Imaging Ca2+ Signals in Small Pulmonary Veins at Physiological Intraluminal Pressures
In This Article
Summary
In this protocol, we present a novel technique for recording and analyzing Ca2+ signals in intrapulmonary veins (small pulmonary veins or PVs) at physiological intraluminal pressures. The technique involves isolating small PVs, incubating them with a Ca2+ indicator, cannulating and pressurizing them, confocal imaging of Ca2+ signals, and data analysis.
Abstract
Pulmonary veins (PVs) carry oxygen-rich blood from the lungs back to the left heart, thus serving an important function in the delivery of oxygen-rich blood to vital organs. However, most studies of pulmonary vasculature have focused on pulmonary arteries and capillaries under normal and disease conditions. Ca2+ signals are critical regulators of vascular function. Despite the critical physiological roles of PVs, Ca2+ signals in small intrapulmonary PVs have not been recorded under physiological conditions. Here, we describe a technique to record Ca2+ signal activity in mouse PVs isolated, cannulated and pressurized at 5 mmHg. By incorporating a Ca2+ indicator, we can study Ca2+ signals in the myocyte layer of small PVs using high-speed, spinning disk confocal imaging under physiological conditions. Our representative data indicates that the Ca2+ signals in small PV myocytes are mediated by openings of ryanodine receptor ion channels. This method will be of considerable interest to researchers in the field of pulmonary vascular physiology and disorders.
Introduction
The pulmonary vasculature plays an important role in exchanging gases, adding oxygen, and removing carbon dioxide from the blood1,2. Pulmonary arteries (PAs) receive oxygen-poor blood from the right heart. The gas exchange occurs at the level of capillaries (alveolar capillaries), and oxygen-rich blood is delivered to the left heart by pulmonary veins (PVs). Unlike other veins that carry oxygen-poor blood, PVs serve an important function of delivering oxygen-rich blood to the left heart and, consequently, to vital organs in the body. However, the signaling mechanisms that regulate PV function under normal and disease conditions are poorly understood.
Studies on pulmonary vasculature have mainly focused on PAs and pulmonary capillaries. Recent studies show that Ca2+ signals in endothelial and smooth muscle cell layers are crucial in regulating the function of PAs3,4,5. Furthermore, abnormal Ca2+ signaling mechanisms are responsible for the impaired function of PAs and pulmonary capillaries in disease conditions6,7,8,9,10. However, studies on intrapulmonary veins (small PVs) remain scarce. Ectopic activity in large PVs adjacent to the left heart (cardiac PVs) has been proposed as a source of atrial fibrillation in humans11,12, and spontaneous pulsation13 and Ca2+ signals12,14,15 have also been shown in large PVs. Small PVs deliver oxygen-rich blood to large PVs. However, Ca2+ signaling mechanisms in small PVs have not been studied under physiological intraluminal pressures.
In this manuscript, we introduce a new method for isolating, cannulating, and pressurizing small PVs and imaging Ca2+ signals in PV myocytes using high-speed spinning disk confocal microscopy. The overall goal of this method is to record spontaneous and induced Ca2+ signals in small PV myocytes under physiological intraluminal pressures. Intraluminal pressure plays a critical role in regulating Ca2+ signals in myocytes16. Typically, PVs are subjected to physiological intraluminal pressures ranging from 4 to 10 mm Hg17,18. Changes in intraluminal pressure throughout the cardiac cycle can influence Ca2+ signals in PV myocytes. Therefore, it is essential to employ a technique that allows us to study these signals under physiological conditions and examine how varying intraluminal pressures affect them. Additionally, most prior research on PVs has concentrated on larger cardiac PVs. The current technique focuses on small PVs located deep within the lungs, which may be more relevant for understanding the regulation of lung function in both health and disease.
Protocol
All animal protocols were approved by the University of Virginia Animal Care and Use Committee (protocols 4100 and 4120). C57BL6/J Mice, 10-12 weeks of age, are euthanized with pentobarbital (90 mg/kg; intraperitoneally), followed by cervical dislocation for harvesting lung tissue3,4,19,20.
1. Isolation of small PVs
- Clean the dissection tools and dishes with 100% ethanol, followed by washing with deionized water.
- Use scissors to open the thoracic cavity. Use forceps to carefully remove the heart and the lungs from the thoracic cavity with minimal touching of the lungs.
- Place the tissue on a Sylgard-coated plate containing cold (4-10 °C) HEPES-buffered physiological salt solution (HEPES-PSS, in mM, 10 HEPES, 134 NaCl, 6 KCl, 1 MgCl2 hexahydrate, 2 CaCl2 dihydrate, and 7 dextrose, pH adjusted to 7.4 using 1 M NaOH)3,4,19. Use dissection pins to pin down the heart and the lungs such that the large PVs and PAs are clearly visible and the left lobe of the lung is slightly stretched, as shown in Figure 1.
- Using large pulmonary veins (PVs) as reference points, carefully remove the tissue surrounding the small intrapulmonary veins (small PVs, approximately 50-100 μm in diameter, indicated by the dashed rectangle in Figure 1) with fine scissors. Make sure to avoid direct contact with the small PVs. Gently isolate the small PVs from the surrounding tissue.
- Isolate approximately 1-2 mm long segments of small PVs. Place small PVs in 4-10 °C HEPES-PSS.
2. Loading small PVs with a Ca2+ indicator
NOTE: Small PVs should be handled carefully, ensuring the forceps touch only the tips of the PVs. This method is a modification of the technique described previously3,7,20,21.
- Prepare a stock concentration of 2.5 mM Fluo-4 AM with DMSO3,4. Using the above stock solution, prepare 10 μM fluo-4 AM and 0.04% pluronic acid with HEPES-PSS.
- Place small PVs in a 1.5 mL tube with the loading solution (10 μM fluo-4 AM and 0.04% pluronic acid in HEPES-PSS)3,4. Cover the tube with aluminum foil and place in a water bath for incubation at 37 °C for 1 h.
- After the incubation, carefully replace the loading solution with 4-10 °C HEPES-PSSand keep on ice for 30 min as a washing step.
3. Cannulation of small PVs
NOTE: This method is a modification of the technique described previously3,20,21.
- Prepare the pressure myography chamber with two glass cannulas, one on each side. Fill the cannulas with PSS (119 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgCl2 160 hexahydrate, 2.5 mM CaCl2 dihydrate3,4,21,22, 7 mM dextrose, and 24 mM NaHCO3, maintained at pH 7.4 by bubbling the solution with 21% O2 and 5% CO2) using a 10 mL syringe.
- Remove a small PV from the washing solution and place it in the pressure myography chamber. Using fine-tipped forceps, carefully cannulate one end of the small PV onto one of the cannulas. Then, using a microfilament made from nylon thread, tie a knot around the cannulated end of the small PV and the tip of the cannula to secure it.
- Push the PSS gently through the cannula to remove the blood inside the PV. Use nylon thread to tie off the other end with a glass cannula using microfilaments, as shown in Figure 1.
4. Pressurizing and Ca2+ imaging of small PV
NOTE: This method is a modification of the technique described previously3,21.
- Using a servo pressure controller attached to a tubing containing PSS, pressurize the small PV from one end at 5 mm Hg. Use a peristaltic pump connected to both an inlet and an outlet, to superfuse the PSS. Maintain the temperature of the superfusate at 37 °C.
- After the equilibration period, acquire Ca2+ images for 1000 frames (frame rate: 30 frames per second) using the 40x water-dipping objective (NA 0.8) and a spinning disk confocal imaging system. Excite Fluo-4 using a 488 nm solid-state laser and capture emitted fluorescence using a 525/36 nm band-pass filter.
- Apply the ryanodine receptor (RyR) antagonist ryanodine (5 μM)21,23 to the superfusate. Wait for 5 min. Retake the Ca2+ images as described above, capturing another 1000 frames at a rate of 30 frames/s.
5. Ca2+ image analysis
NOTE: This method is a modification of the technique described previously21,22,24. Changes in the activity of Ca2+ signals in PV myocytes can provide important insights into the inhibition or enhancement of excitation-contraction coupling in PVs.
- Use custom-designed software (developed by Dr. Adrian Bonev, https://github.com/vesselman/SparkAn) to analyze Ca2+ images.
- Smooth images using 5 x 5 boxcar filter and 5 x 5 median filter. Use a frame outlining a flat region of PV with multiple cells for event autodetection.
- Click on View and Event Autodetection. Use the following settings: event threshold at an amplitude of 1.3 F/F0, Tolerance of 20%, scan box of 7 x 7 pixels, and running average of 7 images. Generate average images automatically every 10 images, starting from the first image.
- Click on Start Search (the eye icon). Find the events table saved as a Mark File under the File menu. To calculate events per µm², divide the number of detected Ca2+ signals by the area of the selected frame.
Results
Figure 2A shows a field of view from a small PV at 5 mm Hg intraluminal pressure. The + symbols indicate the automatically detected Ca2+ signals within a selected frame (Video 1), indicated by the green outline. One field of view was recorded from each PV, and one PV was used from each mouse. One small PV was considered as n=1. The number of Ca2+ signals per μm2 per minute was compared before and 5 min after the addition of RyR inhibitor ryanodine (5 μM)23 in paired experiments. A paired t-test was used for statistical comparisons to detect significant differences. The number of spontaneously occurring Ca2+ signals was 0.73 ± 0.2 events per minute per μm2 (Figure 2B,C) under the conditions described above. Ryanodine treatment almost completely inhibited the Ca2+ signals in small PVs (Figure 2C, Video 2), as indicated by a drastic reduction in events per minute per μm2. These data show that spontaneous activity of Ca2+ signals in the myocyte layer from pressurized small PVs represents openings of RyRs. These results provide evidence that the activity of Ca2+ signals can be studied in small PVs under physiological intraluminal pressures. This method will facilitate future studies on the effects of intraluminal pressure and neurohumoral mediators on Ca2+ signaling in small PV myocytes, potentially offering valuable insights into the regulation of PV function. While the current study focuses on the number of RyR Ca2+ signals, it is also possible to analyze the kinetic properties of these signals, including duration, amplitude, spatial spread, and rise time. Understanding how physiological and pathological stimuli affect the kinetic properties of RyR Ca2+ signals may provide important information regarding the regulation of PV function in both healthy and diseased states.
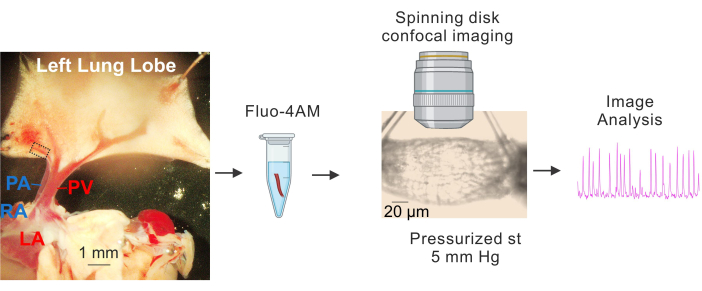
Figure 1: Preparation and Ca2+ imaging of PVs. Sequential images showing the identification and isolation of small PVs, incubation of fluo4-AM, cannulation and pressurization (5 mm Hg), spinning disk high-speed confocal imaging, and image analysis. The dashed rectangle in the image on the left represents the PV used for the study. Abbreviations: PA = pulmonary artery; RA = right atrium; PV = pulmonary vein; LA = left atrium. Please click here to view a larger version of this figure.
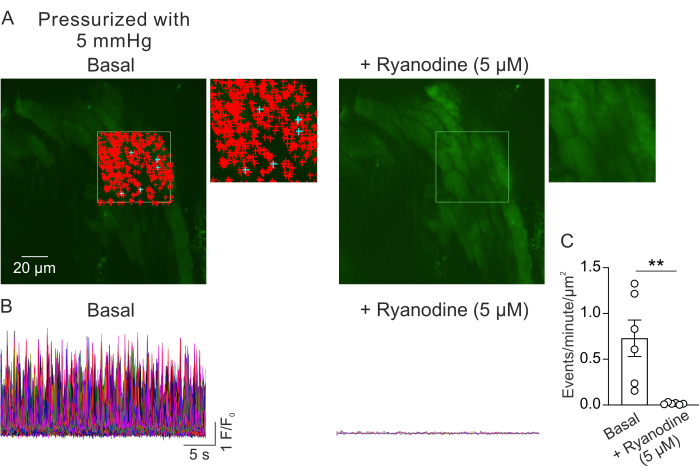
Figure 2: Analysis of Ca2+ imaging. (A) Images of a field of view from a pressurized (5 mm Hg), fluo-4 loaded, small PV in the absence (left) or presence of ryanodine (5 µM; RyR inhibitor, right). The green rectangle denotes the area that was selected for autodetection Ca2+ signals. The + signs represent autodetected events within the frame. Red signs indicate signals with area of less than 3000 µm2, whereas blue signs indicate signals that occupied an area of more than 3000 µm2. Each + sign indicates one Ca2+ event or signal. (B) F/F0 traces of detected events from the selected area indicated in A, showing the activity of Ca2+ signals in the myocyte layer from pressurized small PVs under basal conditions (left) and in the presence of ryanodine (right). (C) Quantification of events per minute per µm2 of Ca2+ signals in small PVs before and after the addition of ryanodine (1 PV per mouse; n=6 mice; **p < 0.01 vs Basal; paired t-test). Please click here to view a larger version of this figure.
Video 1: Ca2+ signals in a small PV pressurized at 5 mm Hg before treatment with ryanodine. The field of view represents the selected frame, shown in green color in Figure 2. Please click here to download this Video.
Video 2: Ca2+ signals in a small PV pressurized at 5 mm Hg after treatment with ryanodine (5 μM). The field of view represents the selected frame, shown in green color in Figure 2. Please click here to download this Video.
Discussion
Ca2+ signals in smooth muscle cells play a crucial role in regulating vascular function25. PVs are an important but relatively understudied vascular bed responsible for delivering oxygen-rich blood to the left heart26. However, Ca2+ signaling mechanisms in small PVs and their impact on physiological function are unknown. The goal of this manuscript is to describe a new method for isolating, cannulating, and pressurizing small PVs to record Ca2+ signals in the myocyte layer and to analyze these Ca2+ signals. The ability to visualize Ca2+ signals in small PVs could help advance our understanding of how these signals work under normal and diseased conditions. While studying Ca2+ signals in PVs within the intact lung would provide physiologically more relevant data, small PVs are not clearly visible in the intact lung, which may introduce technical challenges with loading Ca2+ dyes and confocal imaging. Isolating and pressurizing small PVs offers an alternative way to observe Ca2+ signals under physiological intraluminal pressures, temperature, and ionic solutions.
Ca2+ imaging of PVs described in this manuscript is based on our well-established procedure for Ca2+ imaging in systemic arteries21,22,27. The data indicates that the majority of Ca2+ signals in small PV myocytes are due to openings of RyR ion channels. Therefore, the protocol outlined in this manuscript will enable future studies of RyR regulation and its impact on PV function under normal conditions and in diseases. RyR2 activity is essential for contractions in cardiac myocytes28, while in arterial smooth muscle cells, it has been associated with relaxation29. The precise effect of RyR activity on small PV contraction remains unknown. Pressure myography is a commonly used technique for monitoring changes in the contraction of small blood vessels. Therefore, it is likely that pressurized PV preparation can also be used to monitor small PV contractions.
Ryanodine exhibits concentration-dependent effects on RyR activity, increasing it at submicromolar concentrations and decreasing it at micromolar concentrations23. Ryanodine may also have an indirect inhibitory effect on other Ca2+ signals in small PVs, a possibility that has not been tested. At the concentration used, we did not observe a compensatory increase in Ca2+ signals in PVs. The regulation of Ca2+ signals in isolated and pressurized PVs may differ from those in PVs within an intact lung, a possibility that has yet to be explored. However, recording Ca2+ signals in small PVs in the intact lung from an anesthetized mouse presents significant challenges, arising from the location of the lung in the thoracic cavity and the difficulty of loading a Ca2+ indicator selectively in PVs in an anesthetized mouse.
Blood flow shear stress is known to alter the function of blood vessels. Although the current method allows isolating the effect of intraluminal pressure on Ca2+ signals in small PVs, the lack of flow or shear stress is a potential limitation. However, flow or shear stress can be easily incorporated and performed in combination with Ca2+ imaging using setups described previously21. It is possible that flow/shear stress modifies the activity of Ca2+ signals in small PVs, which would be an interesting topic for future investigation.
We used fluo-4-AM as a Ca2+ indicator to record Ca2+ signals in small PVs. Depending on the experimental needs, it should be possible to use other Ca2+ indicators that can be excited at 488 nm (Calbryte 520 AM)7 or 560 nm (Calbryte 590 AM)30. The use of a spinning disk confocal imaging system allows for imaging of Ca2+ signals, specifically in PV myocytes. Although not studied in the current study, focusing on the endothelial cell layer will allow the recording of endothelial Ca2+ signals in small PVs. PAs and PVs are functionally different and are exposed to distinct microenvironments. Therefore, a comparison of myocyte and endothelial Ca2+ signals between small PAs and small PVs may provide crucial insights into their functional differences.
It is plausible that Ca2+ signals in small PVs regulate the flow of oxygen-rich blood through PVs. The pressure inside the left atrium changes from 4 mmHg to 12 mmHg during each cardiac cycle17,18. Since PVs supply blood to the left atrium, it is likely that the pressure inside the PVs also changes during the cardiac cycle. Previous studies have shown that RyRs can be activated by intraluminal pressure in small arteries16. Therefore, measuring the effect of intraluminal pressure on PV Ca2+ signals may provide novel insights into the delivery of oxygen-rich blood from PVs to the left heart. Nerve stimulation and humoral mediators in the bloodstream can also activate smooth muscle Ca2+ signals. The current method will facilitate future studies on the impact of neurohumoral mediators on PV Ca2+ signals and PV function.
There are a few critical steps within the protocol that should be taken care of. A careful dissection of pulmonary veins (PVs) is extremely important. It is crucial to avoid touching the imaging area of the PVs with dissection tools, as this can negatively affect their health. Only the ends of the PVs should be touched. Reducing dissection time can enhance the success rate. Insufficient dye loading can result in a lower signal-to-noise ratio and increase the likelihood of false positives during event autodetection. Following the protocol outlined here should ensure adequate dye loading in PV myocytes.
In summary, we have presented a method for recording Ca2+ signals in small PVs, which allows for investigating Ca2+ signaling mechanisms in this important but often overlooked vascular bed. Importantly, the ability to record Ca2+ signals in small PVs under normal conditions could provide a valuable understanding of pathological mechanisms for diseases of the lung, including pulmonary hypertension, lung injury, and heart-failure-induced lung edema.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research was funded by awards from the NHLBI (HL167208, HL157407, and HL146914) to SKS and NIDDK (DK138271) to YLC.
Materials
| Name | Company | Catalog Number | Comments |
| Custom-Designed SparkAn Software | University of Vermont | https://github.com/vesselman/SparkAn | |
| Fluo-4, AM | Thermo Fisher Scientific | F14201 | |
| FN1 Upright Microscope | Nikon | ||
| Heater/Temperature Controller | Warner Instruments | ||
| Imaging System | Andor | ||
| iQ 3.x acquisition Software | Andor | ||
| iXon EMCCD Cameras | Andor | ||
| Nylon Thread for Tying Blood Vessels | Living Systems Instrumentation | THR-G | |
| Pressure Myography Chamber | Instrumentation and Model Facility, University of Vermont, Burlington, VT, USA | ||
| Revolution WD (with Borealis) High Speed Spinning Disk Confocal Imaging System | Andor | ||
| Ryanodine | Bio-Techne | 1329 | |
| Servo Pressure Controller | Living Systems Instrumentation | ||
| SS Dissection Pins, 0.2mm dia | Living Systems Instrumentation | PIN-0.2mm | |
| Tubing Pump REGLO | ISMATEC | ISM4212 |
References
- Suresh, K., Shimoda, L. A. Lung circulation. Compr Physiol. 6 (2), 897-943 (2016).
- Sundjaja, J. H., Bordoni, B. . Thorax, heart pulmonic valve. , (2024).
- Daneva, Z., et al. Caveolar peroxynitrite formation impairs endothelial TRPV4 channels and elevates pulmonary arterial pressure in pulmonary hypertension. Proc Natl Acad Sci U S A. 118 (17), e2023130118 (2021).
- Daneva, Z., et al. Endothelial pannexin 1-TRPV4 channel signaling lowers pulmonary arterial pressure in mice. Elife. 10, e67777 (2021).
- Kuhr, F. K., Smith, K. A., Song, M. Y., Levitan, I., Yuan, J. X. New mechanisms of pulmonary arterial hypertension: role of Ca2+ signaling. Am J Physiol Heart Circ Physiol. 302 (8), H1546-H1562 (2012).
- Sonkusare, S. K., Laubach, V. E. Endothelial TRPV4 channels in lung edema and injury. Curr Top Membr. 89, 43-62 (2022).
- Kuppusamy, M., et al. Purinergic P2Y2 receptor-induced activation of endothelial TRPV4 channels mediates lung ischemia-reperfusion injury. Sci Signal. 16 (808), eadg1553 (2023).
- Alvarez, D. F., et al. Transient receptor potential vanilloid 4-mediated disruption of the alveolar septal barrier: a novel mechanism of acute lung injury. Circ Res. 99 (9), 988-995 (2006).
- Balakrishna, S., et al. TRPV4 inhibition counteracts edema and inflammation and improves pulmonary function and oxygen saturation in chemically induced acute lung injury. Am J Physiol Lung Cell Mol Physiol. 307 (2), L158-L172 (2014).
- Suresh, K., et al. Reactive oxygen species induced Ca2+ influx via TRPV4 and microvascular endothelial dysfunction in the SU5416/hypoxia model of pulmonary arterial hypertension. Am J Physiol Lung Cell Mol Physiol. 314 (5), L893-L907 (2018).
- Haissaguerre, M., et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 339 (10), 659-666 (1998).
- Heijman, J., Voigt, N., Nattel, S., Dobrev, D. Cellular and molecular electrophysiology of atrial fibrillation initiation, maintenance, and progression. Circ Res. 114 (9), 1483-1499 (2014).
- Brunton, T. L., Fayrer, J. Note on independent pulsation of the pulmonary veins and vena cava. Proc R Soc Lond. 25, 174-176 (1877).
- Namekata, I., et al. Involvement of the Na(+)/Ca(2+) exchanger in the automaticity of guinea-pig pulmonary vein myocardium as revealed by SEA0400. J Pharmacol Sci. 110 (1), 111-116 (2009).
- Cros, C., et al. Regional differences in Ca2+ signaling and transverse-tubules across left atrium from adult sheep. Int J Mol Sci. 24 (3), 2347 (2023).
- Dabertrand, F., Nelson, M. T., Brayden, J. E. Ryanodine receptors, calcium signaling, and regulation of vascular tone in the cerebral parenchymal microcirculation. Microcirculation. 20 (4), 307-316 (2013).
- Nair, R., Lamaa, N. . Pulmonary capillary wedge pressure. , (2024).
- Fukuta, H., Little, W. C. The cardiac cycle and the physiologic basis of left ventricular contraction, ejection, relaxation, and filling. Heart Fail Clin. 4 (1), 1-11 (2008).
- Daneva, Z., et al. Endothelial IK and SK channel activation decreases pulmonary arterial pressure and vascular remodeling in pulmonary hypertension. Pulm Circ. 13 (1), e12186 (2023).
- Marziano, C., et al. Nitric oxide-dependent feedback loop regulates transient receptor potential vanilloid 4 (TRPV4) channel cooperativity and endothelial function in small pulmonary arteries. J Am Heart Assoc. 6 (12), e007157 (2017).
- Chen, Y. L., et al. Novel smooth muscle Ca2+-signaling nanodomains in blood pressure regulation. Circulation. 146 (7), 548-564 (2022).
- Sonkusare, S. K., et al. Elementary Ca2+ signals through endothelial TRPV4 channels regulate vascular function. Science. 336, 597-601 (2012).
- Arias-Cavieres, A., et al. Ryanodine receptor-mediated calcium release has a key role in hippocampal LTD induction. Front Cell Neurosci. 12, 403 (2018).
- Chen, Y. L., et al. Calcium signal profiles in vascular endothelium from Cdh5-GCaMP8 and Cx40-GCaMP2 mice. J Vasc Res. 58 (3), 159-171 (2021).
- Ottolini, M., Sonkusare, S. K. The calcium signaling mechanisms in arterial smooth muscle and endothelial cells. Compr Physiol. 11 (2), 1831-1869 (2021).
- Boyette, L. C., Burns, B. . Physiology, pulmonary circulation. , (2024).
- Ottolini, M., et al. Local peroxynitrite impairs endothelial transient receptor potential vanilloid 4 channels and elevates blood pressure in obesity. Circulation. 141 (16), 1318-1333 (2020).
- Shan, J., et al. Phosphorylation of the ryanodine receptor mediates the cardiac fight or flight response in mice. J Clin Invest. 120 (12), 4388-4398 (2010).
- Knot, H. J., Standen, N. B., Nelson, M. T. Ryanodine receptors regulate arterial diameter and wall [Ca2+] in cerebral arteries of rat via Ca2+-dependent K+ channels. J Physiol. 1 (Pt 1), 211-221 (1998).
- Tischbirek, C., Birkner, A., Jia, H., Sakmann, B., Konnerth, A. Deep two-photon brain imaging with a red-shifted fluorometric Ca2+ indicator. Proc Natl Acad Sci U S A. 112 (36), 11377-11382 (2015).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved