A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Multi-Stream Perfusion Bioreactor Integrated with Outlet Fractionation for Dynamic Cell Culture
In This Article
Summary
This paper presents a method to construct and operate a low-cost, multichannel perfusion cell culture system for measuring the dynamics of secretion and absorption rates of solutes in cellular processes. The system can also expose cells to dynamic stimulus profiles.
Abstract
Certain cell and tissue functions operate within the dynamic time scale of minutes to hours that are poorly resolved by conventional culture systems. This work has developed a low-cost perfusion bioreactor system that allows culture medium to be continuously perfused into a cell culture module and fractionated in a downstream module to measure dynamics on this scale. The system is constructed almost entirely from commercially available parts and can be parallelized to conduct independent experiments in conventional multi-well cell culture plates simultaneously. This video article demonstrates how to assemble the base setup, which requires only a single multichannel syringe pump and a modified fraction collector to perfuse up to six cultures in parallel. Useful variants on the modular design are also presented that allow for controlled stimulation dynamics, such as solute pulses or pharmacokinetic-like profiles. Importantly, as solute signals travel through the system, they are distorted due to solute dispersion. Furthermore, a method for measuring the residence time distributions (RTDs) of the components of the perfusion setup with a tracer using MATLAB is described. RTDs are useful to calculate how solute signals are distorted by the flow in the multi-compartment system. This system is highly robust and reproducible, so basic researchers can easily adopt it without the need for specialized fabrication facilities.
Introduction
Many important biological processes occur in cell and tissue cultures on the timescale of minutes to hours1,2,3. While some of these phenomena may be observed and recorded in an automated fashion using time-lapse microscopy4, bioluminescence1, or other methods, experiments involving the collection of culture supernatant samples for chemical analysis are often performed manually in static cell cultures. Manual sampling limits the feasibility of certain studies due to the inconvenience of frequent or after-hours sampling timepoints. Further shortcomings of static culture methods include experiments involving controlled, transient exposures to chemical stimuli. In static cultures, stimuli must be added and removed manually, and stimulus profiles are limited to step changes over time, while medium changes also add and remove other medium components, which can affect cells in an uncontrolled manner5. Fluidic systems can overcome these challenges, but existing devices pose other challenges. Microfluidic devices come with the prohibitive costs of specialized equipment and training to produce and use, require microanalytical methods to process samples, and cells are difficult to recover from the devices after perfusion6. Few macrofluidic systems have been created for the types of experiments described here7,8,9,10, and they are built of multiple custom parts made in-house and require multiple pumps or fraction collectors. Furthermore, the authors are not aware of any commercially available macrofluidic perfusion cell culture systems other than stirred tank bioreactors for suspension culture, which are useful for biomanufacturing, though are not designed for modeling and studying physiology.
The authors previously reported on the design of a low-cost perfusion bioreactor system composed almost entirely of commercially available parts11. The base version of the system enables multiple cultures in a well plate to be kept in a CO2 incubator and continuously perfused with medium from a syringe pump, while the effluent medium streams from the cultures are automatically fractionated into samples over time using a fraction collector with a custom modification. Thus, this system enables automated sampling of culture medium supernatant and continuous solute input to the cultures over time. The system is macrofluidic and modular and can be easily modified to meet the needs of novel experiment designs.
The overall goal of the method presented here is to construct, characterize, and use a perfusion cell culture system that enables experiments in which the secretion or absorption rates of substances by cells over time is measured, and/or cells are exposed to precise, transient solute signals. This video article explains how to assemble the base setup, which is capable of perfusing up to six cell cultures simultaneously using a single syringe pump and modified fraction collector. Two useful variants on the base system that make use of additional pumps and parts to allow for experiments that expose cells to transient solute concentration signals, including brief pulses and pharmacokinetic-like profiles12, are also presented, shown in Figure 1.
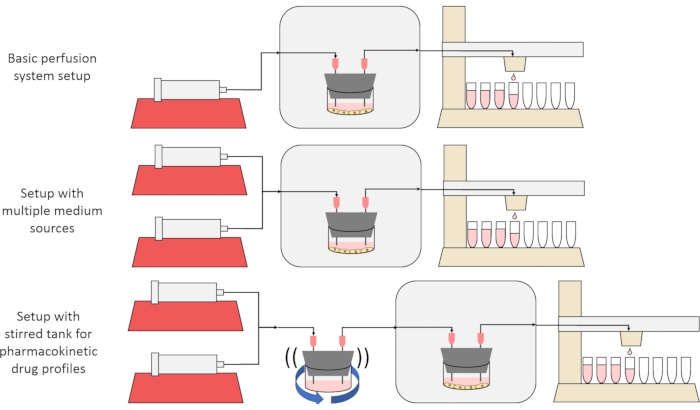
Figure 1: Three variations on the perfusion system design. (Top) The basic perfusion system. (Middle) The perfusion system with a stopcock for multiple medium sources. (Bottom) The perfusion system with a stirred tank to mimic a well-mixed volume of distribution. Please click here to view a larger version of this figure.
Due to dispersion and diffusion within the flow, the solute signals become distorted or "smeared" as they travel through the flow system. This distortion can be quantified through the use of residence time distributions (RTDs)13. This article explains how to perform tracer experiments on components of the perfusion system (Figure 2), and provides MATLAB scripts to generate RTDs from measured data. A detailed explanation of this analysis can be found in the authors' previous paper11. Additional MATLAB scripts fit appropriate functions to the RTDs and extract physical parameters, and perform signal convolution using RTDs to predict how solute signal input by the user will propagate and distort through the perfusion system14.
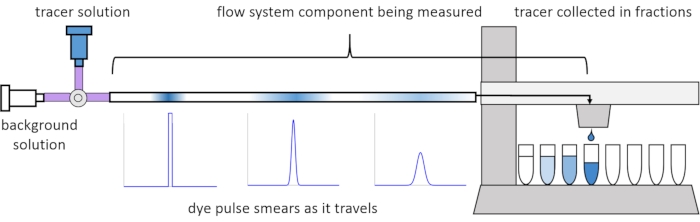
Figure 2: Residence time distributions. The RTDs of flow system components, such as this length of tubing, are measured by inputting a pulse of tracer to the system and measuring how it "smears" by the time it exits into the collected fractions. This figure has been modified from Erickson et al.11. Please click here to view a larger version of this figure.
Access restricted. Please log in or start a trial to view this content.
Protocol
1. Prepare parts for well plate perfusion
- Prepare tubing
- Cut two lengths of silicone tubing (1.6 mm inner diameter) for each cell culture to be perfused. Ensure that the piece used as the upstream tubing is long enough to reach from the syringe pump to the cell culture inside the incubator, and that the downstream piece can reach from the cell culture to the furthest extended position of the fraction collector.
- Give each piece of tubing a unique label on both of its ends with labeled tape.
- Prepare stoppers for the well plate
- Obtain one silicone stopper for each well of the well plate to be perfused, with an appropriate diameter to fit snuggly into the wells with an airtight seal.
- Cut off excess material from the bottoms of the stopper so that they fit into the wells while leaving space inside for air above the intended liquid level.
- Push two blunt 18 G needles through each stopper, into the top and out from the bottom to serve as the inlet and outlet for the flow through the well, diametrically opposite one another to maximize the distance between their tips within the well.
- Adjust the heights of the needles within the plugged well, as the height of the outlet needle will determine the stable height of the liquid level in the well during perfusion.
NOTE: If the perfusion is started with the outlet needle above the liquid level, then the liquid will accumulate in the well until the level reaches the needle. If the perfusion is started with the outlet needle below the liquid level, the liquid level will remain steady unless air bubbles flow into the well, which will cause the liquid height to lower until it is the same height as the outlet needle.
- Gather additional parts
- Obtain one sterile syringe for each cell culture to be perfused that is large enough to contain enough medium for the entire perfusion, plus an additional amount of medium to initially fill the tubing.
- For each culture to be perfused, obtain one female-to-barb and two male-to-barb Luer connectors, as well as two female and two male Luer caps.
- Clean and sterilize parts
- If parts have been used previously, clean them by perfusing them with 0.1 N NaOH, followed by a rinse with deionized water.
- By autoclaving or otherwise, ensure the sterility of all parts listed above.
2. Laser-cut the multi-head dispenser and attach it to a fraction collector
- Recreate the multi-head dispenser model from Figure 3 in a computer-aided design program or download the provided model DXF file (Supplemental File 1).
- Use a laser cutter to cut the design from a 1/8 in acrylic sheet.
- Remove the three screws attaching the dispensing head to the moving base of the fraction collector.
- Align the three smallest holes in the multi-head dispenser with the screw holes in the moving base, and screw the screws back in through the holes to attach.
- Adjust the tightness of the three screws to angle the multi-head dispenser up and down until the row of dispensing holes is aligned with the collection tubes below it.
- Carefully place 300 μL pipette tips through the desired holes to serve as dispensing tips.
NOTE: The fraction collector may be used without the multi-head dispenser to perfuse a single cell culture.
3. Measure component RTDs and perform signal convolution
- Set up pumps and syringes for tracer pulse as shown in Figure 2.
- Obtain two single-channel or multichannel syringe pumps.
- Choose a background solution to represent the medium that will be used in the flow system during cell culture experiments. Ensure that the background solution has similar mass transfer properties to the medium. In many cases, deionized water is an appropriate choice.
- Choose a tracer substance to represent the solute that will be of interest during cell culture experiments. Ensure the tracer has similar mass transfer properties to the solute of interest, and its concentration must be able to be measured. In many cases, food dye is an appropriate choice.
- Dissolve the tracer substance in the background solution to make the tracer solution.
- Fill one syringe with a background solution and load it into one syringe pump. Fill another syringe with tracer solution and load it into the second syringe pump.
- Connect both syringes to two of the three ports of a four-way stopcock using Luer connectors.
- Close the stopcock to the background solution and pump the tracer solution into the stopcock until it begins to drip out the open port. Stop the pump and do not adjust the syringe further.
NOTE: It is important that the moving bar of the syringe pump is pushed up against the syringe plungers before the timed portions of the experiment begin. This will allow the flow to begin immediately when the pump is started. Otherwise, the pump may be started, but flow will not actually begin until the moving bar catches up to the plunger position. - Close the stopcock to the tracer solution and pump the background solution into the stopcock until all residual tracer solution has been flushed out of the open port. Stop the pump and do not adjust the syringe further.
- Set up the flow system component of interest and the fraction collector
- Set up the flow system component desired for RTD analysis. Ensure the component to be measured ends with a piece of downstream tubing of suitable length and flexibility to reach the fraction collector during operation.
- Insert the end of the downstream tubing into a pipette tip dispenser in the multi-head dispenser such that it is snuggly connected.
- Attach the open port of the four-way stopcock to the inlet of the component to be measured. Pump background solution through the component until it is entirely filled as it would be during a cell culture experiment, and it begins to drip out of the fraction collector dispensing tip. Stop the pump.
- Inject tracer pulse, collect fractions, and measure tracer
- Set the pump for the tracer solution to the desired flow rate. Close the stopcock to the background solution and start the flow of the tracer solution. At the same time, start the fraction collector.
- Continue the flow of tracer solution for a short period of time to approximate an impulse input of the tracer. A pulse duration of 10 min was found to work well for RTDs at a flow rate of 1 mL/h.
NOTE: If the tracer pulse is too brief, not enough tracer will enter the flow to be measurable. If the pulse is too long, it will no longer approximate an impulse and will change the shape of the RTD. - At the end of the tracer solution pulse period, stop the tracer solution pump. Quickly close the stopcock to the tracer solution and start the flow of the background solution at the same flow rate.
- Allow the background solution to flow and fractions to be collected until all of the tracer has passed through the system and into the collected fractions.
- Stop the system and measure the tracer concentration in the fractions. Only include fractions that were dispensed completely. If the collection is stopped part-way through the collection of a fraction, do not include that fraction.
- Calculate the residence time distribution (RTD) from measured data in MATLAB
NOTE: A written explanation of the analysis performed by this MATLAB script can be found in the authors' previous publication11, and discussions of the theory are widely available in literature13.- Produce a .xlsx file containing the concentration data in the format of the example_tracer_data.xlsx spreadsheet provided in Supplemental File 2. Enter the concentration values of the tracer in the fractions (any units) in chronological order from left to right in row 2. Enter the time elapsed from the start of the pulse to the end of the last fraction in cell A5, and enter the length of the tracer pulse in minutes in cell A8.
- Save the .xlsx file in the MATLAB directory.
- Open the RTD_From_Data.m script, from Supplementary File 3, in the MATLAB editor.
- Replace the name of the .xlsx file in the parentheses in the first line of the Load Data section of the script with the name of the new .xlsx data file, following the instructions written within the script file. Run the script.
- Ensure that the script successfully performs RTD analysis13, producing a plot of the RTD and returning the value of the numerical integral over the RTD equaling 1. Find the time vector (t) and associated RTD values vector (Et) saved by the script to the MATLAB directory.
- Fit a model function to the RTD in MATLAB
- Open the Fit_RTD_Function.m script in the MATLAB editor from Supplementary File 4.
- Choose one of the three commented-out model functions to fit to the RTD: the axial dispersion model13, which fits RTDs for laminar flow in cylindrical tubes; the CSTR model13, which fits well-stirred tanks; and the n-CSTR model15, which approximately fits larger well plates. To fit another model not included here, add it to the script in the same format.
- Remove the comments in the section of the script containing the model chosen for fitting.
- Change the values of the initial guesses for the parameters to ones appropriate for the RTD.
- Run the script to produce a plot of the fit function overlaid on the RTD data and to print the fit parameter values for the function. If the fit is very poor or errors occur, change the parameter initial guesses and run the script again.
- Perform signal convolution in MATLAB
- Choose either one signal and one RTD or two RTDs to convolve.
- Open the Signal_Convolution.m script, from Supplemental File 5, in the MATLAB editor.
- For each of the two signals to be convolved (i.e., one signal and one RTD, or two RTDs), define one vector of evenly spaced time points in the desired units and a corresponding vector of signal values at those times.
NOTE: The vectors of the two signals must have the same number of elements and the same size time steps. This is why it's useful to have the RTD as a continuous function that can be sampled for an arbitrary number of points at any time interval. - Enter the two signals into MATLAB and run the script to obtain the time and signal vectors of the output signal.
4. Set up the basic perfusion system with cells in a well plate
- Prepare the well plate
- Ensure the well plate culture has the appropriate medium depth for the perfusion experiment. Perform any final medium changes, stimulations, or other steps as desired prior to beginning perfusion. If suspension cells are being perfused, centrifuge the plate to ensure they are on the bottom.
- Under sterile conditions, insert the stoppers with needles into the well plate cultures with the needles pulled up. After the stopper is in place, lower the needles to the desired height for perfusion, as the height of the outlet needle determines the stable liquid level.
- Cap the needles with male Luer caps and keep the whole well plate in an incubator until use.
- Prepare the syringes and upstream tubing
- Under sterile conditions, fill one syringe for each culture to be perfused with enough medium for the desired duration of the perfusion, plus enough additional medium to fill the upstream tubing.
- Attach the upstream tubing to the syringe using a female-to-barb Luer connector. On the other end of the tube, insert a male-to-barb Luer connector.
- Dispense medium from the syringe until the upstream tube is entirely filled with medium.
- Cap the open end of the tube with a female Luer cap.
NOTE: All replicate syringes must have exactly the same volume at this point. If their volumes are not equal, their plungers will be in different positions, and they will not all fit well into a single multichannel syringe pump.
- Under sterile conditions, insert a male-to-barb Luer connector into one end of the downstream tubing, and cap it with a female Luer cap.
- Carefully bring all prepared tubing, syringes, and the well plate to the incubator that will be used for perfusion.
- Place the syringe pump and fraction collector in the desired locations near the incubator. Place the syringe pump on top of or near the incubator, and place the fraction collector next to the incubator, near the port.
- Bundle together the capped ends of all the upstream and downstream tubes and push them from the outside of the incubator to the inside through the port.
- Load the syringes into the syringe pump and insert the open ends of the downstream tubes into the dispensing pipette tips of the multi-head dispenser of the fraction collector.
- Inside the incubator, pull as much slack of the upstream tubes as possible into the incubator to maximize the length of tubing through which the flowing medium can receive heat and CO2 from the incubator air. While holding these in place, pull the downstream tubes out of the incubator, just enough so that they are able to reach the furthest extended point on the fraction collector while still keeping the capped ends inside the incubator.
- For each plugged well, quickly uncap the needles and the upstream and downstream tubes for that well and attach them together with their Luer connectors.
- Once all parts are connected, briefly run the syringe pump at a relatively high speed to ensure that all streams are flowing properly.
- At this point, if it is desired to begin the experiment with the downstream tubes full of the medium, continue running the pump until all are filled. Otherwise, stop the pump.
- Set the syringe pump flow rate and the frequency of fraction collection and start both machines simultaneously to begin the experiment. Collect fractions for the desired experiment duration.
5. Set up the perfusion system with a stopcock for multiple medium sources
- Perform all sub-steps of step 4.1 above.
- Prepare the two media to be used in the perfusion, labeling the medium that will be dispensed first as 1, and the other as medium 2.
- For each culture to be perfused, fill one syringe with enough medium 1 for the duration of its dispensation, plus enough volume to initially fill the perfusion system. Fill a second syringe with enough medium 2 for the duration of its dispensation.
- Connect both syringes to two of the three ports of a four-way stopcock.
NOTE: A length of tubing to connect the syringes to the stopcocks may be required. - Prepare the stopcock and syringes in a similar manner as steps 3.1.7-3.1.8 above by closing the stopcock to medium 1 and dispensing medium 2 into the stopcock until it just begins to drip out the open port.
- Close the stopcock to medium 2 and dispense medium 1 into the stopcock until all residual medium 2 has been flushed out of the open port.
- Attach the upstream tubing to the open stopcock port using a female-to-barb Luer connector. On the other end of the tube, insert a male-to-barb Luer connector.
- Dispense medium 1 from the syringe until the upstream tube is entirely filled with medium.
- Proceed with steps 4.3-4.11 above, loading both syringes into separate syringe pumps and only dispensing medium 1.
- Set the syringe pump flow rate for medium 1 and the frequency of fraction collection, and start both machines simultaneously to begin the experiment.
- When the medium source is to be changed, quickly stop the syringe pump for medium 1, turn the stopcock closed to medium 1, and start the syringe pump for medium 2. If desired later, switch the source back to medium 1 in a similar manner.
- Collect fractions for the desired experiment duration.
6. Set up the perfusion system with a stirred tank to mimic pharmacokinetics
- Obtain pharmacokinetic data for the drug of interest and ensure that it consists of a concentration peak followed by an exponential decay.
- After setting the peak to time 0 and removing data points prior to the peak, use the Stirred_Tank_Fit.m script (Supplemental File 6) to fit the stirred tank RTD equation to the data. Input v (the desired perfusion flow rate) and the data to be fit as a pair of vectors directly into the script, along with t and C for time values and concentration values, respectively. Run the script to print the parameter V, which is the required stirred tank volume.
- Plan a layout for the perfusion system to include two syringe pumps and a plate shaker upstream of the incubator.
- Measure the RTDs of the perfusion system components beyond the stopcock and perform signal convolution of the RTDs with various drug pulse durations and concentrations to find an appropriate pharmacokinetic profile. Use the fit stirred tank RTD equation in this calculation.
- Proceed with steps 5.1-5.8 above.
- Use an additional well plate of appropriate size as the stirred tank for the setup. Each well may serve as a stirred tank for one perfused culture. Fill the well with the required volume of medium, V, and plug the well with the needles initially pulled up, then pushed to the bottom of the well, and cap the needles.
- Load the syringes into the syringe pumps and quickly connect the tubing from the syringes to the capped inlet needles of the stirred tanks. Then connect the cell culture upstream tube inlets to the outlet needles of the stirred tanks.
- Proceed with steps 5.9-5.10.
- At the desired time, switch to dispensing medium 2 containing the drug, as described in step 5.11. Switch back to medium 1 when the drug infusion is complete.
- Proceed with step 5.12.
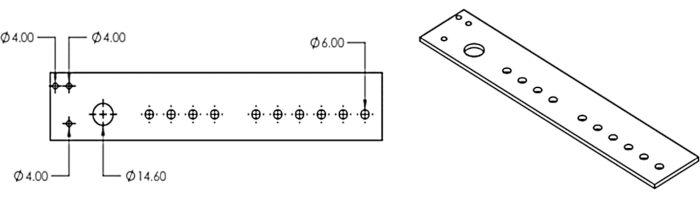
Figure 3: The multi-head dispenser. Design for the laser-cut multi-head dispenser. This figure has been modified from Erickson et al.11. Please click here to view a larger version of this figure.
Access restricted. Please log in or start a trial to view this content.
Results
The perfusion system with multiple medium sources from section 5 of the protocol was used to measure the expression dynamics of a reporter gene driven by the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) transcription factor in human embryonic kidney 293 (HEK293) cells in response to a 1.5 h pulse of tumor necrosis factor alpha (TNF-α). HEK293 cells were stably transduced using lentiviral vectors with a gene construct containing Gaussia luciferase (GLuc), driven by a promoter called NFK...
Access restricted. Please log in or start a trial to view this content.
Discussion
This work describes the assembly and operation of a perfusion cell culture system with multiple medium sources demonstrated with a specific example in which the dynamics of NF-κB-driven gene expression in response to a transient pulse of TNF-α were measured. The RTDs of the perfusion system components were measured and modeled, and signal convolution was used to predict both the exposure of the cells to the TNF-α pulse and the TNF-α distribution in the collected effluent medium fractions. The cells we...
Access restricted. Please log in or start a trial to view this content.
Disclosures
The authors declare no competing interests.
Acknowledgements
This research was conducted with support under Grant Nos. R01EB012521, R01EB028782, and T32 GM008339 from the National Institutes of Health.
Access restricted. Please log in or start a trial to view this content.
Materials
| Name | Company | Catalog Number | Comments |
| 18 Gauge 1 1/2- in Disposable Probe Needle For Use With Syringes and Dispensing Machines | Grainger | 5FVK2 | |
| 293T Cells | ATCC | CRL-3216 | HEK 293T cells used in the Representative Results experiment. |
| 96-Well Clear Bottom Plates, Corning | VWR | 89091-010 | Plates for measuring dye concentrations in RTD experiments and GLuc in representative results experiment. |
| BD Disposable Syringes with Luer-Lok Tips, 5 mL | Fisher Scientific | 14-829-45 | |
| BioFrac Fraction Collector | Bio-Rad | 7410002 | Fraction collector that can be used for a single stream, or modified using our method to enable collection from multiple streams. |
| Clear High-Strength UV-Resistant Acrylic 12" x 12" x 1/8" | McMaster-Carr | 4615T93 | This sheet is cut using a laser cutter according to the DXF file in the supplemental materials to produce the multi-head dispenser that can be attached to the BioFrac fraction collector. |
| Coelenterazine native | NanoLight Technology | 303 | Substrate used in Gaussia luciferase bioluminescence assay in representative results. |
| Corning Costar TC-Treated Multiple Well Plates, size 48 wells, polystyrene plate, flat bottom wells | Millipore Sigma | CLS3548 | Used to grow and perfuse 293T cells in representative results. |
| Corning Costar Flat Bottom Cell Culture Plates, size 12 wells | Fisher Scientific | 720081 | Can be plugged and used as a stirred tank to produce pharmacokinetic profiles in perfusion. Can also contain cells for perfusion. |
| DMEM, high glucose | ThermoFisher Scientific | 11965126 | |
| Epilog Zing 24 Laser | Cutting Edge Systems | Epilog Zing 24 | Laser cutter used to produce multi-head dispenser from acrylic sheet. Other laser cutters may be used. |
| Fisherbrand Sterile Syringes for Single Use, Luer-Lock, 20 mL | Fisher Scientific | 14-955-460 | |
| Fisherbrand Sterile Syringes for Single Use, Luer-Lock, 60 mL | Fisher Scientific | 14-955-461 | |
| Fisherbrand Premium Microcentrifuge Tubes: 1.5mL | Fisher Scientific | 05-408-129 | Microcentrifuge tubes for collecting fractions. |
| Fisherbrand Round Bottom Disposable Borosilicate Glass Tubes with Plain End | Fisher Scientific | 14-961-26 | Glass tubes for collecting fractions. |
| Fisherbrand SureOne Micropoint Pipette Tips, Universal Fit, Non-Filtered | Fisher Scientific | 2707410 | 300 ul pipette tips that best fit the multi-head dispenser and tubing to act as dispensing tips. |
| Gibco DPBS, powder, no calcium, no magnesium | Fisher Scientific | 21600010 | Phosphate buffered saline. |
| Labline 4625 Titer Shaker | Marshall Scientific | Labline 4625 Titer Shaker | Orbital shaker used to keep stirred tanks mixed. |
| Masterflex Fitting, Polycarbonate, Four-Way Stopcock, Male Luer Lock, Non-Sterile; 10/PK | Cole-Parmer | EW-30600-04 | Used to join multiple inlet streams for RTD experiments and cell culture experiments. |
| Masterflex Fitting, Polycarbonate, Straight, Female Luer x Cap; 25/PK | Masterflex | UX-45501-28 | |
| Masterflex Fitting, Polypropylene, Straight, Female Luer to Hosebarb Adapters, 1/16" | Cole-Parmer | EW-45508-00 | |
| Masterflex Fitting, Polypropylene, Straight, Male Luer Lock to Hosebarb Adapter, 1/16" ID | Cole-Parmer | EW-45518-00 | |
| Masterflex Fitting, Polypropylene, Straight, Male Luer Lock to Plug Adapter; 25/PK | Masterflex | EW-30800-30 | |
| Masterflex L/S Precision Pump Tubing, Platinum-Cured Silicone, L/S 14; 25 ft | Masterflex | EW-96410-14 | |
| MATLAB | MathWorks | R2019b | Version R2019b. Newer versions may also be used. Some older versions may work. |
| NE-1600 Six Channel Programmable Syringe Pump | New Era Pump Systems | NE-1600 | |
| Rack Set F1 | Bio-Rad | 7410010 | Racks to hold collecting tubes in the fraction collector. |
| Recombinant Human TNF-alpha (HEK293-expressed) Protein, CF | Bio-Techne | 10291-TA-020 | Cytokine used to stimulate 293T cells in representative results. |
| Saint Gobain Solid Stoppers, Versilic Silicone, Size: 00, Bottom 10.5mm | Saint Gobain | DX263015-50 | Fits 48-well plates. |
| Saint Gobain Solid Stoppers, Versilic Silicone, Size: 4 Bottom 21mm | Saint Gobain | DX263027-10 | Fits 12-well plates. |
| Sodium Hydroxide, 10.0 N Aqueous Solution APHA; 1 L | Spectrum Chemicals | S-395-1LT | |
| SolidWorks | Dassault Systems | SolidWorks | CAD software used to create the multi-head dispenser DXF file. |
| Varioskan LUX multimode microplate reader | ThermoFisher Scientific | VL0000D0 | Plate reader. |
| Wilton Color Right Performance Color System Base Refill, Blue | Michaels | 10404779 | Blue food dye containing Brilliant Blue FCF, used as a tracer in RTD experiments. Absorbance spectrum peaks at 628 nm. |
References
- Welsh, D. K., Yoo, S. H., Liu, A. C., Takahashi, J. S., Kay, S. A. Bioluminescence imaging of individual fibroblasts reveals persistent, independently phased circadian rhythms of clock gene expression. Current Biology. 14 (24), 2289-2295 (2004).
- Talaei, K., et al. A mathematical model of the dynamics of cytokine expression and human immune cell activation in response to the pathogen Staphylococcus aureus. Frontiers in Cellular and Infection Microbiology. 11, 711153(2021).
- Kemas, A. M., Youhanna, S., Zandi Shafagh, R., Lauschke, V. M. Insulin-dependent glucose consumption dynamics in 3D primary human liver cultures measured by a sensitive and specific glucose sensor with nanoliter input volume. FASEB Journal. 35 (3), 21305(2021).
- Muzzey, D., van Oudenaarden, A. Quantitative time-lapse fluorescence microscopy in single cells. Annual Review of Cell and Developmental Biology. 25, 301-327 (2009).
- Calligaro, H., Kinane, C., Bennis, M., Coutanson, C., Dkhissi-Benyahya, O. A standardized method to assess the endogenous activity and the light-response of the retinal clock in mammals. Molecular Vision. 26, 106-116 (2020).
- Battat, S., Weitz, D. A., Whitesides, G. M. An outlook on microfluidics: the promise and the challenge. Lab on a Chip. 22 (3), 530-536 (2022).
- Petrenko, V., Saini, C., Perrin, L., Dibner, C. Parallel measurement of circadian clock gene expression and hormone secretion in human primary cell cultures. Journal of Visualized Experiments. (117), e54673(2016).
- Yamagishi, K., Enomoto, T., Ohmiya, Y. Perfusion-culture-based secreted bioluminescence reporter assay in living cells. Analytical Biochemistry. 354 (1), 15-21 (2006).
- Watanabe, T., et al. Multichannel perfusion culture bioluminescence reporter system for long-term detection in living cells. Analytical Biochemistry. 402 (1), 107-109 (2010).
- Murakami, N., Nakamura, H., Nishi, R., Marumoto, N., Nasu, T. Comparison of circadian oscillation of melatonin release in pineal cells of house sparrow, pigeon and Japanese quail, using cell perfusion systems. Brain Research. 651 (1-2), 209-214 (1994).
- Erickson, P., Houwayek, T., Burr, A., Teryek, M., Parekkadan, B. A continuous flow cell culture system for precision cell stimulation and time-resolved profiling of cell secretion. Analytical Biochemistry. 625, 114213(2021).
- Saltzman, W. M. Drug Delivery: Engineering Principles for Drug Therapy. , Oxford University Press. (2001).
- Fogler, H. S. Elements of Chemical Reaction Engineering. 4th edn. , Prentice Hall PTR. Boston. (2006).
- Conesa, J. A. Chemical Reactor Design: Mathematical Modeling and Applications. , Wiley. (2019).
- Toson, P., Doshi, P., Jajcevic, D. Explicit residence time distribution of a generalised cascade of continuous stirred tank reactors for a description of short recirculation time (bypassing). Processes. 7 (9), 615(2019).
- Tamayo, A. G., Shukor, S., Burr, A., Erickson, P., Parekkadan, B. Tracking leukemic T-cell transcriptional dynamics in vivo with a blood-based reporter assay. FEBS Open Biology. 10 (9), 1868-1879 (2020).
- Newell, B., Bailey, J., Islam, A., Hopkins, L., Lant, P. Characterising bioreactor mixing with residence time distribution (RTD) tests. Water Science and Technology. 37 (12), 43-47 (1998).
- Dubois, J., Tremblay, L., Lepage, M., Vermette, P. Flow dynamics within a bioreactor for tissue engineering by residence time distribution analysis combined with fluorescence and magnetic resonance imaging to investigate forced permeability and apparent diffusion coefficient in a perfusion cell culture chamber. Biotechnology and Bioengineering. 108 (10), 2488-2498 (2011).
- Gaida, L. B., et al. Liquid and gas residence time distribution in a two-stage bioreactor with cell recycle. HAL Open Science. , (2008).
- Rodrigues, M. E., Costa, A. R., Henriques, M., Azeredo, J., Oliveira, R. Wave characterization for mammalian cell culture: residence time distribution. New Biotechnology. 29 (3), 402-408 (2012).
- Olivet, D., Valls, J., Gordillo, M. A., Freixó, A., Sánchez, A. Application of residence time distribution technique to the study of the hydrodynamic behaviour of a full-scale wastewater treatment plant plug-flow bioreactor. Journal of Chemical Technology and Biotechnology. 80 (4), 425-432 (2005).
Access restricted. Please log in or start a trial to view this content.
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved