Preparing Anhydrous Reagents and Equipment
Overview
Source: Laboratory of Dr. Dana Lashley - College of William and Mary
Demonstrated by: Timothy Beck and Lucas Arney
Many reactions in organic chemistry are moisture-sensitive and must be carried out under careful exclusion of water. In these cases the reagents have a high affinity to react with water from the atmosphere and if left exposed the desired reaction will not take place or give poor yields, because the reactants are chemically altered.
In order to prevent undesired reactions with H2O these reactions have to be carried out under an inert atmosphere. An inert atmosphere is generated by running the reaction under nitrogen gas, or in more sensitive cases, under a noble gas such as argon.
Every component in such a reaction must be completely anhydrous, or free of water. This includes all reagents and solvents used as well as all glassware and equipment that will come into contact with the reagents. Extremely water-sensitive reactions must be carried out inside of a glovebox which provides a completely sealed off anhydrous environment to work under via a pair of gloves which protrudes out to one of the sides of the chamber.
Procedure
Drying of Glassware
1. Oven-Drying
- Remove all pieces that are not made of glass, such as the stopcock of an addition funnel.
- Place all glassware that are part of the apparatus in a drying-oven set to about 125 °C for at least 24 h before use.
- Put on heat protection gloves and remove glassware from the oven. Be very careful when handling hot glass while assembling the apparatus.
- For best results flush the apparatus with an inert gas such as N2&#
Results
A classical example for a reaction that must be done under anhydrous conditions is the Grignard reaction. (Equation 1)
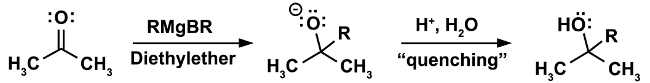
In the first step of the reaction, the nucleophilic attack of the Grignard reagent RMgX occurs on an electrophile (in this case a ketone). In this step
References
- Burfield, D. R. and Smithers, R. H. Desiccant efficiency in solvent and reagent drying. 7. Alcohols. J. Org. Chem. 48 (14), 2420-2422 (1983).
- Williams, D. B. G. and Lawton, M. Drying of Organic Solvents: Quantitative Evaluation of the Efficiency of Several Desiccants. J. Org. Chem. 75 (24), 8351-8354 (2010).
Skip to...
Videos from this collection:

Now Playing
Preparing Anhydrous Reagents and Equipment
Organic Chemistry
79.3K Views

Introduction to Catalysis
Organic Chemistry
34.4K Views

Assembly of a Reflux System for Heated Chemical Reactions
Organic Chemistry
167.2K Views

Conducting Reactions Below Room Temperature
Organic Chemistry
70.5K Views

Schlenk Lines Transfer of Solvents
Organic Chemistry
41.6K Views

Degassing Liquids with Freeze-Pump-Thaw Cycling
Organic Chemistry
56.1K Views

Purifying Compounds by Recrystallization
Organic Chemistry
707.9K Views

Separation of Mixtures via Precipitation
Organic Chemistry
157.6K Views

Solid-Liquid Extraction
Organic Chemistry
237.7K Views

Rotary Evaporation to Remove Solvent
Organic Chemistry
212.8K Views

Fractional Distillation
Organic Chemistry
334.1K Views

Growing Crystals for X-ray Diffraction Analysis
Organic Chemistry
32.4K Views

Performing 1D Thin Layer Chromatography
Organic Chemistry
289.3K Views

Column Chromatography
Organic Chemistry
359.8K Views

Nuclear Magnetic Resonance (NMR) Spectroscopy
Organic Chemistry
247.5K Views
Copyright © 2025 MyJoVE Corporation. All rights reserved