A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Virus-induced Gene Knockdown in the CA3 and Subsequent Spatial Relocation Test to Assess CA3-dependent Cognitive Function
In This Article
Summary
Hippocampal cognitive function can be modulated by locally manipulating gene expression. The spatial object relocation test is a short and robust spatial memory test. We combined this test with virus-induced manipulation of gene expression in the CA3 region to assess the impact of the target gene in shaping cognitive performance.
Abstract
Investigating the role of a gene of interest in a specific brain region allows further understanding of brain physiology and pathophysiology. Modulation of gene expression by local injection of adeno-associated virus (AAV) has been proven to be efficient and safe. The stability and long-term expression of the AAV construct allows the use of a battery of behavioral tests to screen the animals for a region-specific involvement of the target gene in shaping performance in different behavioral domains.
The spatial object relocation (SOR) test is a hippocampal-dependent one-trial memory test based on the natural spontaneous exploratory behavior of rodents. This test gives robust information on memory function and can be easily integrated in a battery of behavioral testing for phenotype screening.
In this video-article, we provide a detailed protocol to assess the role of a particular target gene in shaping hippocampus-dependent spatial memory function. The protocol includes stereotactic AAV-induced gene transfer specifically into the mouse hippocampal CA3 region and combines this with the SOR test. Due to the variability in SOR protocols in the literature, we carefully described relevant aspects of the protocol to ensure the optimal behavioral protocol and setup selection. Also, detailed analyses of the results are described to guarantee the proper interpretation of the results.
Introduction
Dissecting the specific function of an individual gene, expressed in a defined brain region, is a key milestone to a better understanding of brain physiology and pathophysiology. One valuable approach is to investigate the behavioral consequences of local gene expression changes in the brain. Adeno-associated virus (AAV) based gene transfer has been proven to be efficient, safe and to induce long-term gene expression in the central nervous system1–5. In rodents, the stability of AAV-induced gene expression is suitable for extensive behavioral characterization, which usually requires several sessions in different days.
The rationale to select the most suitable behavioral tests to decipher the region-specific function of a target gene might depend on several factors. First, the region of interest might be associated with a prominent behavioral function and measured using a specific set of behavioral tests: e.g. the hippocampal CA3 area is linked to spatial memory6, prefrontal cortex is related to executive function7, amygdala is related to fear8, etc. Second, the gene (and corresponding protein) of interest might be associated with specific functions, such as the glucocorticoid receptor is related to stress9, the serotonin transporter is related to depression10, etc. Also, a battery of behavioral tests could be performed to study different aspects of behavior. However, repeated testing might influence behavior11, therefore considering the order and minimizing the number of tests is important for the validity of the results.
The spatial object relocation test (SOR) is an interesting test to specifically monitor changes in hippocampus-dependent spatial memory function. Similar to the novel object recognition test, the SOR is a one-trial memory test based on rodents spontaneous exploratory behavior12–17 where the hippocampus plays a prominent role18,19. Compared to other spatial memory tests (i.e. Morris water maze20,21, radial arm maze, Barnes maze22), the SOR is short, less stressful (e.g. compared to the swimming effort demanded in the Morris Ware maze), does not require food deprivation (such as the radial arm maze) or repeated training (e.g. Morris water maze, radial arm maze, Barnes maze), and provides a clear readout on the memory function of the animal. For these reasons, it can be easily added to a battery of tests to assess the role of a particular target gene in shaping brain-region dependent changes in cognitive behavior.
The SOR consists of the presentation of two similar objects during an acquisition trial, and after a defined inter-trial-interval (ITI), the animal is exposed to the same arena with one of the objects placed in a new location (retrieval). Based on the natural exploratory behavior of rodents, animals that remember the old location of the objects are expected to explore more the object placed in the new location. However, several protocols have been used in the literature 16,23–27, which show considerable variability in the use of arenas (open field of different sizes, T-maze, Y-maze, other); objects (number of objects presented to the animal, shapes, material, colors); number of habituation and acquisition trials; duration of the different trials; and also the length of the inter-trial-interval time, which is useful to identify changes in short (1 min up to 3 hr) or long-term memory (24 and 48 hr). This variety of different conditions and potential influencing parameters makes it difficult to select the best conditions for a particular experiment and, in particular, to compare results from different laboratories.
In a recent study28, we were interested in further understanding the functional role of the down-regulated in renal-cell carcinoma 1 gene (DRR1) in the hippocampal CA3 region. DRR1 is a glucocorticoid-regulated gene recently suggested to promote stress resilience28,29. This gene shows particularly high constitutive expression in the hippocampal CA3 region28,30,31. In order to study the role of DRR1 in the CA3, we used an AAV containing a short hairpin RNA against DRR1 (shDRR1) to knock-down DRR1 expression and a scrambled version (shSCR) as control28. The phenotype of these mice was characterized using a battery of tests including cognitive (Y-maze, novel object recognition test, SOR and cross water-maze) and anxiety-like domains (open field, elevated plus-maze and forced swimming test). In this particular context, the SOR test was robust and efficient to detect the behavioral changes induced by DRR1 knock-down.
For this reason, the SOR test was chosen to be presented in this video article, in combination with providing detailed information on the stereotactic delivery of AAV-shDRR1 and AVV-shSCR to the mouse hippocampal CA3 region. Moreover, we carefully describe the protocol to perform the SOR and its subsequent analysis. We also provide the rationale used to select our optimal conditions for the SOR test, including data from the pre-test optimization phase. Finally, we show how the knock-down of DRR1 impacts on hippocampus-dependent cognitive function by reducing the performance of the mice in the SOR test.
Protocol
C57Bl6/N male mouse (<8 weeks old) were used for all the procedures. Animals were individually housed and kept on a 12-hr light/dark cycle (lights on at 7:00 AM), at room temperature of 23 ± 2 °C with food and water ad libitum. All experiments were conducted in accordance with European Communities Council Directive 2010/63/EU. All efforts were made to minimize animal suffering during the experiments. The protocols were approved by the committee for the Care and Use of Laboratory Animals of the Government of Upper Bavaria, Germany.
1. Stereotactic Adeno-associated Virus Injection in the Mouse CA3
- Pull capillary glass tubes to create the injection glass pipettes. Cut the tips of the pipette formed to ensure an open end of approximately 45 μm outer and 15 μm inner diameters. The length of the pipette should be enough to allow reaching deep structures with minimum brain tissue damage.
- Fill the isoflurane vaporizer system with isoflurane (approx. 2/3 of one bottle).
- Quickly anaesthetize the mouse in a isoflurane induction chamber or using a closed container. In the second case, add 1-2 ml isoflurane to the closed container, with paper at the bottom, and let the isoflurane vapor fill the container. Place the mouse in the induction chamber or the container until the mouse is completely anaesthetized (approximately 10-20 sec).
- Quickly transfer the animal to the stereotactic frame equipped with isoflurane vaporizer system. Use high isoflurane percentage while fixing the mouse head with the ear bars of the stereotactic apparatus (e.g. 4% vol) and, once the mouse head is correctly fixed, reduce and keep it constant until the end of the surgery (e.g. 1.5% vol). Maintain the mouse body temperature at 37 °C with a heating pad and use a cold light source for illumination.
- Administer a systemic pain killer (non-steroidal anti-inflammatory drug) to prevent postsurgical pain and inflammation.
- Apply a humectant, emollient and moisturizer to the eyes to prevent them from drying.
- Remove the fur on the head with scissors or shaver.
- Administer local anesthesia and disinfect the scalp with povidone-iodine.
- Cut the skin along the midline with a scalpel. Be careful not to harm the muscles from the external occipital crest on the back of the skull.
- Push aside the skin and the membranes above the skull from the midline with cotton swabs, cleaning specially bregma, lambda and around the future injection site.
- Measure bregma and lambda coordinates. If the head of the mouse is properly fixed in the stereotactic frame, the lateral and ventral coordinates should be the same for both bregma and lambda. Anterior-posterior coordinates might vary depending on the age/body weight of the mouse. For mice around 8-10 weeks, expect around 3.9 mm between both anatomical points.
- Use the measured bregma coordinates to calculate the injection sites coordinates. For bilateral hippocampal CA3 injection the coordinates are 1.9 mm anterior to bregma, 2.2 mm lateral from midline and 1.8 mm below dura mater 32.
- Mark the injection sites, and make a corresponding hole through the skull using a micro drill. Avoid high pressure onto the skull to minimize inflammation. Also, discontinuous drilling facilitates heat dissipation and avoids excessive heating of the area.
- Connect the injection system with the pulled pipette and take 0.5-1 µl of viral solution.
- Lower the tip of the pipette containing the virus solution into the drilled hole until the surface of the dura mater. Measure the ventral coordinate and calculate the depth of the injection.
- Carefully descend the pipette using the micromanipulator of the stereotactic system to the desired coordinates and when in position, slowly inject the viral solution (0.5 µl ~ 5 min).
NOTE: The injection speed is important to minimize tissue damage and allow diffusion of the solution through the tissue. Leave the pipette additional 5 min after each injection to avoid solution reflux through the injection site and remove the pipette slowly afterwards. - Discard the pipette in the waste.
- Repeat the steps 1.14 to 1.17 for the second injection site.
- Stitch the skin with surgical suture and anaesthetize again locally.
- Remove the animal from stereotactic system and place it in the home cage over a heating pad until recovery from the anesthesia.
- Dilute an anti-inflammatory such as Metacam 1:1,000 in the drinking water (0.5 ml in 0.5 L tap water) for post-surgical pain killer treatment, change water after 1 week.
- AAV infection is stable 4 weeks after surgery.
2. Behavioral Test
- Preparation of the behavioral setup.
- Start the behavioral testing at least 4 weeks after the surgery. Allow the mice to habituate to the testing room: when possible, house the animals in the room 1 week in advance or move the animals to the room at least 1 hr prior the test.
- Handle the mice daily one week before testing to habituate the animals to the experimenter. Handling involves repeating the procedure of taking out the mouse from the home cage, let the mice walk on the subjects hand and arms and return them to the home cage again.
- Place the Y-maze, consisting of three arms (30 x 10 x 15 cm) at 120° from each other made of gray PVC, in the center of the testing chamber. We used four different chambers (110 x 110 x 130 cm) in order to test four mice at the same time.
- To avoid arm preferences of the animals, make sure none of the arms is perpendicular to any chamber wall and the light in the arena is evenly distributed with a peak around 15-20 Lux in the center of the arena. Optionally, pre-test the arm preference by measuring the distance and times spend in each arm during habituation or priory using another group of mice.
- Locate extra-maze spatial cues at the top of the testing chamber walls. These spatial cues could be white DIN A4 paper cut in different shapes (e.g. two triangles, one rectangle and one rhomboid respectively in three of the chamber walls) to contrast with the black wall of the chamber and visible for the animals performing the test.
- Include also intra-maze cues at the side wall and at the end of each arm in the Y-maze to easily distinguish between them. These cues could be made of white tape in different shapes: e.g. one long vertical stripe, one cross and one triangle, respectively on each arm.
- When more than one apparatus is used at the same time, counterbalance the position of the arms within the chambers.
- Setup the videotracking software to record the tracking and videos of the test.
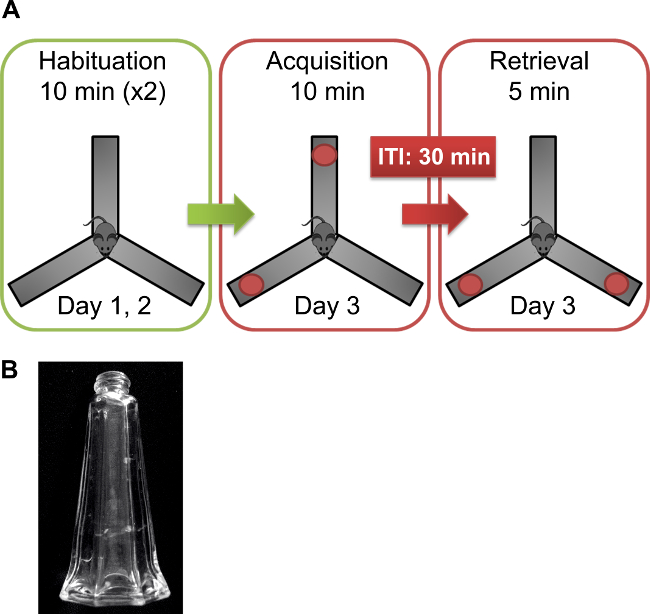
Figure 1. Spatial object relocation protocol. (A) Mice are placed in the center of the Y-maze on two consecutive days (days 1 and 2) for habituation to the arena (10 min/day). On the day of the test (day 3), mice are placed again in the center of the Y-maze containing two similar objects at the end of two of the arms and allowed to explore the arena for 10 min (Acquisition). After an inter-trial interval (ITI), the mice are returned to the Y-maze containing two clean similar objects, one of them in a new location (Retrieval). The arena and objects are thoroughly cleaned after each trial with water and dried before the next trial. (B) The objects used in the SOR test were glass salt shakers.
- Test
- Habituate the mice to the empty arena on two consecutive days. Each day, place the mice in the center of the Y-maze and let them explore for 10 min. Then, return the mice to the respective home cages and clean the surface of the Y-maze thoroughly with water and dry it with paper afterwards.
- On the day of the test, put two identical objects at the end of two of the arms in the Y-maze. Place the objects at the end of the arm approximately 5 cm to the wall, allowing the mice to travel around the object. Counterbalance the position of the objects between chambers and mazes.
NOTE: Ensure that these objects are attractive for the mice, evoking exploratory activity. This might be pre-tested in a group of mice to ensure the mice explore the objects more than 10 sec during 5 min. Glass salt shakers were used in this study as objects. - Let the mice explore the arena with the objects for 10 min → Acquisition trial.
- Return the mice to the respective home cage for the desired inter-trial-interval (in our case 30 min).
- Clean the arena and the objects thoroughly with water and dry them. Place two new clean objects, one in the previous position (old object location) and the second one in the previously empty arm (new object location).
- Let the mice explore the arena with the objects for 5 min → Retrieval trial.
- Return the mice to the respective home cage and clean the arena and the objects thoroughly with water and dry them.
- Disinfect the objects and arenas with e.g. 70% ethanol at the end of the experiment.
3. Analysis
- For each experimental group, measure the total distance travelled. Different distance travelled might bias exploration time of the objects and mask the results.
- Analyze the videos measuring the exploration time of each of the objects. Consider exploration of the objects when the mice touch the objects with the nose, forepaws or vibrissae. Exclude mice when they do not explore both objects. Also, data from mice that explore in total less than 5 sec might be discarded. Total exploration time of the objects should not differ between groups.
- Calculate the index of recognition as a measure of memory function for each mouse, i.e. percentage of exploration of the object in the new location compared to total exploration time during the retrieval phase:
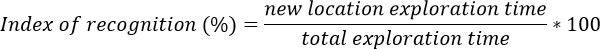
4. Validation of AAV-injection
- Evaluate the correctness of the AAV-injection site and the efficacy of the AVV-induced gene modulation. Most of the AAV co-express fluorescent markers allowing ex vivo fluorescence visualization in brain slices. Also, the efficacy of the AAV-induced gene expression modulation can be quantified by in situ hybridization.
Results
The functional role of DRR1 expression was studied using a combination of AAV-induced mRNA changes in the CA3 region and spatial memory assessment using the SOR test in C57Bl/6N mice 28. We used an AAV containing a short hairpin RNA against DRR1 (shDRR1) to knock-down DRR1 expression and a scrambled version (shSCR) as control. Here we present (1) data showing how the SOR test conditions were previously selected, using naive C56Bl/6N mice; (2) the results of the SOR in mice injected in the CA3 with AAV-shDRR1 o...
Discussion
The SOR is an robust and valid test to investigate changes in hippocampus-dependent cognitive function. The test can be easily included in a screening battery of tests because it is short, non-aversive, and does not require food deprivation or repeated training. In the present video article, we combined the SOR test with an AAV-mediated local gene knock-down of DRR1. The results of the study showed that the SOR was efficient in detecting memory impairment induced by DRR1 knock-down locally in the CA3.
Disclosures
The authors have nothing to disclose.
Acknowledgements
A.U-M is a recipient of a DAAD predoctoral fellowship.
Materials
| Name | Company | Catalog Number | Comments |
| LightCycler Capillaries | Roche | 4929292001 | 1-5 µl glass capillar |
| Micropipette puller | Narishige | PC-10 | |
| Warming pad | |||
| Oxygen/Isofluran system | General anesthesia | ||
| Stereotactic system for mouse | Kofp | ||
| Cold light source | Leica/SCHOTT | KL1600LED | |
| Micro drill: Ultimate-XL, UMXL-T + UHRXL-T | NSK | Y141440 + H269 | |
| Stereo Microscope | Leica | M80 | |
| Panthenol | Bepanthen, Bayer | Humectant, emollient and moisturizer | |
| Lidocain | Xylocain | Local anesthesia | |
| Povidone-Iodine (or propanol) | Betadine (or Kodan) | Antiseptic for desinfection | |
| Meloxicam | Metacam, Heiland | Pain killer | |
| Cotton swabs | |||
| Ethilon sutures | Ethicon | EH7500H | |
| Y-maze | Custom made | Three arms (30 x 10 x 15 cm) at 120° from each other made of grey PVC | |
| Objects | Glass salt shakers | ||
| AAV-shDRR1/AVV-shSCR | Gene Detect | AAV1/2-U6-DRR1 shRNA-terminator-CAG-EGFP-WPRE-BGH-polyA | DRR1 knock-down virus expression cassette |
| AAV-shshSCR | Gene Detect | AAV1/2-U6-scrambled shRNA-terminator-CAG-EGFP-WPRE-BGH-polyA | Control virus expression cassette |
| Videotracking software | ANY-maze | Any videotracking software can be used |
References
- McCown, T. J., Xiao, X., Li, J., Breese, G. R., & Samulski, R. J. Differential and persistent expression patterns of CNS gene transfer by an adeno-associated virus (AAV) vector. Brain researc. 713 (1-2), 99–107, doi:10.1016/0006-8993(95)01488-8 (1996).
- Tenenbaum, L., Chtarto, A., Lehtonen, E., Velu, T., Brotchi, J., & Levivier, M. Recombinant AAV-mediated gene delivery to the central nervous system. The journal of gene medicin. 6 Suppl 1, S212–22, doi:10.1002/jgm.506 (2004).
- Burger, C., Gorbatyuk, O. S., et al. Recombinant AAV viral vectors pseudotyped with viral capsids from serotypes 1, 2, and 5 display differential efficiency and cell tropism after delivery to different regions of the central nervous system. Molecular therapy : the journal of the American Society of Gene Therap. 10 (2), 302–17, doi:10.1016/j.ymthe.2004.05.024 (2004).
- Davidson, B. L., Stein, C. S., et al. Recombinant adeno-associated virus type 2, 4, and 5 vectors: transduction of variant cell types and regions in the mammalian central nervous system. Proceedings of the National Academy of Sciences of the United States of America. 97 (7), 3428–32, doi:10.1073/pnas.050581197 (2000).
- Burger, C., Nash, K., & Mandel, R. J. Recombinant adeno-associated viral vectors in the nervous system. Human gene therap. 16 (7), 781–91, doi:10.1089/hum.2005.16.781 (2005).
- Kesner, R. P. Behavioral functions of the CA3 subregion of the hippocampus. Learning, & memory (Cold Spring Harbor, N.Y.. 14 (11), 771–81, doi:10.1101/lm.688207 (2007).
- Chudasama, Y., & Robbins, T. W. Functions of frontostriatal systems in cognition: comparative neuropsychopharmacological studies in rats, monkeys and humans. Biological psycholog. 73 (1), 19–38, doi:10.1016/j.biopsycho.2006.01.005 (2006).
- Johansen, J. P., Cain, C. K., Ostroff, L. E., & LeDoux, J. E. Molecular mechanisms of fear learning and memory. Cel. 147 (3), 509–24, doi:10.1016/j.cell.2011.10.009 (2011).
- Reul, J. M., & de Kloet, E. R. Two receptor systems for corticosterone in rat brain: microdistribution and differential occupation. Endocrinolog. 117 (6), 2505–11at doi:10.1210/endo-117-6-2505 (1985).
- Levinson, D. F. The genetics of depression: a review. Biological psychiatr. 60 (2), 84–92, doi:10.1016/j.biopsych.2005.08.024 (2006).
- Voikar, V., Vasar, E., & Rauvala, H. Behavioral alterations induced by repeated testing in C57BL/6J and 129S2/Sv mice: implications for phenotyping screens. Genes, Brain and Behavio. 3 (1), 27–38, doi:10.1046/j.1601-183X.2003.0044.x (2004).
- Ennaceur, A., & Delacour, J. A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behavioural brain researc. 31 (1), 47–59, doi:10.1016/0166-4328(88)90157-X (1988).
- Ennaceur, A. One-trial object recognition in rats and mice: methodological and theoretical issues. Behavioural brain researc. 215 (2), 244–54, doi:10.1016/j.bbr.2009.12.036 (2010).
- Dere, E., Huston, J. P., & De Souza Silva, M. a The pharmacology, neuroanatomy and neurogenetics of one-trial object recognition in rodents. Neuroscience and biobehavioral review. 31 (5), 673–704, doi:10.1016/j.neubiorev.2007.01.005 (2007).
- Antunes, M., & Biala, G. The novel object recognition memory: neurobiology, test procedure, and its modifications. Cognitive processin. 13 (2), 93–110, doi:10.1007/s10339-011-0430-z (2012).
- Dix, S. L., & Aggleton, J. P. Extending the spontaneous preference test of recognition: evidence of object-location and object-context recognition. Behavioural brain researc. 99 (2), 191–200, doi:10.1016/S0166-4328(98)00079-5 (1999).
- Murai, T., Okuda, S., Tanaka, T., & Ohta, H. Characteristics of object location memory in mice: Behavioral and pharmacological studies. Physiology, & behavio. 90 (1), 116–24, doi:10.1016/j.physbeh.2006.09.013 (2007).
- Barker, G. R. I., & Warburton, E. C. When is the hippocampus involved in recognition memory? The Journal of neuroscience : the official journal of the Society for Neuroscienc. 31 (29), 10721–31, doi:10.1523/JNEUROSCI.6413-10.2011 (2011).
- Warburton, E. C., & Brown, M. W. Findings from animals concerning when interactions between perirhinal cortex, hippocampus and medial prefrontal cortex are necessary for recognition memory. Neuropsychologi. 48 (8), 2262–72, doi:10.1016/j.neuropsychologia.2009.12.022 (2010).
- Nunez, J. Morris Water Maze Experiment. Journal of visualized experiments : JoV. (19), e897, doi:10.3791/897 (2008).
- Bromley-Brits, K., Deng, Y., & Song, W. Morris water maze test for learning and memory deficits in Alzheimer’s disease model mice. Journal of visualized experiments : JoV. (53), e2920, doi:10.3791/2920 (2011).
- Rosenfeld, C. S., & Ferguson, S. A. Barnes maze testing strategies with small and large rodent models. Journal of visualized experiments : JoV. (84), e51194, doi:10.3791/51194 (2014).
- Zlomuzica, a, Viggiano, D., et al. Behavioral alterations and changes in Ca/calmodulin kinase II levels in the striatum of connexin36 deficient mice. Behavioural brain researc. 226 (1), 293–300, doi:10.1016/j.bbr.2011.08.028 (2012).
- Assini, F. L., Duzzioni, M., & Takahashi, R. N. Object location memory in mice: pharmacological validation and further evidence of hippocampal CA1 participation. Behavioural brain researc. 204 (1), 206–11, doi:10.1016/j.bbr.2009.06.005 (2009).
- Heyward, F. D., Walton, R. G., Carle, M. S., Coleman, M. A., Garvey, W. T., & Sweatt, J. D. Adult mice maintained on a high-fat diet exhibit object location memory deficits and reduced hippocampal SIRT1 gene expression. , 98 (1), 25-32, doi: 10.1016/j.nlm.2012.04.005 (2012).
- Kenney, J. W., Adoff, M. D., Wilkinson, D. S., & Gould, T. J. The effects of acute, chronic, and withdrawal from chronic nicotine on novel and spatial object recognition in male C57BL/6J mice. Psychopharmacolog. 217 (3), 353–65, doi:10.1007/s00213-011-2283-7 (2011).
- Oliveira, A. M. M., Hawk, J. D., Abel, T., & Havekes, R. Post-training reversible inactivation of the hippocampus enhances novel object recognition memory. Learning, & memory (Cold Spring Harbor, N.Y.. 17 (3), 155–60, doi:10.1101/lm.1625310 (2010).
- Masana, M.,Dine, J., et al. Cognitive performance and synaptic function are dependent on the resilience-promoting protein DRR1. In preparation
- Masana, M., Su, Y.-A., et al. The stress-inducible actin-interacting protein DRR1 shapes social behavior. Psychoneuroendocrinolog. 48, 98–110, doi:10.1016/j.psyneuen.2014.06.009 (2014).
- Schmidt, M. V, Schülke, J.-P., et al. Tumor suppressor down-regulated in renal cell carcinoma 1 (DRR1) is a stress-induced actin bundling factor that modulates synaptic efficacy and cognition. Proceedings of the National Academy of Sciences of the United States of Americ. 108 (41), 17213–8, doi:10.1073/pnas.1103318108 (2011).
- Masana, M., Jukic, M. M., et al. Deciphering the spatio-temporal expression and stress regulation of Fam107B, the paralog of the resiliencepromoting protein DRR1 in the mouse brain. Neuroscienc. 290:147–158, doi:10.1016/j.neuroscience.2015.01.026 (2015).
- Paxinos, G., & Franklin, K. B. J. Mouse Brain in Stereotaxic Coordinate.. Academic Press: New York, (2008).
- Knapman, a, Heinzmann, J.-M., Hellweg, R., Holsboer, F., Landgraf, R., & Touma, C. Increased stress reactivity is associated with cognitive deficits and decreased hippocampal brain-derived neurotrophic factor in a mouse model of affective disorders. Journal of psychiatric researc. 44 (9), 566–75, doi:10.1016/j.jpsychires.2009.11.014 (2010).
- Kamei, H., Nagai, T., et al. Repeated methamphetamine treatment impairs recognition memory through a failure of novelty-induced ERK1/2 activation in the prefrontal cortex of mice. Biological psychiatr. 59 (1), 75–84, doi:10.1016/j.biopsych.2005.06.006 (2006).
- Mizoguchi, H., Takuma, K., et al. Improvement by minocycline of methamphetamine-induced impairment of recognition memory in mice. Psychopharmacolog. 196 (2), 233–41, doi:10.1007/s00213-007-0955-0 (2008).
- Koike, H., Ibi, D., et al. Behavioral abnormality and pharmacologic response in social isolation-reared mice. Behavioural brain researc. 202 (1), 114–21, doi:10.1016/j.bbr.2009.03.028 (2009).
- Lowery, R. L., & Majewska, A. K. Intracranial injection of adeno-associated viral vectors. Journal of visualized experiments : JoV. (45), e2140, doi:10.3791/2140 (2010).
- Lamirault, L., & Simon, H. Enhancement of place and object recognition memory in young adult and old rats by RS 67333, a partial agonist of 5-HT4 receptors. Neuropharmacolog. 41 (7), 844–53, doi:10.1016/S0028-3908(01)00123-X (2001).
- Rolls, A., Colas, D., et al. Optogenetic disruption of sleep continuity impairs memory consolidation. Proceedings of the National Academy of Sciences of the United States of Americ. 108 (32), 13305–10, doi:10.1073/pnas.1015633108 (2011).
- Bettis, T., & Jacobs, L. F. Sex differences in object recognition are modulated by object similarity. Behavioural brain researc. 233 (2), 288–292, doi:10.1016/j.bbr.2012.04.028 (2012).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved