Summary
Abstract
Introduction
Protocol
Representative Results
Discussion
Acknowledgements
Materials
References
Cancer Research
High-Content Screening Assay for the Identification of Antibody-Dependent Cellular Cytotoxicity Modifying Compounds
This protocol presents an automated, image-based high-throughput technique to identify compounds modulating natural killer cell-mediated breast cancer cell killing in the presence of a therapeutic anti-HER-2 antibody.
Immunotherapy with antigen-specific antibodies or immune checkpoint inhibitors has revolutionized the therapy of breast cancer. Breast cancer cells expressing the epidermal growth factor receptor HER2 can be targeted by the anti-HER-2 antibody trastuzumab. Antibody-dependent cellular cytotoxicity (ADCC) is an important mechanism implicated in the antitumor action of HER-2. Trastuzumab bound to cancer cells can be recognized by the Fc receptors of ADCC effector cells (e.g., natural killer (NK) cells, macrophages, and granulocytes), triggering the cytotoxic activity of these immune cells leading to cancer cell death. We set out to develop an image-based assay for the quantification of ADCC to identify novel ADCC modulator compounds by high-content screening. In the assay, HER2 overexpressing JIMT-1 breast cancer cells are co-cultured with NK-92 cells in the presence of trastuzumab, and target cell death is quantified by automated microscopy and quantitative image analysis. Target cells are distinguished from effector cells based on their EGFP fluorescence. We show how compound libraries can be tested in the assay to identify ADCC modulator drugs. For this purpose, a compound library test plate was set up using randomly selected fine chemicals off the lab shelf. Three microtubule destabilizing compounds (colchicine, vincristine, podophyllotoxin) expected to interfere with NK cell migration and degranulation were also included in the test library. The test screen identified all three positive control compounds as hits proving the suitability of the method to identify ADCC-modifying drugs in a chemical library. With this assay, compound library screens can be performed to identify ADCC-enhancing compounds that could be used as adjuvant therapeutic agents for the treatment of patients receiving anticancer immunotherapies. In addition, the method can also be used to identify any undesirable ADCC-inhibiting side effects of therapeutic drugs taken by cancer patients for different indications.
Immunotherapy with anticancer antibodies, immune checkpoint inhibitors, or chimeric antigen receptor-expressing T (CAR-T) cells represents a powerful approach to cancer treatment1,2,3. Trastuzumab is a humanized monoclonal anti-HER-2 (human epidermal growth factor receptor 2) antibody used for treating HER-2 positive early stage or metastatic breast cancer, as well as HER-2 positive metastatic gastric cancer4,5,6. It primarily acts by inhibiting the proliferation stimulating effect o....
NOTE: Key steps of the assay workflow are presented in Figure 2.
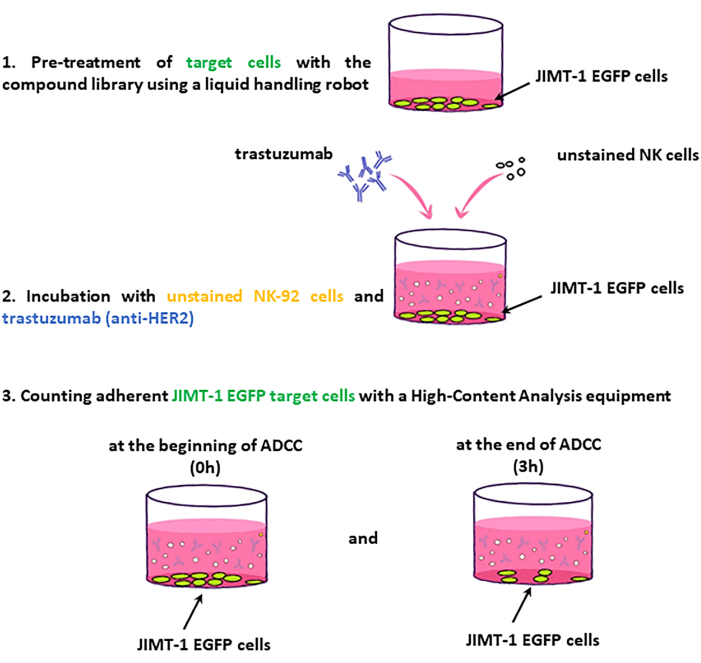
Figure 2: Workflow of the ADCC screen. JIMT-1-EGFP target cells seeded into 96 well HCS plates are treated with drugs of the compound library. In turn, unstained NK (effector) cells and trastuzumab are added, and the plate .......
To demonstrate how the assay works in real life, we created a test library of 16 compounds selected randomly from the lab shelves (Figure 3). In addition, DMSO was also included as a negative control, and three microtubule polymerization inhibitor compounds (colchicine, vincristine, and podophyllotoxin) as positive controls. The latter were expected to inhibit ADCC by interfering with NK cell migration to the cancer cells and NK cell degranulation. All test compounds and DMSO were placed ont.......
The ADCC reaction has been described a relatively long time ago. Key molecular events of the process have also been described19. Methods for measuring ADCC range from the gold standard radioactive chromium release assay, cytoplasmic enzyme release assays to several fluorescence-based flow cytometry or microplate assays20. However, a common limitation of these assays is that they are not amenable to high-throughput applications. Previously, we developed an image-based HCS as.......
LV received funding from National Research, Development and Innovation Office grants GINOP-2.3.2-15-2016-00010 TUMORDNS", GINOP-2.3.2-15-2016-00048-STAYALIVE and OTKA K132193, K147482. CD16.176V.NK-92 cells were obtained from Dr. Kerry S. Campbell (Fox Chase Center, Philapedlphia, PA, on behalf of Brink Biologics, lnc. San Diego, CA), are protected by patents worldwide, and were licensed by Nantkwest, lnc. Authors are thankful to György Vereb and Árpád Szöőr for their help with the use of the NK-92 cell line and for technical advice.
....| Name | Company | Catalog Number | Comments |
| 5-fluorouracil | Applichem | A7686 | in compound library |
| 96-well Cell Carrier Ultra plate | PerkinElmer | LLC 6055302 | |
| Betulin | Sigma | B9757 | in compound library |
| CD16.176V.NK92 cells | Nankwest Inc. | ||
| Cerulenin | ChemCruz | sc-396822 | in compound library |
| Cisplatin | Santa Cruz Biotechnology | sc-200896 | in compound library |
| Colchicine | Sigma | C9754 | in compound library |
| Concanavalin-A | Calbiochem | 234567 | in compound library |
| Dexamethasone | Sigma | D4902 | in compound library |
| DMEM/F-12 medium | Sigma | D8437 | in JIMT-1 EGFP medium |
| DMSO | Sigma | D2650 | in compound library |
| Etoposide | Sigma | E1383 | E1383 |
| Fetal bovine serum (FBS) | Biosera | FB-1090/500 | JIMT-1 EGFP and NK medium |
| Fisetin | Sigma | F4043 | in compound library |
| Freedom EVO liquid handling robot | TECAN | ||
| Gallotannin | Fluka Chemical Corp. | 16201 | in compound library |
| Glutamine | Gibco | 35,050–061 | in NK medium |
| Harmony software | PerkinElmer | ||
| Humanized anti-HER2 monoclonal antibody (Herzuma) | EGIS Pharmaceuticals, Budapest Hungary | N/A | |
| Humulin R (insulin) | Eli Lilly | HI0219 | JIMT-1 EGFP medium |
| IL-2 | Novartis Hungária Kft. | PHC0026 | in NK medium |
| Isatin | Sigma | 114618 | in compound library |
| MEM Non-essential Amino Acids (MEM-NEAA) | Gibco | 11,140–050 | in NK medium |
| Na-pyruvate | Lonza | BE13-115E | in NK medium |
| Naringenin | Sigma | N5893 | in compound library |
| NQDI-1 | Sigma | SML0185 | in compound library |
| Opera Phenix High-Content Analysis equipment | PerkinElmer | ||
| Penicillin–streptomycin | Biosera | LM-A4118 | JIMT-1 EGFP and NK medium |
| Pentoxyfilline | Sigma | P1784 | in compound library |
| Phosphate buffered saline (PBS) | Lonza | BE17-517Q | to wash the cells |
| Podophyllotoxin | Sigma | P4405 | in compound library |
| Quercetin | Sigma | Q4951 | in compound library |
| Tannic acid | Sigma | T8406 | in compound library |
| Temozolomide | Sigma | T2577 | in compound library |
| Trypan blue 0.4% solution | Sigma | T8154 | for cell counting |
| Vincristine sulfate | Sigma | V0400000 | in compound library |
| α-MEM | Sigma | M8042 | in NK medium |
- Gupta, S. L., Basu, S., Soni, V., Jaiswal, R. K. Immunotherapy: an alternative promising therapeutic approach against cancers. Molecular Biology Reports. 49 (10), 9903-9913 (2022).
- Moretti, A., et al. The past, present, and future of non-viral CAR T cells. Frontiers in Immunology. 13, 867013 (2022).
- June, C. H., O'Connor, R. S., Kawalekar, O. U., Ghassemi, S., Milone, M. C. CAR T cell immunotherapy for human cancer. Science. 359 (6382), 1361-1365 (2018).
- Ross, J. S., et al. The HER-2 receptor and breast cancer: ten years of targeted anti-HER-2 therapy and personalized medicine. Oncologist. 14 (4), 320-368 (2009).
- Shitara, K., et al. Discovery and development of trastuzumab deruxtecan and safety management for patients with HER2-positive gastric cancer. Gastric Cancer. 24 (4), 780-789 (2021).
- Gianni, L., et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncology. 13 (1), 25-32 (2012).
- Barok, M., et al. Trastuzumab causes antibody-dependent cellular cytotoxicity-mediated growth inhibition of submacroscopic JIMT-1 breast cancer xenografts despite intrinsic drug resistance. Molecular and Cancer Therapy. 6 (7), 2065-2072 (2007).
- Gauthier, M., Laroye, C., Bensoussan, D., Boura, C., Decot, V. Natural Killer cells and monoclonal antibodies: Two partners for successful antibody dependent cytotoxicity against tumor cells. Crit Rev Oncol Hematol. 160, 103261 (2021).
- Gruijs, M., Sewnath, C. A. N., van Egmond, M. Therapeutic exploitation of neutrophils to fight cancer. Semin Immunol. 57, 101581 (2021).
- Mando, P., Rivero, S. G., Rizzo, M. M., Pinkasz, M., Levy, E. M. Targeting ADCC: A different approach to HER2 breast cancer in the immunotherapy era. Breast. 60, 15-25 (2021).
- van der Haar Avila, I., Marmol, P., Kiessling, R., Pico de Coana, Y. Evaluating antibody-dependent cell-mediated cytotoxicity by chromium release assay. Methods in Molecular Biology. 1913, 167-179 (2019).
- Broussas, M., Broyer, L., Goetsch, L. Evaluation of antibody-dependent cell cytotoxicity using lactate dehydrogenase (LDH) measurement. Methods in Molecular Biology. 988, 305-317 (2013).
- Toth, G., Szollosi, J., Vereb, G. Quantitating ADCC against adherent cells: Impedance-based detection is superior to release, membrane permeability, or caspase activation assays in resolving antibody dose response. Cytometry A. 91 (10), 1021-1029 (2017).
- Chung, S., Nguyen, V., Lin, Y. L., Kamen, L., Song, A. Thaw-and-use target cells pre-labeled with calcein AM for antibody-dependent cell-mediated cytotoxicity assays. Journal of Immunological Methods. 447, 37-46 (2017).
- Lee-MacAry, A. E., et al. Development of a novel flow cytometric cell-mediated cytotoxicity assay using the fluorophores PKH-26 and TO-PRO-3 iodide. Journal of Immunological Methods. 252 (1-2), 83-92 (2001).
- Tanito, K., et al. Comparative evaluation of natural killer cell-mediated cell killing assay based on the leakage of an endogenous enzyme or a pre-loaded fluorophore. Analytical Science. 37 (11), 1571-1575 (2021).
- Lin, S., Schorpp, K., Rothenaigner, I., Hadian, K. Image-based high-content screening in drug discovery. Drug Discovery Today. 25 (8), 1348-1361 (2020).
- Guti, E., et al. The multitargeted receptor tyrosine kinase inhibitor sunitinib induces resistance of HER2 positive breast cancer cells to trastuzumab-mediated ADCC. Cancer Immunology, Immunotherapy. 71 (9), 2151-2168 (2022).
- Li, F., Liu, S. Focusing on NK cells and ADCC: A promising immunotherapy approach in targeted therapy for HER2-positive breast cancer. Frontiers in Immunology. 13, 1083462 (2022).
- Perussia, B., Loza, M. J. Assays for antibody-dependent cell-mediated cytotoxicity (ADCC) and reverse ADCC (redirected cytotoxicity) in human natural killer cells. Methods in Molecular Biology. 121, 179-192 (2000).
- Garcia-Alonso, S., Ocana, A., Pandiella, A. Trastuzumab emtansine: Mechanisms of action and resistance, clinical progress, and beyond. Trends in Cancer. 6 (2), 130-146 (2020).
ABOUT JoVE
Copyright © 2024 MyJoVE Corporation. All rights reserved