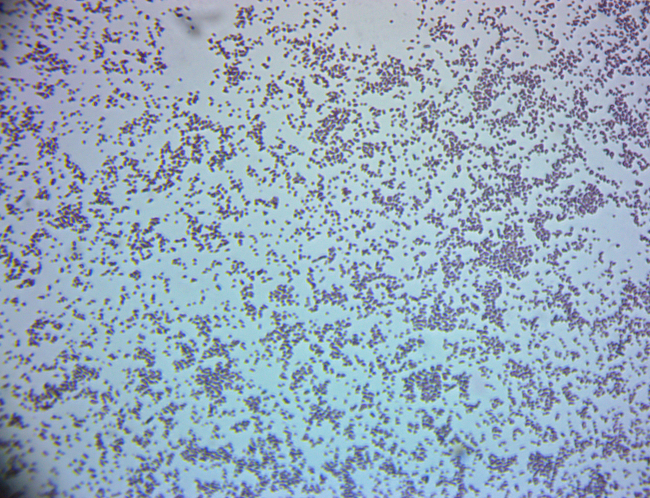
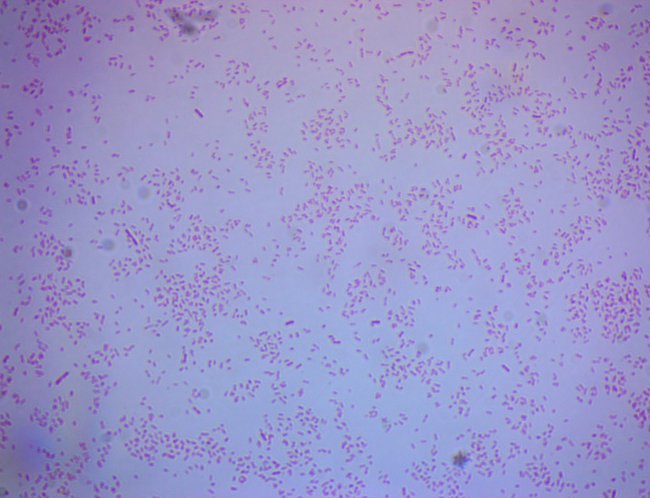
環境ソースから細菌のグラム染色
概要
ソース: ドクターペッパー イアン博士チャールズ Gerba - アリゾナ大学所
示す著者: ルイーザ Ikner
スコープと潜在的な応用環境微生物学研究のスペクトルは広いです。ままかどうか仕事は、ベンチ スケールの知られている細菌の菌株や未知の細菌分離株を含む土壌や水のサンプルを収集するフィールドには、迅速かつ視覚的に金利の人為的集団を識別する能力環境微生物学者たちに大きい輸入の今日でも使用可能な分子技術の豊富な。このビデオは、グラム染色として知られているそのようなの 1 つの手法を説明します。
手順
1. サンプル コレクション
- 微生物分析検査室に土のサンプルとトランスポートを収集します。
- 演習では、10 g サンプル分析用天秤を使用して重量を量る。
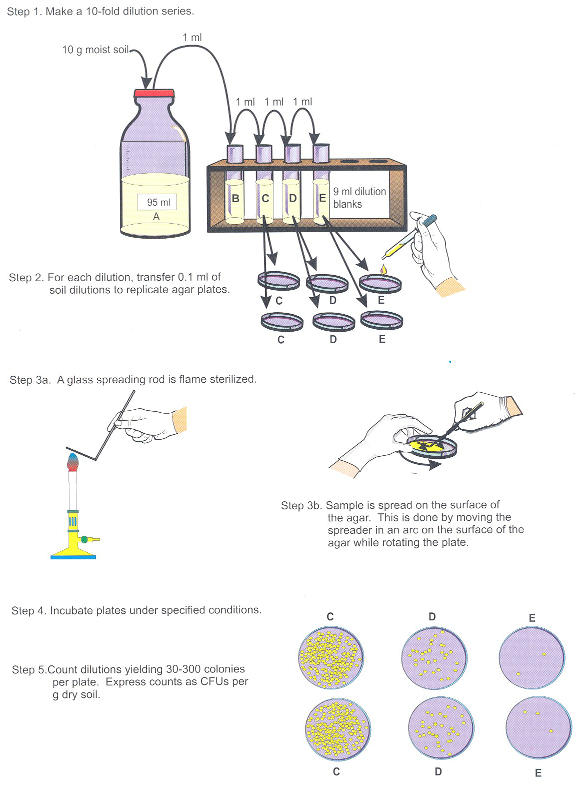
- リン酸緩衝生理食塩水 95 mL に、サンプル 1:10 の希釈 (10 の部分の土壌は 5 パーツ水性液に相当) と (図 2ステップ 1) をミックスする渦。
- その後 1:10 を実行、mL 当たり少なくとも 10-5 g まで希釈土壌し、拡散板 2 つまたは低栄養寒天媒体 (例えばR2A) に 3 つのレプリケートで希釈を選択した (図 2、手順 2-3)。
- (図 2手順 4) 室温で 1 週間の版を孵化させなさい。
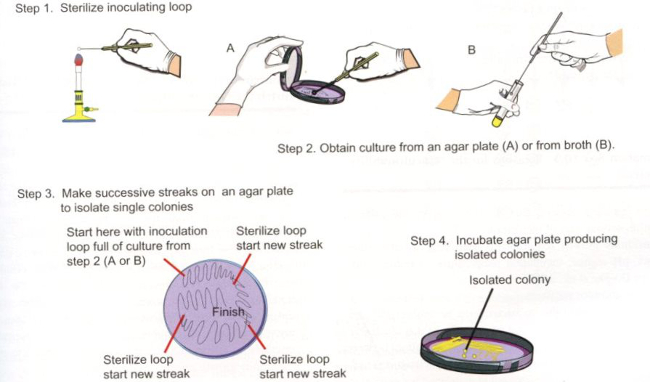
- 隔離、および (図 3、ステップ 1-3) 新鮮な寒天に連勝の 1 つまたは 2 つのコロニーを選択します。
- 縞板室温 (図 3
申請書と概要
タグ
スキップ先...
このコレクションのビデオ:

Now Playing
環境ソースから細菌のグラム染色
Environmental Microbiology
101.0K 閲覧数

土壌中の水分の定量
Environmental Microbiology
360.0K 閲覧数

無菌環境科学
Environmental Microbiology
126.8K 閲覧数

連絡先を介して可視化の土壌微生物スライド法と顕微鏡
Environmental Microbiology
42.8K 閲覧数

糸状菌
Environmental Microbiology
58.8K 閲覧数

細菌のコロニーからコミュニティの DNA の抽出
Environmental Microbiology
29.1K 閲覧数

ポリメラーゼ連鎖反応およびゲル電気泳動法と環境微生物の検出
Environmental Microbiology
44.9K 閲覧数

RT-PCR 法を用いた環境試料の RNA 解析
Environmental Microbiology
40.9K 閲覧数

環境微生物やウイルスを使用しての qPCR の定量化
Environmental Microbiology
48.2K 閲覧数

指標生物による水質分析
Environmental Microbiology
30.1K 閲覧数

ろ過による水試料からの糞便細菌の分離
Environmental Microbiology
39.7K 閲覧数

環境試料中のバクテリオファージの検出
Environmental Microbiology
41.0K 閲覧数

養殖および土壌試料からの細菌を列挙します。
Environmental Microbiology
185.2K 閲覧数

細菌の成長曲線の解析と環境応用
Environmental Microbiology
296.7K 閲覧数

藻類培養手法を介して列挙
Environmental Microbiology
14.1K 閲覧数
Copyright © 2023 MyJoVE Corporation. All rights reserved