextran, in mg/mL, is divided by 100 kDa to give a molar concentration. The hydrazide concentration is determined using ADH as the standard and the results expressed as µM Hz. (C) Calculation of hydrazide: dextran ratios.The level of derivatization was calculated as hydrazides per 100 kDa of dextran to facilitate the comparison between polymers of different average molecular weights. The % weight ratio of g ADH/g dextran was calculated using a MW of 174 g/mole for ADH. Please click here to view a larger version of this figure.
ABOUT JoVE
Copyright © 2024 MyJoVE Corporation. All rights reserved