Microscopy and Staining: Gram, Capsule, and Endospore Staining
Overview
Source: Rhiannon M. LeVeque1, Natalia Martin1, Andrew J. Van Alst1, and Victor J. DiRita1
1 Department of Microbiology and Molecular Genetics, Michigan State University, East Lansing, Michigan, United States of America
Bacteria are diverse microorganisms found nearly everywhere on Earth. Many properties help distinguish them from each other, including but not limited to Gram-staining type, shape and arrangement, production of capsule, and formation of spores. To observe these properties, one can use light microscopy; however, some bacterial characteristics (for example size, lack of coloration, and refractive properties) make it hard to distinguish bacteria solely with a light microscope (1, 2). Staining bacteria is necessary when distinguishing bacterial types with light microscopy. The two main types of light microscopes are simple and compound. The main difference between them is the number of lenses used to magnify the object. Simple microscopes (for example a magnifying glass) have only one lens to magnify an object, while compound microscopes have several lenses to enhance magnification (Figure 1). Compound microscopes have an objective lens close to the object which collects light to create an image of the object. This is then magnified by the eyepiece (ocular lens) which enlarges the image. Combining the objective lens and eyepiece allows for higher magnification than using a single lens alone. Typically, compound microscopes have multiple objective lenses of varying powers to allow for different magnification (1, 2). Here, we will discuss visualizing bacteria with Gram stains, Capsule stains, and Endospore stains.

Figure 1: A typical compound microscope. The most important parts of the microscope are labeled.
The Gram stain, developed in 1884 by the Danish bacteriologist Hans Christian Gram (1), differentiates bacteria based on the composition of the cell wall (1, 2, 3, 4). Briefly, a bacterial smear is placed on a microscope slide and then heat-fixed to adhere the cells to the slide and make them more readily accepting of stains (1). The heat-fixed sample is stained with Crystal Violet, turning the cells purple. The slide is flushed with an Iodine solution, which fixes the Crystal Violet to the cell wall, followed by a decolorizer (an alcohol) to wash away any non-fixed Crystal Violet. In the final step, a counterstain, Safranin, is added to color cells red (Figure 2). Gram-positive bacteria stain purple due to the thick peptidoglycan layer which is not easily penetrated by the decolorizer; Gram-negative bacteria, with their thinner peptidoglycan layer and higher lipid content, destain with the decolorizer and are counterstained red when Safranin is added (Figure 3). Gram staining is used to differentiate cells into two types (Gram-positive and Gram-negative) and is also useful to distinguish cell shape (spheres or cocci, rods, curved rods, and spirals) and arrangement (single cells, pairs, chains, groups, and clusters) (1, 3).
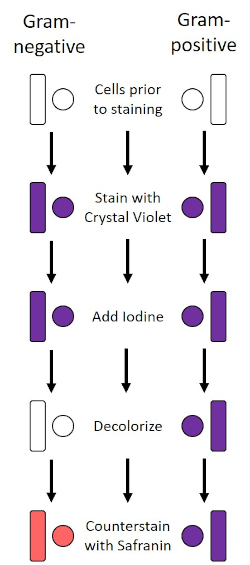
Figure 2: Schematic of the Gram Staining Protocol. The left column shows how Gram-negative bacteria react at each step of the protocol. The right column shows how Gram-positive bacteria react. Also, shown are two typical bacterial cell shapes: the bacilli (or rods) and the cocci (or spheres).
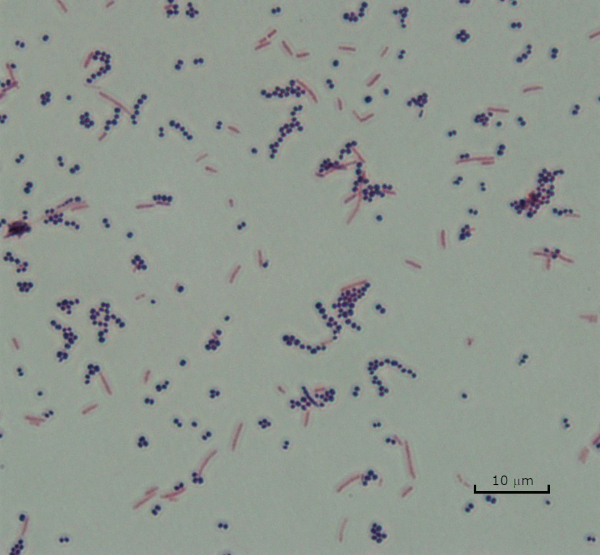
Figure 3: Gram Staining Results. A Gram stain of a mixture of Staphylococcus aureus (Gram-positive purple cocci) and Escherichia coli (Gram-negative red rods).
Some bacteria produce an extracellular viscous outer layer called a capsule (3, 5). Capsules are protective structures with various functions, including but not limited to adherence to surfaces and other bacteria, protection from desiccation, and protection from phagocytosis. Capsules are typically composed of polysaccharides containing more than 95% water, but some may contain polyalcohols and polyamines (5). Due to their mostly non-ionic composition and tendency to repel stains, simple staining methods do not work with capsule; instead, capsule staining uses a negative staining technique which stains the cells and the background, leaving the capsule as a clear halo around the cells (1, 3) (Figure 4). Capsule staining involves smearing a bacterial sample into an acidic stain on a microscope slide. Unlike Gram staining, the bacterial smear is not heat-fixed during a Capsule Stain. Heat-fixing can disrupt or dehydrate the capsule, leading to false negatives (5). Furthermore, heat-fixing can shrink cells resulting in a clearing around the cell which can be mistaken as a capsule, leading to false positives (3). The acidic stain colors the slide background; while follow up with a basic stain, Crystal Violet, colors the bacterial cells themselves, leaving the capsule unstained and appearing as a clear halo between the cells and the slide background (Figure 5). Traditionally, India ink has been used as the acidic stain because these particles cannot penetrate the capsule. Therefore, neither the capsule nor the cell is stained by India ink; instead, the background is stained. Congo Red, Nigrosin, or Eosin can be used in place of India ink. Capsule staining can help doctors diagnose bacterial infections when looking at cultures from patient samples and guide appropriate patient treatment. Common diseases caused by encapsulated bacteria include pneumonia, meningitis, and salmonellosis.
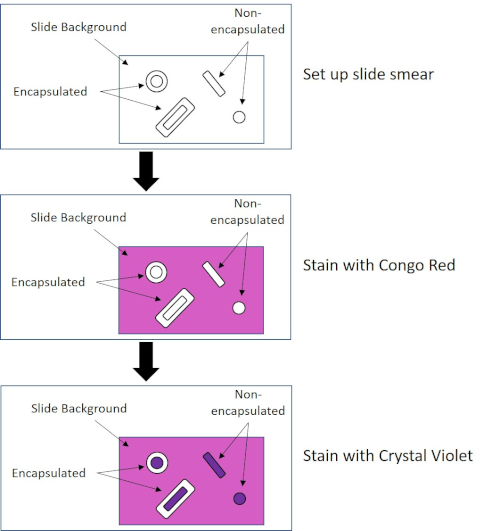
Figure 4: Schematic of the Capsule Staining Protocol. The top panel shows the slide smear prior to any stain application. The middle panel shows how the slide and bacteria look after the primary stain, Congo Red. The final panel shows how the slide and bacteria look after the counterstain, Crystal Violet.
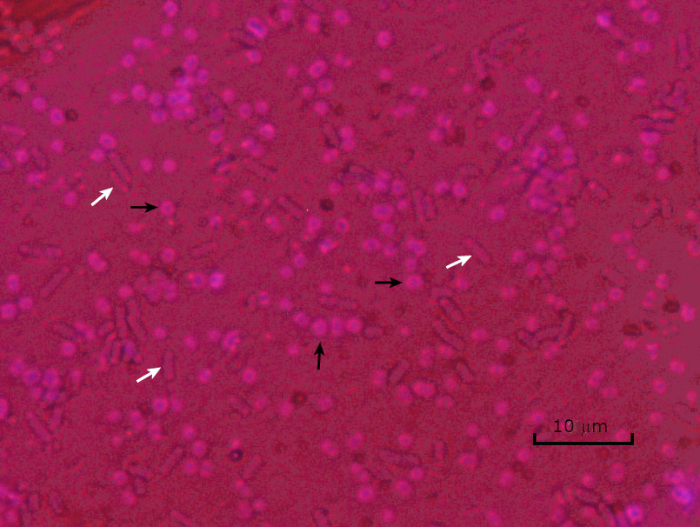
Figure 5: Capsule Staining Results. Capsule staining of encapsulated Acinetobacter baumannii (denoted with black arrows) and non-encapsulated Escherichia coli (denoted with white arrows). Notice the background is dark and A. baumannii cells are stained purple. The capsule around A. baumannii cells is evident as a halo, while E. coli has no halo.
In adverse conditions (for example nutrient limitation, extreme temperatures, or dehydration), some bacteria produce endospores, metabolically inactive structures that are resistant to physical and chemical damage (1, 2, 8, 9). Endospores allow the bacterium to survive harsh conditions by protecting the genetic material of the cells; once conditions are favorable for growth, spores germinate, and bacterial growth continues. Endospores are difficult to stain with standard staining techniques because they are impermeable to dyes typically used for staining (1, 9). The technique routinely used to stain endospores is the Schaeffer-Fulton Method (Figure 6), which uses the primary stain Malachite Green, a water soluble stain that binds relatively weakly to cellular material, and heat, to allow the stain to break through the cortex of the spore (Figure 7). These steps color the growing cells (termed vegetative cells in the context of endospore biology), as well as endospores and any free spores (those no longer within the former cell envelope). Vegetative cells are washed with water to remove Malachite Green; endospores retain the stain due to heating the Malachite Green within the spore. Finally, the vegetative cells are counterstained with Safranin to visualize (Figure 8). Staining for endospores helps differentiate bacteria into spore formers and non-spore formers, as well as determines whether spores are present in a sample which, if present, could lead to bacterial contamination upon germination.
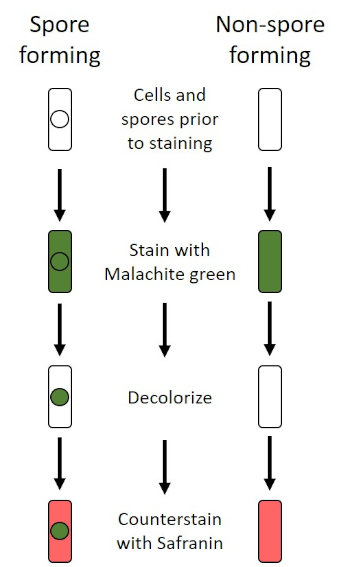
Figure 6: Schematic of the Endospore Staining Protocol. The left column shows how spore forming bacteria react at each step of the protocol. The right column shows how non-spore forming bacteria react.
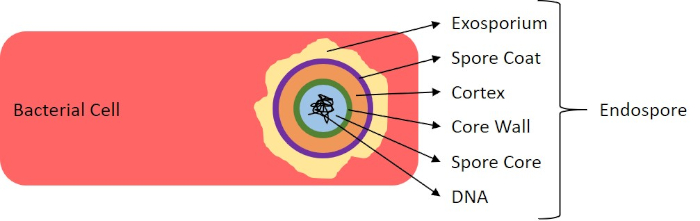
Figure 7: Diagram of Endospore Structure. Bacterial cell containing an endospore with the various spore structures labeled.
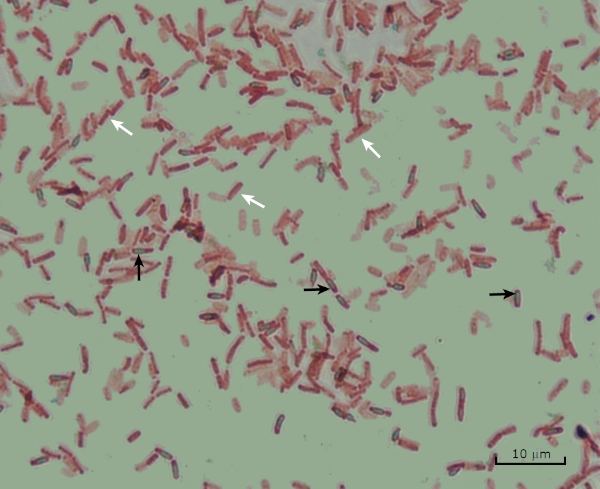
Figure 8: Endospore Staining Results. A typical staining of endospores of Bacillus subtilis. The vegetative cells (denoted with the white arrows) are stained red, while the endospores (denoted with the black arrows) are stained green.
Procedure
1. Gram Staining
- Set-up
- Wear gloves and a non-flammable lab coat, as dyes will stain hands and clothing.
- A Bunsen burner is used to heat-fix the bacteria. Use care when working with flame; tie back long hair.
- Commercially available Gram stain reagents will be used.
- Clean microscope slides with laboratory wipes.
- Protocol
- Pipet 10 µL phosphate-buffered saline or culture broth
Application and Summary
Bacteria have distinguishing characteristics that can aid in their identification. Some of these characteristics can be observed by staining and light microscopy. Three staining techniques useful for observing these characteristics are Gram staining, Capsule staining, and Endospore staining. Each technique identifies different characteristics of bacteria and can be used to help physicians recommend treatments for patients, identify potential contaminants in samples or food products, and verify sample sterility.
References
- Black, J. G. Microbiology Principles and Explorations, 4th edition. Prentice-Hall, Inc., Upper Saddle River, New Jersey. (1999)
- Madigan, M. T. and J. M. Martinko. Brock Biology of Microorganisms, 11th edition. Pearson Prentice Hall, Upper Saddle River, New Jersey. (2006).
- Leboffe, M. J., and B. E. Pierce. A Photographic Atlas for the Microbiology Laboratory, 2nd ed. Morton Publishing Company, Englewood, Colorado. (1996).
- Smith, A. C. and M. A. Hussey. Gram stain protocols. Laboratory Protocols. American Society for Microbiology, Washington, DC. Available from: http://www.asmscience.org/content/education/protocol/protocol.2886. (2005).
- Hughes, R. B. and A. C. Smith. Capsule Stain Protocols Laboratory Protocols. American Society for Microbiology, Washington, DC. Available from: http://www.asmscience.org/content/education/protocol/protocol.3041. (2007).
- Anthony, E. E. Jr. A note on capsule staining. Science 73(1890):319-320 (1931).
- Finegold, S. M., W. J. Martin, and E. G. Scott. Bailey and Scott's Diagnostic Microbiology, 5th edition. The C. V. Mosby Company, St. Louis, Missouri. (1978).
- Gerhardt, P., R. G. E. Murray, W. A. Wood, and N. R. Krieg. Methods for general and molecular bacteriology. ASM Press, Washington, DC. (1994).
- Hussey, M. A. and A. Zayaitz. Endospore Stain Protocol. Laboratory Protocols. American Society for Microbiology, Washington, DC. Available from: http://www.asmscience.org/content/education/protocol/protocol.3112. (2007).
Tags
Skip to...
Videos from this collection:

Now Playing
Microscopy and Staining: Gram, Capsule, and Endospore Staining
Microbiology
362.4K Views

Creating a Winogradsky Column: A Method to Enrich the Microbial Species in a Sediment Sample
Microbiology
128.1K Views

Serial Dilutions and Plating: Microbial Enumeration
Microbiology
313.8K Views

Enrichment Cultures: Culturing Aerobic and Anaerobic Microbes on Selective and Differential Medias
Microbiology
131.7K Views

Pure Cultures and Streak Plating: Isolation of Single Bacterial Colonies from a Mixed Sample
Microbiology
165.6K Views

16S rRNA Sequencing: A PCR-based Technique to Identify Bacterial Species
Microbiology
187.8K Views

Growth Curves: Generating Growth Curves Using Colony Forming Units and Optical Density Measurements
Microbiology
292.8K Views

Antibiotic Susceptibility Testing: Epsilometer Tests to Determine MIC Values of Two Antibiotics and Evaluate Antibiotic Synergy
Microbiology
93.4K Views

Plaque Assay: A Method to Determine Viral Titer as Plaque Forming Units (PFU)
Microbiology
185.6K Views

Transformation of E. coli Cells Using an Adapted Calcium Chloride Procedure
Microbiology
86.2K Views

Conjugation: A Method to Transfer Ampicillin Resistance from Donor to Recipient E. coli
Microbiology
38.0K Views

Phage Transduction: A Method to Transfer Ampicillin Resistance from Donor to Recipient E. coli
Microbiology
28.9K Views
Copyright © 2025 MyJoVE Corporation. All rights reserved