Method Article
Laboratory Scale Slow Cook-Off Testing of Rocket Propellants: The Combustion Rate Analysis of a Slowly Heated Propellant (CRASH-P) Test
In This Article
Summary
We present a protocol for a laboratory-scale slow cook-off test for solid rocket propellants called the Combustion Rate Analysis of a Slowly Heated Propellant (CRASH-P) test. Confined rocket propellants are slowly heated until autoignition, and both the cook-off temperature and the violence of reaction are measured with dynamic pressure sensors.
Abstract
Solid rocket propellants are widely used for propulsion applications by military and space agencies. Although highly effective, they can be dangerous to personnel and equipment under certain conditions, with slow heating in confined conditions being a particular danger. This paper describes a more affordable laboratory test that is easier to set up and was developed for screening rocket propellant ingredients. Rocket propellants are cast into sample holders that have been designed to have the same confinement as standard rocket motors (propellant volume to total volume in the container) and ensure that the propellant is not easily vented. Reaction violence is quantified by the time it takes to reach 90% of the maximum pressure after autoignition, which is analogous to blast overpressure gauges used to measure violence in a full-scale test. A positive correlation was observed between the speed and pressure produced from the reaction and the power produced by the rocket propellant during the reaction.
Introduction
Solid rocket propellants are used extensively in defense, space, and gas-generating applications. They are relatively reliable fuels that perform many functions extremely well. However, many rocket propellants contain dangerous ingredients such as ammonium perchlorate (AP). Rocket propellants with these oxidizers can explode violently when slowly heated1,2,3. There have been several high-profile accidents with the slow heating of rocket propellant or rocket propellant ingredients that have drawn attention to these issues such as the fire and subsequent cook-off of munitions on the USS Forrestal4 and the PEPCON explosion1. While these are thankfully rare events, they can be devastating because of the personnel and equipment losses that occur. Therefore, there is motivation to understand the violence of these reactions and drive them down whenever possible. One of the main causes of violent cook-off events with rocket propellant is that many of the ingredients partially decompose, leaving reactive product gases behind along with the oxidizer with an enhanced reactive surface area.
One specific example of this is the ionic salt, ammonium perchlorate. The low-temperature decomposition of ammonium perchlorate is drawn out and incomplete, leaving reactive intermediate products within a propellant framework with substantial porosity and surface area available for subsequent reactions5,6,7,8,9. In addition, rocket propellants that contain ammonium nitrate and explosive nitramine compounds can have very violent reactions when heated slowly10,11,12. Slow cook-off violence is an important insensitive munition metric because many rockets are required by law to pass these tests13. Currently, the best way to determine whether a rocket propellant formulation reacts too violently under slow heating conditions is to run a slow cook-off (SCO) test on a full-scale rocket motor. These tests involve taking a full-size rocket motor and heating it slowly in a disposable convection oven.
Temperature traces are provided in multiple locations up until the reaction where the violence is then assessed based on various indicators ranging from container damage and fragmentation to simple overpressure gauges and dynamic pressure sensors for measuring blast pressure. These full-scale tests are often expensive and are not practical for investigating minor changes in propellant ingredients14. A few laboratory-scale tests have been developed that involve heating propellants or explosives in a variety of configurations and assessing container damage after the autoignition event. Although current laboratory-scale tests predict time to cook-off well and sometimes the autoignition temperature15,16,17, they are less able to predict the violence.
One commonly used test is the variable confinement cook-off test18 that slowly heats a cylinder of propellant until it ignites. The violence of the reaction is determined by the fragmentation of the chamber and bolts during the exothermic autoignition reaction. The most common laboratory tests use the final condition of the chamber to rank reaction violence, and there is a degree of subjectivity to the assessment. Small differences in reaction violence are difficult to determine. This assessment of violence is qualitative in nature, and it can be difficult to assess whether a change in a formulation ingredient altered the SCO violence. Furthermore, unlike an actual rocket motor, current laboratory tests do not confine the propellant inside a case. Product gases can easily escape, and this is important because the gases can react with the propellant heterogeneously or be reactive themselves, as in the case of ammonia and perchloric acid if ammonium perchlorate is used.
One of the best efforts in instrumenting a laboratory scale test involved the use of a dynamic pressure sensor on a small-scale cook-off bomb19. This allowed for higher resolution, quantifiable differences in reaction violence to be determined for relatively minor changes in rocket propellant formulation. However, a critical problem with this test is that it did not confine the rocket propellants in the same manner as an actual rocket motor, and numerous modeling and subscale experiments have shown this to be an important factor for consideration20. In addition, the propellant usually does not have the same amount of exposed surface area or the same free volume and is not geometrically confined in the same way as a full-scale test. The Combustion Rate Analysis of a Slowly Heated Propellant (CRASH-P) test was conceived to improve upon these previous tests. Samples between 25 g and 100 g can be tested under similar propellant confinement conditions as a full-scale test21. It also provides a means of measuring the power produced from the reaction event quantitatively through dynamic pressure sensor measurements, which is something current subscale tests do not provide. The results have been found to correlate well with full-scale SCO tests.
Protocol
1. Propellant sample preparation
- Carefully mix propellant ingredients (polymeric resin, plasticizers, and solid fuel and oxidizer particles) together in a rotating planetary mixer for a set duration.
NOTE: The duration of mixing depends on the specific formulation, but most mixes take at least 2 h. - Cast uncured rocket propellant into a specially made CRASH-P sample holder. Place a polytetrafluoroethylene mandrel in the center of the sample holder while casting to create a center perforation when the propellant cures. Use a mandrel holder (Figure 1) to ensure the center perforation in the propellant is straight and consistent.
NOTE: CRASH-P sample holders should scale to have the same propellant volume to internal chamber volume as an actual rocket motor to mimic the propellant confinement of a full-scale rocket motor. CRASH-P sample holders are made of polyether ether ketone (PEEK) or aluminum. Although rocket formulations without a metal fuel can use PEEK, metallized formulations should use aluminum holders so that they do not melt prematurely during autoignition. - Place CRASH-P samples in an oven to accelerate any polyurethane reactions or other chemistry required to cure the propellant. Keep the oven temperature at 60 °C for urethane cures, and increase or decrease the temperature depending on the rocket propellant ingredients.
- After the propellants have cured, trim them so that excess propellant does not protrude from the sample holder surface and interfere with the O-ring face seal. Safely remove the mandrel from each formulation by gently pulling it out.
NOTE: The propellant should be trimmed with a razor blade or other sharp object to minimize frictional abrasion against the propellant surface. - Place a silicone O-ring inside the CRASH-P sample holder face for a proper pressure seal (Figure 1).
NOTE: The O-ring size will vary depending on the size of the CRASH-P sample holder. For example, a 025 size O-ring is used for the 25 g test and a 128 size O-ring is used for the 50 g test. - Bolt the cover onto the CRASH-P sample holder, and tighten it with an Allen wrench. Tighten the bolts in a star pattern to distribute the sealing force more evenly.
2. CRASH-P chamber preparation
- Ensure that the CRASH-P chamber is not pressurized by opening the exhaust valve attached to the CRASH-P chamber. Remove the chamber cover, cap, and thrust washer from the CRASH-P body. Attach a plank to the CRASH-P cap for holding the CRASH-P samples.
- Clean the CRASH-P chamber to remove traces of the last test. Scrub all combustion residue with a wire brush, and clean the chamber with an organic solvent such as ethanol, isopropanol, acetone, or methyl ethyl ketone. Dispose of any single-use cleaning materials as hazardous waste according to local and national regulations.
NOTE: Personal protective equipment should be used when cleaning with the listed solvents, such as eye protection, an appropriate laboratory coat, or chemically resistant gloves. - Inspect the CRASH-P dynamic pressure sensors for any unusual wear and tear.
NOTE: The sensors use a recessed mounting with the CRASH-P chamber because they can only handle a maximum temperature of 204 °C to prevent damage to their internal electronics. These high-temperature, charge-amplified sensors use a downstream convertor (see the Table of Materials) to change the signal to an integrated circuit piezoelectric (ICP) signal. - Remove the 1/8 inch American National Pipe Thread (NPT) fittings that attach the pressure sensors to the CRASH-P main body. Clean out any combustion residue with a spatula or organic solvent. Unthread the pressure sensor from the NPT coupling.
- Fill the NPT coupling with room temperature vulcanizing silicone sealant. Thread the pressure sensor back inside, making sure some of the sealant is extruded. Wipe the sealant off so it is flush with the 1/8 inch NPT fitting.
- Let the sealant cure for at least 12 h. Reinstall the NPT-coupled pressure sensors to protect the sensors from blast-induced temperature errors in the dynamic pressure readings.
- Prepare electrical feedthroughs for the temperature diagnostics. Strip the thermocouple wires of their insulation, and run the bare wires through the feedthrough insulating sleeve.
NOTE: The model and type of the electrical feedthroughs will vary depending on the wire gauge and amounts of feedthroughs needed. See the Table of Materials for the electrical feedthroughs used in the CRASH-P chamber. - Use standard K-type thermocouples for the CRASH-P test as the temperature and sampling rates of the test are fairly standard. Install a mating connection on the other end of the feedthrough.
NOTE: For productivity reasons, it is encouraged to make multiple electrical feedthroughs. - Thread the two electrical feedthroughs through the chamber cap. Leave at least 0.3 m of thermocouple for each feedthrough inside the chamber. Ensure that the beaded side of the thermocouples is inside the CRASH-P chamber.
3. Propellant sample installation
- Bolt the sealed CRASH-P sample to the steel plank (Figure 2B) attached to the chamber cap of the CRASH-P test to keep the sample in the middle of the chamber.
NOTE: Ensuring that the sample is in the middle of the chamber without touching the vessel wall ensures that the sample is heated by convection instead of conduction. - Place one of the thermocouples from the electrical feedthroughs inside the propellant sample holder to capture any exothermic reactions. Place another thermocouple on the steel plank, pointing up to sample the air temperature inside the CRASH-P chamber (Figure 2). Ensure that the thermocouple sampling the air temperature is the controlling thermocouple for the temperature controller.
- Place the sealing ring in the ring-like indent on the CRASH-P chamber. Make sure the sealing ring is clean of any foreign object debris.
- Once the sample is secured properly on the plank and the thermocouples are placed properly, slide the chamber cap into the body of the chamber. Take care not to rotate the chamber cap by marking the chamber cap.
- Use a cylindrical rod to insert the thrust washer and completely thread and tighten the retaining head onto the chamber.
- Install the 7/8"-9 set screws hex bolt into the chamber head. Tighten them in a star pattern to ensure that the chamber is tightened evenly. Use a torque wrench for the final chamber tightening to ensure uniform sealing.
NOTE: Generally, 169.48 N∙m are sufficient for uniform sealing. - Install the chamber retainer clamps, and hold them in place with dowel pins. If necessary, use a rubber mallet to ensure a snug fit for the clam and prevent vertical movement from the chamber.
- Install the chamber end plate by bolting this to the testing table to prevent the CRASH-P test from axial movement during an ignition event.
- Plug the dynamic pressure sensor co-axial cables into the signal conditioner. Plug the electrical band heaters (Figure 2D) into the outlet sockets that connect to the temperature controllers so that the band heaters can be controlled by a temperature controller that supplies 220 VAC power to the heaters.
4. Setting up and checking test instrumentation
- Program the temperature controller (requiring 120 VAC power) so that it transmits a 24 V signal to a solid-state relay-a switch that determines when the heating power is turned on or off.
NOTE: Like any cook-off test, programming the temperature controller is crucial for running reliable tests. - Tune the temperature controller before the test to get the appropriate heating characteristics.
NOTE: The proportional gain, integral characteristics, and rate should all be set to minimize oscillations and overshoot. - Set the temperature values needed for the 16 time intervals on the temperature controller. Use the first three intervals to set up a ramp and soak period where the temperature is kept at 50 °C for at least 2 h. Then, enter the intervals to supply the data points for the test to have a linear heating profile that does not change slope during the test (15 °C/h is the goal), and set the final temperature to 300 °C.
- Make sure the inlet and outlet wires are plugged into the dynamic pressure signal conditioner. Turn on the dynamic pressure signal conditioner. If there are no shorts indicated, proceed to the next step.
NOTE: A red light illuminates for a shorted sensor. - Use three K-type thermocouples whose ends terminate inside a thermocouple amplifier, and make sure the amplifier is turned on. Turn on the monitoring camera for the test to record the CRASH-P test by video so operators can see if anything happens to the chamber remotely. Turn on the electrical power to the heaters on the control console (Figure 3), and turn the temperature controller on to run the test remotely.
- On the CTRL page of the temperature controller, turn RSEN to on. Press the aux button on the temperature controller to change the test condition from standby to run so that the test starts heating the chamber.
5. Data acquisition and test clean-up
- Construct a workbench in the data acquisition system software to set up two distinct regions for test data collection: one for pressure to be measured by the main board and the another for temperatures to be taken for the thermocouple amplifier (Figure 3).
- Check the data acquisition system to see if there was a triggered event, which implies that the sample experienced an exothermic reaction and can be stopped. Set the system to run on a triggered sweep mechanism so that after a threshold voltage is reached, the pressure sampling rate goes from one sample a second to 50,000 samples/s to accurately resolve the work done by the reacting sample during autoignition.
NOTE: Inert tests should be run beforehand to investigate how to control the heating rate. The charge amplified sensors can sample at a rate up to 500,000 samples/s, but that speed is usually not necessary for this test. - If an exothermic triggered reaction is observed, press the stop button on the data acquisition software. As the data acquisition does not end on its own, periodically check on the test to check for either a temperature exotherm or a triggered pressure response. If either of these are observed, manually stop the recording, and turn off the heater power, video, and temperature controller.
- Manually export the temperature and pressure data into text files that are tab delimited, making sure that pressure and temperature data are exported separately because of the different sampling rates. Transfer the text files to another computer to perform data analysis on the results.
- Wait for at least 12 h for the test to cool down before disassembling the test chamber. Vent the chamber to release any product gases from the exothermic reaction. Carefully disassemble the test chamber.
NOTE: Wear personal protective equipment-chemical/flame resistant lab coat, appropriate gloves, and a respirator-as rocket propellant products can be hazardous. - Clean the chamber and all components, and capture sample container fragments of the sample holder.
6. CRASH-P data analysis
NOTE: Data analysis consists of the actual temperature traces and the triggered dynamic pressure data. The data acquisition system marks the location of the trigger, and the user can see the time when this occurred. The trigger corresponds to a dynamic pressure value that is 5% higher than the baseline.
- Stop the recording in the software, and export the temperature and pressure data to tab delimited text files.
- Open the text files with graphing software. Check the data for temperature exotherms from which the ignition temperature can be determined, and check how quickly the chamber pressurizes.
- Compare the CRASH-P results to full-scale SCO test data for the formulation being tested, if these are available. Compare autoignition temperature and reaction violence.
Results
To help the reader visualize how the subsystems of the CRASH-P test interact with each other, an experimental schematic is shown in Figure 4. Thermocouples inside the CRASH-P chamber control the feed data to the data acquisition system through a thermocouple amplifier. The temperature controller operates an electric relay, which turns the electric band heaters on and off. This ensures that the correct heating profile is achieved for the rocket propellant sample. When autoignition of the sample occurs, the data acquisition system triggers the collection of high-speed dynamic pressure data at 50,000 samples/s. The test then ends, the data are saved, and the temperature-controlling system is turned off. After at least 12 h, the CRASH-P chamber should be at room temperature, and any product gases can be exhausted.
Typical representative results are seen in Figure 5. Temperature traces are provided for the inside chamber air and the internal propellant temperature by the data acquisition system. Minor exothermic reactions before ignition are often measured along with the main exothermic reaction. Usually, the exothermic reaction is not violent enough to break the thermocouple bead, so the entire event can be captured. In addition, dynamic pressure readings for the reaction are recorded for the front, back, and rear dynamic pressure gauges. Like most laboratory cook-off events, the state of the sample container after reaction can be assessed for damage (Figure 5C). Finally, Figure 5D shows that there can be quite a large degree of measured variation in the reaction violence of different propellant samples, allowing the violence to be quantified and compared for the different reactions. In general, faster pressurizing reactions had more scatter or noise in the pressure data (Figure 5D), which is consistent with the greater oscillation of the chamber due to a more violent response.

Figure 1: Preparation and sealing of CRASH-P samples. (A) Rocket propellant ingredients are mixed in a planetary mixer. (B) Rocket propellant is cast into a sample holder with a polytetrafluoroethylene mandrel. (C) Propellant samples are trimmed, and an O-ring is placed in the container for sealing purposes. (D) Sample container is sealed and bolted. Sample confinement is the same as that of actual rocket motors. Abbreviation: CRASH-P = Combustion Rate Analysis of a Slowly Heated Propellant. Please click here to view a larger version of this figure.

Figure 2: Sample loading and preparation of the CRASH-P test. Sample placement is critical. (A) Samples are placed on a plank and centrally heated by natural convection during testing. (B) Sample is bolted and held in place on the plank. (C) Thermocouples are placed on the plank and inside the propellant sample for temperature control and diagnostic purposes. (D) CRASH-P chamber is sealed, and band heaters are connected to a 220 VAC power supply controlled by the temperature controller. Abbreviation: CRASH-P = Combustion Rate Analysis of a Slowly Heated Propellant. Please click here to view a larger version of this figure.

Figure 3: Instrumentation and data acquisition for the CRASH-P test. (A) Dynamic pressure signal conditioner, (B) thermocouple amplifier, (C) test heating controls, and (D) data acquisition during test. . Abbreviation: CRASH-P = Combustion Rate Analysis of a Slowly Heated Propellant. Please click here to view a larger version of this figure.
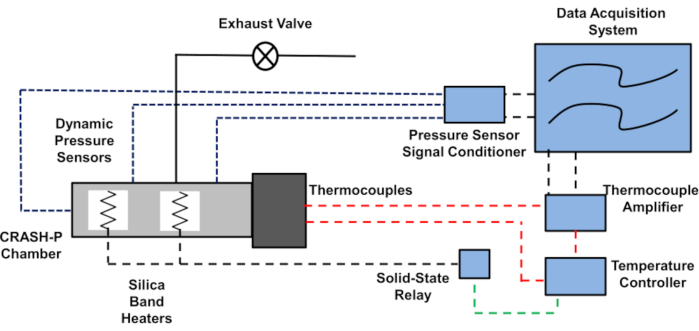
Figure 4: Experimental schematic of the CRASH-P test. The temperature-monitoring system controls the heating rate. Dynamic pressure sensors quantify the reaction violence of the autoignition event, and a data acquisition system records all these test data for the experiment. CRASH-P = Combustion Rate Analysis of a Slowly Heated Propellant. Please click here to view a larger version of this figure.
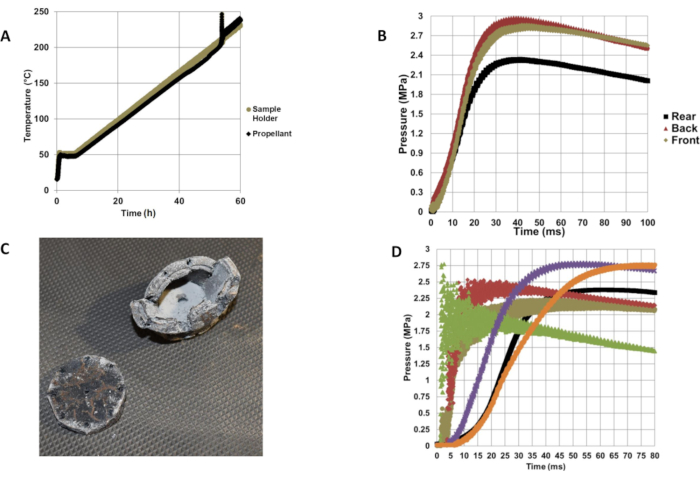
Figure 5: Representative test data for CRASH-P test run. (A) Temperature traces during a test. (B) Rear, back, and front dynamic pressure readings. (C) CRASH-P sample container after test. (D) Comparison of front dynamic pressure readings for six different rocket propellant formulations. CRASH-P = Combustion Rate Analysis of a Slowly Heated Propellant. Please click here to view a larger version of this figure.
Discussion
One of the most important parts of establishing the CRASH-P test was deciding what metric from the test would be best used to quantify the reaction violence of the rocket propellant formulations. The speed and amount of pressure produced from the reaction is directly proportional to the power produced by the rocket propellant when reacting. It is also directly analogous to the blast overpressure gauge used in a full-scale SCO test. Initially, pressurization rate (dP/dt) was used, but these data were misleading because different formulations contain different amounts of fuel and oxidizer and produce different amounts of gas with varying composition. To minimize this bias from the effects of changing the formulation ingredients, the time to 90% peak pressure was used instead, and it correlated well with full-scale SCO test violence.
Another testing operation that was found to be important is confinement. Early sample holders were made with thermoplastic materials designed to handle the high temperatures of the test. Unfortunately, while these samples did not melt, they softened and did not provide the same confinement as metal sample holders. The reaction violence for these samples was noticeably less than the reaction violence for metal sample holders. Another key finding about the test was that some rocket propellant formulations had critical sizes to autoignite reliably. Aluminized formulations had difficulty cooking off and autoigniting if they were under 50 g. This was attributed to the requirement of a threshold amount of ammonium perchlorate that was required for the violent reaction. In addition, another insight was that thermoplastic bolts did not work. The original CRASH-P sample holder bolts were made from PEEK, and this had to be changed to stainless steel. Confinement was not strong enough because of the PEEK material thermally expanding before propellant autoignition was achieved.
For some formulations that ignite at higher temperatures, mainly aluminized formulations, using an aluminum propellant holder case is desirable as they do not soften at higher temperatures. Finally, ICP dynamic pressure sensors were the original pressure sensors used. However, after ~10 tests, the results got increasingly noisy, probably from being exposed to too high a temperature. The dynamic pressure sensors were switched from ICP sensors to charge amplifier sensors. However, charge amplifier sensors lose charge if left on for too long. To minimize this effect, an in-line charge amp-to-ICP convertor was used downstream at a safe temperature region. As the maximum sampling rate of the pressure sensor is 500,000 samples/s, sampling rates faster than 50,000 sample/s could be recorded. However, there was no need for this as the events were not that fast.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank the Joint Enhanced Munitions Technology Program. Mr. Anthony DiStasio and Jeffrey Brock were instrumental in making sure this work was completed.
Materials
| Name | Company | Catalog Number | Comments |
| ½ x 24 x 12’ Ceramic Insulative Blanket | Cotronics Corporation | 370-3 | Thermal Insulation for CRASH-P Chamber |
| 20 gauge K-Type Thermocouple | Omega Engineering | EXPP-K-20-SLE-500 | Thermocouple wire for temperature measurements |
| Dynamic Pressure Signal Conditioner | PCB Piezotronics | 482C16 | Converts ICP signal to voltage for data acquisition system |
| Electrical feedthrough of CRASH-P chamber | Conax | ||
| GC-35 Reaction Chamber | High Pressure Equipment Company | GC-35 | Main Reaction Chamber of CRASH-P Test |
| Gen 3i and Perception software | HBM Inc. | Gen3i | Main Data Acquisition System for CRASH-P Data |
| High-Temperature Charge-Amplified Pressure Sensor | PCB Piezotronics | 113B03 | Dynamic Pressure Sensors used in CRASH-P Test |
| In-Line Charge Amp-to-ICP Converter | PCB Piezotronics | 422E53 | Converters pressure sensor charge amp signal to ICP signal |
| Mica Band Heaters | Omega Engineering | MBH00295 | Resistive Element for Heating up CRASH-P Test |
| Quantum X Thermocouple Amplifier | HBM Inc. | 1-MX1609KB | Used for getting Temperature Measurements |
| Teflon Insulated K-type thermocouple (0.02 inch diameter) | Omega Engineering | 5TC-TT-K-24-36 | K-Type Thermocouples |
| Temperature Controller | Omega Engineering | CN3251 | PID Temperature Controller |
References
- Ibitayo, O. O., Mushkatel, A., Pijawka, K. D. Social and political amplification of technological hazards: The case of the PEPCON explosion. Journal of Hazardous Materials. 114 (1-3), 15-25 (2004).
- Boggs, T. L. The hazards of solid propellant combustion. International Journal of Energetic Materials and Chemical Propulsion. 4 (1-6), 233-267 (1997).
- Price, D., Clairmont, A. R., Jaffe, I. Explosive behavior of ammonium perchlorate. Combustion and Flame. 11 (5), 415-425 (1967).
- Stewart, H. P. The impact of the USS Forrestal's 1967 fire on United States navy shipboard damage control. Master's Thesis, U.S. Army Command and General Staff College. , (2004).
- Bircumshaw, L. L., Newman, B. H. The thermal decomposition of ammonium perchlorate. I. Introduction, experimental analysis of gaseous products, and thermal decomposition experiments. Proceedings of the Royal Society A. Mathematical and Physical Sciences. 227 (1168), 115-132 (1954).
- Bircumshaw, L. L., Newman, B. H. The thermal decomposition of ammonium perchlorate, II. The kinetics of the decomposition, the effect of particle size, and discussion of results. Proceedings of the Royal Society of London . Series A. Mathematical and Physical Sciences. 227 (1169), 228-241 (1955).
- Bircumshaw, L. L., Phillips, T. R. The kinetics of thermal decomposition of ammonium perchlorate. Journal of the Chemical Society (Resumed). 12, 4741-4747 (1957).
- Boldyrev, V. V. Thermal decomposition of ammonium perchlorate. Thermochimica Acta. 443 (1), 1-36 (2006).
- Tolmachoff, E. D., Essel, J. T. Evidence and modeling of heterogeneous reactions of low temperature ammonium perchlorate decomposition. Combustion and Flame. 200, 316-324 (2019).
- Van Dolah, R. W., Mason, C. M., Perzak, F. J. P., Hay, J. E., Forshey, D. R. Explosion hazards of ammonium nitrate under fire exposure. Report of Investigations 6773, United States Department of the Interior, Bureau of Mines. , (1966).
- Doriath, G. Energetic insensitive propellants for solid and ducted rockets. Journal of Propulsion and Power. 11 (4), 870-882 (1995).
- Oxiey, J. C., Kaushik, S. M., Gilson, N. S. Thermal stability and compatibility of ammonium nitrate explosives on a small and large scale. Thermochimica Acta. 212 (21), 77-85 (1992).
- Melita, A. J. US IM Position. Proceedings of the 2006 Insensitive Munitions and Energetic Materials Technology Symposium. , (2007).
- Hayden, H. F., Lustig, E. A., Lawrence, B. G. Development of small-scale slow cook-off (SCO) testing protocol for granular propellants. NDIA Insensitive Munitions and Energetic Materials Conference. , (2015).
- Victor, A. C Simple calculation methods for munitions cookoff times and temperatures. Propellants, Explosives, Pyrotechnics. 20 (5), 252-259 (1995).
- Sandusky, H. W., Chambers, G. P., Erikson, W. W., Schmitt, R. G. Validation experiments for modelling slow cook off. Proceedings of the 12th International Detonation Symposium. , 863-872 (2002).
- Cook, M. P., Stennet, C., Hobbs, M. L. Development of a small scale thermal violence test. No. SAND2018-7274C. Sandia National Lab. , (2018).
- Alexander, K., Gibson, K., Baudler, B. Development of the Variable Confinement Cook-off Test. Indian Head Technical Report 1840. NAVSEA Indian Head Division. , (1996).
- Ho, S. Y. Thermomechanical properties of rocket propellants and correlation with cookoff behavior. Propellants, Explosives, Pyrotechnics. 20 (4), 206-214 (1995).
- Erikson, W. W., Kaneshige, M. J. Pressure and free volume dependence in the cook-off of AP Composite Propellants. No. SAND2014-20085C. 46th JANNAF Combustion Subcommittee. , (2014).
- Essel, J. T., et al. Investigating the effect of chemical ingredient modifications on the slow cook-off violence of ammonium perchlorate solid propellants on the laboratory scale. Journal of Energetic Materials. 38 (2), 127-141 (2020).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved