조정 화학 단지
Overview
출처: 닐 에이브람스 박사 연구소 — SUNY 환경과학 및 임업 대학
전이 금속은 비타민 보충제에서 전기 도금 목욕에 이르기까지 모든 곳에서 발견됩니다. 전이 금속은 또한 많은 페인트의 안료를 구성하고 모든 미네랄을 구성합니다. 전형적으로, 전이 금속은 쉽게 산화되거나 전자를 잃어버리기 때문에 양이온 형태로 발견되며, 리간드라고 불리는 전자 기증자에 둘러싸여 있습니다. 이러한 리간드금속 센터와 이온 또는 공유 결합을 형성하지 않고 오히려 좌표-공유로 알려진 세 번째 유형의 채권을 수주합니다. 리간드와 금속 간의 좌표-공유 결합은 동적이므로 리간드가 금속 중심 을 지속적으로 교환하고 재조정한다는 것을 의미합니다. 금속과 리간드의 정체성은 리간드가 다른 리간드보다 우선적으로 결합될 것을 지시합니다. 또한, 색상 및 자기 특성은 형성되는 복합체의 종류때문이기도 하다. 형성하는 조정 화합물은 다양한 계측기 및 도구를 사용하여 분석됩니다. 이 실험은 왜 이렇게 많은 복합체가 가능한지 탐구하고 형성되는 조정 복합체의 모형을 확인하는 것을 돕기 위하여 분광화학 (색 및 화학) 방법을 이용합니다.
Procedure
1. 니켈 복합체 및 색상
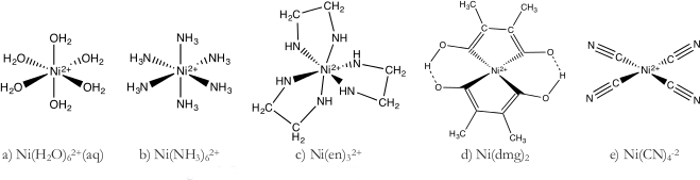
- 니(H2O)62+ 복합체(그림 1a)
- 니소4를 적절한 물량으로 용해하여 Ni(H2O)62+의 1M 용액을 준비합니다.
- 또한 1M 용액의 70mL를 1,000mL의 탈온수에 추가하여 Ni(H2O)62+용액을 희석시켰다.
- 니 (H2O)62 + 일곱 400 mL 비커 중 분할.
- 수성 니켈 용액은 물이 약한 필드 리간드이기 때문에 밝은 녹색 색상을 취합니다.
- 흡광도 스펙트럼은 붉은 파장이 흡수되어 관찰되는 반대, 녹색을 정당화하는 것을 나타냅니다.
- 니(NH3)
Application and Summary
안료에서 사람에 이르기까지, 과도기 금속은 화학, 생물학, 지질학 및 공학 분야 전반에 걸쳐 발견됩니다. 다른 화학 상태 하에서 전이 금속의 동작을 이해하는 것은 색상 또는 자기 행동을 모니터링하는 것만큼 간단할 수 있습니다. 거의 모든 3d (4행) 전이 금속은 생리 적 기능에 필수적이며, 모든 경우에, 이러한 금속은 조정 복합체를 형성하기 위해 리간드에 의해 구속된다. 예를 들어, ?...
References
- Shakhashiri, B. Z.; G. E. Dirreen, G. E; Juergens, F. Color, Solubility, and Complex Ion Equilibria of Nickel (II) Species in Aqueous Solution. J. Chem. Ed. 52 (12), 900-901 (1980).
Tags
건너뛰기...
이 컬렉션의 비디오:

Now Playing
조정 화학 단지
General Chemistry
91.7K Views

일반적인 실험실 유리 제품 및 사용
General Chemistry
658.3K Views

솔루션 및 농도
General Chemistry
275.1K Views

고체 및 액체밀도 결정
General Chemistry
556.8K Views

수성 솔루션에서 질량 백분율 구성 결정
General Chemistry
383.8K Views

경험적 공식 결정
General Chemistry
183.7K Views

이온 화합물의 용해도 규칙 결정
General Chemistry
141.6K Views

pH 미터 사용
General Chemistry
346.7K Views

적정 소개
General Chemistry
425.4K Views

이상적인 가스 법
General Chemistry
79.0K Views

평형 상수의 분광측정 결정
General Chemistry
158.7K Views

르 샤텔리에의 원리
General Chemistry
265.8K Views

알 수 없는 화합물을 결정 하는 동결 포인트 우울증
General Chemistry
160.8K Views

금리법 및 대응 순서 결정
General Chemistry
196.3K Views

차동 스캐닝 열량측정을 사용하여 엔탈피의 변화를 측정합니다.
General Chemistry
44.7K Views
Copyright © 2025 MyJoVE Corporation. 판권 소유