A subscription to JoVE is required to view this content. Sign in or start your free trial.
Development and Characterization of Fusidic Acid-Loaded Alginate-Aloe vera Based Hydrogel Film
In This Article
Summary
A formulation of fusidic acid-loaded hydrogel film was developed with different Aloe vera ratios and characterized in this study.
Abstract
A new fusidic acid-loaded hydrogel film was prepared via the solvent casting technique using alginate and Aloe vera. The hydrogel films were optimized using different ratios of sodium alginate, Aloe vera, and glycerin. The films containing 10% glycerin (w/w of alginate) exhibited the best appearance. Incorporating Aloe vera influenced the thickness, swelling behavior, water vapor permeability, and drug release profile of the hydrogel films. Higher Aloe vera content resulted in thicker films (up to a certain ratio), increased swelling, reduced water vapor permeability, and a prolonged drug release of up to 93% over 12 h. Fourier-transform infrared spectroscopy (FTIR) analysis confirmed the presence of key functional groups and the interaction between the hydrogel components. The study suggests that the combination of sodium alginate, Aloe vera, and glycerin can improve the mechanical properties and drug release profiles of hydrogel films, making them a promising option for enhanced topical drug delivery and wound healing applications.
Introduction
Fusidic acid is a potent steroidal antibiotic derived from Fusidium coccineum. It is widely used to treat staphylococcal skin infections and impetigo. This drug has high efficacy, even against antibiotic-resistant strains, and low toxicity, making it a suitable option for managing skin infections1. Unlike other topical antibiotics, fusidic acid penetrates deeply into the skin, enhancing its antimicrobial effects, especially in areas where the skin's protective barrier is damaged. Its structure allows for versatile solubility characteristics, enabling widespread distribution throughout body tissues, which is further enhanced in its sodium fusidate form for better water solubility and penetration rates2,3. Fusidic acid can be administered in various ways, including orally, intravenously, and topically, adapting to different treatment needs and making it a flexible choice for various infections4.
Hydrogels, with their high water content and biocompatibility, have emerged as innovative carriers for antibiotics in wound treatment, providing moisture retention, controlled drug release, and improved patient outcomes. They are made from natural or synthetic polymers like alginate, which is favored for its strong biocompatibility, affordability, and gelation properties that mimic natural tissue extracellular matrices5. Alginate-based hydrogels, in particular, offer a moist environment conducive to wound healing, do not adhere to the wound, and allow for easy removal, making them ideal for delivering bioactive substances and supporting tissue regeneration. The development of hydrogels encapsulating antibiotics represents a significant advancement in wound care, ensuring targeted drug delivery and sustained therapeutic effects6,7.
The proposed fusidic acid-loaded alginate/Aloe vera hydrogel aims to overcome fusidic acid's solubility challenges and enhance its dermal penetration. Aloe vera, known for its skin-friendly, moisturizing, and antibacterial properties, also serves as a chemical penetration enhancer, potentially improving the efficacy of fusidic acid in treating skin infections8,9. This innovative formulation leverages the synergistic effects of Aloe vera and alginate hydrogel to create a conducive healing environment, optimize drug delivery, and offer a promising solution for enhanced treatment of skin infections, making the most of fusidic acid's potent antimicrobial activity with improved delivery and healing outcomes10.
Protocol
The details of the reagents and equipment used in this study are listed in the Table of Materials.
1. Preparation of fusidic acid hydrogel films
- Prepare solutions of sodium alginate (2.0% w/v) and Aloe vera (1.0% w/v) by dissolving them in distilled water.
- Add 200 mg of fusidic acid to the sodium alginate solution and stir for 1 h.
- Add glycerol to the alginate solution at concentrations of 10%, 12%, and 14% (w/w, based on the alginate mass).
- Combine the alginate and Aloe vera solutions to achieve final alginate/Aloe vera ratios (v/v) of 100:0, 90:10, 80:20, and 75:25.
- Cast 25 mL of each mixture into glass Petri dishes (100 mm x 20 mm) and leave to dry at room temperature (25 °C) and controlled humidity (50%) for 2 days.
- Immerse the dried films into a calcium chloride (CaCl2) aqueous solution (5.0% w/v) for 5 min to obtain the hydrogel films.
- Wash the resulting films with distilled water and dry them at room temperature before use.
2. Determining the film thickness
- Use a digital micrometer to measure the thickness of the film11.
- Take measurements at five distinct points across the film.
- Record the measurements obtained from each point.
- Calculate the average film thickness based on the measurements taken.
3. Determining the swelling index
- Cut the film samples into 2 cm x 2 cm squares using a lab knife.
- Weigh each sample accurately.
- Soak the samples in PBS buffer (pH 6.8) at room temperature for 24 h.
- Absorb any excess water on the film surface using filter paper.
- Weigh the samples again.
- Weigh the films at different time intervals. Calculate the swelling capacity using the following equation:
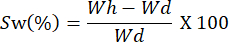
NOTE: Wh represents the hydrated weight of the sample, and Wd corresponds to the dry weight of the sample.
4. Water vapor permeability or occlusion test
- Cover the opening of a glass beaker containing 50 mL of water with filter paper11.
- Apply a film-forming solution to one of the papers and permit it to develop a film.
- Store the beaker at room temperature and humidity.
- Assess the film's permeability to water based on the decrease in water weight within the beaker.
- To determine the evaluation of water vapor permeability for the prepared hydrogel film, use the following formula:
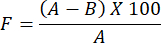
NOTE: The occlusivity factor, F, measures the permeability of a film. It is determined by calculating the difference in water weight between a glass beaker covered with filter paper without a film and a glass beaker covered with filter paper coated with the film. A smaller occlusivity factor value indicates greater film permeability.
5. Fourier transform infrared spectroscopy (FTIR)
- Place the hydrogel sample onto the ATR crystal of the FTIR spectroscopy machine.
- Set the wavenumber range to 4000-400 cm−1.
- Set the scan rate to 60 scans per sample.
- Set the resolution to 4 cm−1.
- Record an air background before each sample run.
- Run the analysis for each hydrogel sample individually.
- Analyze the FTIR spectra for each hydrogel sample to confirm functional groups and examine interactions among all components12.
- Repeat the analysis for raw Aloe vera, sodium alginate, glycerin, and fusidic acid separately.
6. Differential Scanning Calorimetry (DSC)
- Use a commercially available thermal analysis instrument to acquire thermal profiles.
- Craft the measurement samples from multiple circular pieces cut from the polymer film.
- Investigate the temperature range from 25-230 °C.
- Set the heating/cooling rate at 20 °C/min.
- Carry out temperature and enthalpy calibration using standard indium with compatible software.
7. Drug release test
- Prepare a solution of 50 mL PBS (pH = 7.4) as the dissolution medium.
- Immerse the hydrogel films in PBS at 37 °C for 24 h while stirring at a rate of 50 rpm.
- At predetermined intervals, collect 2 mL of the samples.
- Dilute and filter the collected samples through a syringe filter.
- Promptly replenish an equivalent volume of PBS.
- Assess the concentrations of fusidic acid released using a UV spectrophotometer, measuring absorption at 285 nm.
- Evaluate the release kinetics of fusidic acid using zero-order, first-order, Higuchi, and Korsmeyer-Peppas models4.
Results
Preparation of alginate/Aloe vera hydrogel film
Different ratios of sodium alginate, Aloe vera, and glycerin were used to prepare the formulation of hydrogel film. The final preparation and assessment of the formulations were carried out only with 10% glycerin (w/w of alginate). Figure 1 shows the hydrogel film with different Aloe vera ratios. This selection was made because other glycerin ratios did not provide the necessary adhesiveness and...
Discussion
Fusidic acid was incorporated in alginate hydrogel films with various ratios of Aloe vera and glycerin. The presence of Aloe vera did not affect the appearance, but a decrease in viscosity was observed in the solutions containing Aloe vera during preparation. The findings from thickness testing suggest that the inclusion of Aloe vera may have some impact on film thickness to a certain extent, but it is not considered significant. In general, there is not a single ideal thickness that s...
Disclosures
The authors have nothing to disclose.
Acknowledgements
This study was supported by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R30), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. This research was funded by the Researchers Supporting Project number (RSPD2024R811), King Saud University, Riyadh, Saudi Arabia.
Materials
| Name | Company | Catalog Number | Comments |
| Aloe vera | Local supplier, Kuala Lumpur, Malaysia | It was raw aloe vera bark and prepared the materials in our lab | |
| Calcium Chloride | R&M Chemicals | ||
| Differential Scanning Calorimetry | Netzsch-Gruppe | DSC 300 Caliris | |
| FTIR spectroscopy | Perkin Elmer | 107914 | |
| Fusidic Acid | Sigma-Aldrich, St. Louis, MO, USA | PHR2810 | certified reference material, pharmaceutical secondary standard |
| Glycerin | Sigma-Aldrich, St. Louis, MO, USA | PHR1020 | United States Pharmacopeia (USP) Reference Standard |
| Micrometer Screw Gauge | Blomker Industries, Malaysia | ||
| NETZSCH proteus software | Netzsch-Gruppe | DSC 300 Caliris | |
| Phosphate Buffer Saline | Sigma-Aldrich, St. Louis, MO, USA | P4417 | Tablets |
| Sodium alginate | Sigma-Aldrich, St. Louis, MO, USA | W201502 | |
| thermal analysis instrument | NETZSCH | DSC Caliris | |
| UV-SPECTROPHOTOMETER / UV LINE-9400 | SECOMAM | / 8512047 |
References
- Bandyopadhyay, D. Topical antibacterials in dermatology. Indian J Dermatol. 66 (2), 117-125 (2021).
- Williamson, D. A., Carter, G. P., Howden, B. P. Current and emerging topical antibacterials and antiseptics: agents, action, and resistance patterns. Clin Microbiol Rev. 30, 827-860 (2017).
- Bonamonte, D., Belloni Fortina, A., Neri, L., Patrizi, A. Fusidic acid in skin infections and infected atopic eczema. G Ital Dermatol Venereol. 149 (4), 453-459 (2014).
- Curbete, M. M., Nunes, R. A. Critical review of the properties of fusidic acid and analytical methods for its determination. Crit Rev Anal Chem. 46 (4), 352-360 (2015).
- Zhao, C., Zhou, L., Chiao, M., Yang, W. Antibacterial hydrogel coating: Strategies in surface chemistry. Adv Colloid Interface. 285, 102280-102280 (2020).
- Ahmed, E. M. Hydrogel- Preparation, characterization, and applications: A review. J Adv Res. 6, 105-121 (2015).
- Stan, D., et al. Wound healing applications of creams and "smart" hydrogels. Exp Dermatol. 30 (9), 1218-1232 (2021).
- Cole, L., Heard, C. M. Skin permeation enhancement potential of Aloe vera and a proposed mechanism of action based upon size exclusion and pull effect. Int J Pharm. 333 (1-2), 10-16 (2007).
- Sharma, K., Mittal, A., Chauhan, N. Aloe vera as penetration enhancer. Int J Drug Dev Res. 7, 31-43 (2015).
- Parhi, R., Suresh, P., Mondal, S., Kumar, P. M. Novel penetration enhancers for skin applications: A Review. Curr Drug Deliv. 9 (2), 219-230 (2012).
- Hadi, A., Nawab, A., Alam, F., Zehra, K. Alginate/Aloe vera films reinforced with tragacanth gum. Food Chemistry: Mol Sci. 30 (4), 100105 (2022).
- Gull, N., et al. In vitro study of chitosan-based multi-responsive hydrogels as drug release vehicles: a preclinical study. RSC Adv. 9 (53), 31078-31091 (2019).
- Ciarleglio, G., Cinti, F., Toto, E., Santonicola, M. G. Synthesis and characterization of alginate gel beads with embedded zeolite structures as carriers of hydrophobic curcumin. Gels. 9 (9), 714 (2023).
- Dong, H., et al. Smart polycationic hydrogel dressing for dynamic wound healing. Small. 18 (25), 2201620 (2022).
- Giz, A. S., et al. A detailed investigation of the effect of calcium cross-linking and glycerol plasticizing on the physical properties of alginate films. Int J Biol Macromol. 148, 49-55 (2020).
- Fadhilah, S., Aisyah, N., Mohd, N., Mat, K. A. Sodium alginate film: The effect of crosslinker on physical and mechanical properties. IOP Conf Ser Mater Sci. 509, 012063 (2019).
- Kudłacik-Kramarczyk, S., et al. Investigations on the impact of the introduction of the Aloe vera into the hydrogel matrix on cytotoxic and hydrophilic properties of these systems considered as potential wound dressings. Mater Sci Eng C. 123, 111977 (2021).
- Pereira, R., et al. Development of novel alginate based hydrogel films for wound healing applications. Int J Biol Macromol. 52, 221-230 (2013).
- Hadi, A., Nawab, A., Alam, F., Zehra, K. Physical, mechanical, optical, barrier, and antioxidant properties of sodium alginate-Aloe vera biocomposite film. J Food Process. 45 (5), e15444 (2021).
- Yoshida, C. M., et al. Effect of chitosan and Aloe vera extract concentrations on the physicochemical properties of chitosan biofilms. Polymers. 13 (8), 1187 (2021).
- Nuutila, K., Eriksson, E. Moist wound healing with commonly available dressings. Adv Wound Care. 10 (12), 685-698 (2021).
- Naeem, F., et al. pH-responsive cross-linked polymeric matrices based on natural polymers: effect of process variables on swelling characterization and drug delivery properties. BioImpacts: BI. 7 (3), 177 (2017).
- Mahmood, A., et al. Aloe vera-based polymeric network: A promising approach for sustained drug delivery, development, characterization, and in vitro evaluation. Gels. 9 (6), 474 (2023).
- Kamaraj, N., Rajaguru, P. Y., Issac, P., Sundaresan, S. Fabrication, characterization, in vitro drug release and glucose uptake activity of 14-deoxy, 11, 12-didehydroandrographolide loaded polycaprolactone nanoparticles. Asian J Pharm Sci. 11 (4), 353-362 (2017).
- Chalitangkoon, J., Wongkittisin, M., Monvisade, P. Silver loaded hydroxyethylacryl chitosan/sodium alginate hydrogel films for controlled drug release wound dressings. Int J Biol Macromol. 159, 194-203 (2020).
- Laracuente, M. L., Yu, M. H., McHugh, K. J. Zero-order drug delivery: State of the art and future prospects. J Control Release. 327, 834-856 (2020).
- Wang, Y., et al. A sustained zero-order release carrier for long-acting, peakless basal insulin therapy. J Mater Chem B. 8 (9), 1952-1959 (2020).
- Unagolla, J. M., Jayasuriya, A. C. Drug transport mechanisms and in vitro release kinetics of vancomycin encapsulated chitosan-alginate polyelectrolyte microparticles as a controlled drug delivery system. Eur J Pharm Sci. 114, 199-209 (2018).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved