Palladium-Catalyzed Cross Coupling
Source: Vy M. Dong and Faben Cruz, Department of Chemistry, University of California, Irvine, CA
This experiment will demonstrate the concept of a palladium-catalyzed cross coupling. The set-up of a typical Pd-catalyzed cross coupling reaction will be illustrated. Pd-catalyzed cross coupling reactions have had a profound effect on how synthetic chemists create molecules. These reactions have enabled chemists to construct bonds in new and more efficient ways. Such reactions have found widespread applications in the fine chemical and pharmaceutical industries. Pd-catalyzed cross coupling reactions add another tool to the chemist's toolbox for constructing carbon-carbon bonds, which are central to organic chemistry. The combination of the importance of making carbon-carbon bonds and the impact of Pd-catalyzed cross coupling have resulted in these reactions being the subject of the 2010 Nobel Prize in Chemistry. Ei-ichi Negishi, one of the recipients of the 2010 Nobel Prize in chemistry, explained in his Nobel lecture that one of his motivations for developing this chemistry was to develop "widely applicable straightforward Lego-like methods for hooking up two different organic groups".
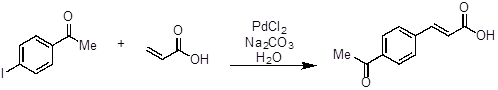
- Add 4-iodoacetophenone (246 mg, 1 equivalent, 1 mmol), acrylic acid (100 μL, 1.5 equivalents, 1.5 mmol), sodium carbonate (Na2CO3, 318 mg, 3 equivalents, 3 mmol), PdCl2 (2 mg, 0.01 equivalents, 0.01 mmol), and water (5 mL, 0.2 M) to a round bottom flask (~ 20 mL) equipped with a magnetic stir bar.
- Heat the reaction to approximately 100 °C and stir until complete consumption of 4-iodoacetoph
The product should be a solid with the follow 1H NMR spectrum: 1H NMR (400 MHz, DMSO-d6): δ (ppm) 2.60 (s, 3H), 6.67 (d, J = 16.0 Hz, 1H), 7.65 (d, J = 16.0 Hz, 1H). 7.83 (d, J = 8.4 Hz, 2H). 7.97 (d, J = 8.4 Hz, 2H).
These Pd-catalyzed cross coupling reactions have changed the way molecules are synthesized in academic and industrial settings. The impact of this technology can be seen in how chemists construct complex structures for pharmaceuticals, agriculture chemicals, and materials. Beyond Pd-catalyzed cross couplings, transition metal catalysis has changed (and is continuing to change) the way synthetic chemists prepare molecules that can have an impact on society through their potential therapeutic use.
Log in or to access full content. Learn more about your institution’s access to JoVE content here
スキップ先...
このコレクションのビデオ:

Now Playing
Palladium-Catalyzed Cross Coupling
Organic Chemistry II
33.7K 閲覧数

クリーニングのガラス製品
Organic Chemistry II
122.8K 閲覧数

求核置換反応
Organic Chemistry II
99.0K 閲覧数

還元剤
Organic Chemistry II
42.6K 閲覧数

グリニャール反応
Organic Chemistry II
148.5K 閲覧数

n- ブチル リチウムの滴定
Organic Chemistry II
47.5K 閲覧数

ディーン ・ スターク ・ トラップ
Organic Chemistry II
99.5K 閲覧数

アルケンのオゾン分解
Organic Chemistry II
66.6K 閲覧数

有機触媒反応
Organic Chemistry II
16.5K 閲覧数

固相合成
Organic Chemistry II
40.6K 閲覧数

水素化
Organic Chemistry II
48.7K 閲覧数

重合
Organic Chemistry II
93.0K 閲覧数

融点
Organic Chemistry II
149.0K 閲覧数

赤外分光法
Organic Chemistry II
213.4K 閲覧数

旋光計
Organic Chemistry II
99.6K 閲覧数
Copyright © 2023 MyJoVE Corporation. All rights reserved