Aby wyświetlić tę treść, wymagana jest subskrypcja JoVE. Zaloguj się lub rozpocznij bezpłatny okres próbny.
Method Article
Green and Low-cost Production of Thermally Stable and Carboxylated Cellulose Nanocrystals and Nanofibrils Using Highly Recyclable Dicarboxylic Acids
W tym Artykule
Podsumowanie
Here we demonstrate a novel method for green and sustainable productions of highly thermally stable and carboxylated cellulose nanocrystals (CNC) and nanofibrils (CNF) using highly recyclable solid dicarboxylic acids.
Streszczenie
Here we demonstrate potentially low cost and green productions of high thermally stable and carboxylated cellulose nanocrystals (CNCs) and nanofibrils (CNF) from bleached eucalyptus pulp (BEP) and unbleached mixed hardwood kraft pulp (UMHP) fibers using highly recyclable dicarboxylic solid acids. Typical operating conditions were acid concentrations of 50 - 70 wt% at 100 °C for 60 min and 120 °C (no boiling at atmospheric pressure) for 120 min, for BEP and UMHP, respectively. The resultant CNCs have a higher thermal degradation temperature than their corresponding feed fibers and carboxylic acid group content from 0.2 - 0.4 mmol/g. The low strength (high pKa of 1.0 - 3.0) of organic acids also resulted in CNCs with both longer lengths of approximately 239 - 336 nm and higher crystallinity than CNCs produced using mineral acids. Cellulose loss to sugar was minimal. Fibrous cellulosic solid residue (FCSR) from the dicarboxylic acid hydrolysis was used to produce carboxylated CNFs through subsequent mechanical fibrillation with low energy input.
Wprowadzenie
Sustainable economic development requires not only using feedstocks that are renewable and biodegradable but also uses green and environmental friendly manufacturing technologies to produce a variety of bioproducts and biochemicals from these renewable feedstocks. Cellulose nanomaterials, such as cellulose nanocrystals (CNC) and cellulose nanofibrils (CNF), produced from renewable lignocelluloses are biodegradable and have unique mechanical and optical properties suitable for developing a range of bioproducts 1, 2. Unfortunately, existing technologies for producing cellulose nanomaterials are either energy intensive when using pure mechanical fibrillation or environmentally unsustainable due to non-recycling or insufficient recycling of processing chemicals, such as when using the concentrated mineral acid hydrolysis process 3-8 or oxidation methods 9-11. Furthermore, oxidation methods may also produce environmentally toxic compounds by reacting with lignocelluloses. Therefore, developing green manufacturing technologies for producing cellulose nanomaterials is critically important to make full use of the abundant and renewable material - lignocelluloses.
Using acid hydrolysis to dissolve hemicellulose and depolymerize cellulose is an effective approach for producing cellulose nanomaterials. Solid acids have been used for sugar production from cellulose with the advantage of easing acid recovery 12, 13. Previous studies using concentrated mineral acids indicated that a lower acid concentration improved CNC yield and crystallinity 3, 5. This suggests that a strong acid may damage cellulose crystals while a milder acid hydrolysis might improve the properties and yield of cellulose nanomaterials through the approach of integrated production and CNC with CNF 3, 14. Here we document a method using concentrated solid dicarboxylic acids hydrolysis to produce CNC along with CNF 15. These dicarboxylic acids have low solubility at low or ambient temperatures, and therefore they can be easily recovered through the mature crystallization technology. They also have good solubility at elevated temperatures which facilitates concentrated acid hydrolysis without boiling or using pressure vessels. Since these acids also have a higher pKa than typical mineral acids used for CNC production, their use results in good CNC crystallinity, and despite lower CNC yields, with a substantial amount of fibrous cellulosic solid residue (FCSR or partially hydrolyzed fibers) remaining due to incomplete cellulose depolymerization. The FCSR can be used to produce CNF through subsequent mechanical fibrillation using low energy inputs. Therefore, cellulose loss to sugars is minimal as compared to using mineral acids.
It is well known that carboxylic acids can esterify cellulose through Fisher-Speier esterification 16. Applying dicarboxylic acids to cellulose can result in semi-acid un-crosslinked esters 17 (or carboxylation), to produce carboxylated CNC and CNF as we demonstrated 15 previously. The method documented here can produce carboxylated and thermally stable CNF and CNC that is also highly crystalline from either bleached or unbleached pulps while having relatively simple and high chemical recovery and using low energy inputs.
Protokół
NOTE: Bleached eucalyptus kraft pulp (BEP) and unbleached mixed hardwood kraft pulp (UMHP) fibers from commercial sources were used as feedstock for producing CNC and CNF. Commercial maleic acids purchased were used for hydrolysis. Hydrolysis conditions were acid concentrations of 60 wt% at 100 °C for 60 min and 120 °C (no boiling at atmospheric pressure) for 120 min, for BEP and UMHP, respectively.
1. Preparation of Concentrated Dicarboxylic Acid Solution
- Heat 40 mL deionized (DI) water in a multiple-neck flask in a liquid glycerol bath on a heating plate to approximately 85 °C.
- Add 60 g anhydrous maleic acid into the flask to make a 60 wt% solution with magnetic stirring. Using the densities of the acid solutions reported previously 15, calculate the required amount of water and acid for making the acid solution at the specified mass concentration.
- Heat the solution to the desired hydrolysis temperature of 100 or 120 °C (no boiling due to high dicarboxylic acid concentrations).
2. Hydrolysis Reaction
- Once the acid solution is at temperature, add 10 g oven dried (OD) of BEP or UMHP fibers into 80 mL dicarboxylic acid solution (1.1) with continuous stirring.
- Take an aliquot of the acid hydrolysate (approximately 2 mL) at the end of the predetermined reaction time of 60 min before terminating hydrolysis by adding 160 mL of 80 °C DI water.
- Dilute 0.5 mL of the sampled hydrolysate for sugar and acid concentration analyses. 15 Observe the remaining hydrolysate sample for crystallization taking place while cooing down to room temperature.
- Separate the hydrolysate from the hydrolyzed pulp by vacuum filtration using a filter paper in a Buchner funnel.
NOTE: This separation needs to occur rather quickly before the temperature drops and the acid begins to crystallize out of solution. Due to high ionic strength from the acid solution, the CNC's generated in hydrolysis agglomerate and stay with the FCSR residuals. Approximately 80-90% of the acid will be removed with the filtrate.
3. CNC Separation
- Wash the filtered solids from section 2 using DI water and dilute to approximately 1% total solids with DI water. Centrifuge the filtrate at 11,960 x g for 10 min.
- Decant off the supernatant. Repeat the washing and filtration process using fresh DI water until the supernatant is turbid. The turbidity indicates that the ionic strength of the solution has fallen enough for the CNC to disperse and start to become colloidal.
- Mix the turbid supernatant with the settled hydrolyzed pulp (2.3). Dialyze the mixture in a dialysis bag (MWCO 14 kDa) using DI water until the conductivity of the liquid approaches that of DI water. Measure the conductivity using a conductance meter.
- Centrifuge the dialyzed sample at 3,500 x g for 10 min to obtain a CNC dispersion in the aqueous phase. Retain the precipitate phase, i.e., FCSR, for CNF production.
- Determine CNC yield from the measured amount of CNC in the dispersion using a COD method described previously 3, 18.
4. CNF Production
- Determine the yield of precipitated FCSR by gravimetric measurements after separating the CNC dispersion. Dry the FCSR at 105 °C and measure the oven dry weight of the FCSR relative to the initial oven dry weight of BEP or UMHP fibers used.
- Mechanically fibrillate the FCSR at fiber suspension of 0.5% by consecutively passing the suspension 3 times through a 200 µm orifice chamber followed by 2 times through an 87 µm orifice chamber, all at 100 MPa.
5. Atomic Force Microscopy (AFM) Imaging
- Sonicate approximately 0.01 wt% CNC or CNF suspensions for 2 min. Deposit a drop of the dispersed suspension on a mica substrate. Air-dry the deposited suspension at ambient temperature.
- Take AFM images of the air-dried CNCs and CNFs in vibrating tapping mode using manufacturer's protocol. Analyze the AFM images of approximately 100 individual CNCs or CNFs using commercial software to obtain diameter and length distributions.
6. Fourier Transform Inferred (FTIR) Measurements
- Use a commercial FTIR spectrophotometer with a universal attenuated-total-reflection (ATR) probe to analyze the resultant CNC and CNF samples along with the original BEP and UMHP fibers to identify ester groups.
- Record the absorption spectra of the samples in a wavelength range between 450 - 4,000 cm-1 with a resolution of 4 cm-1 and 4 scans for each sample.
7. Conductometric Titration
- Use conductometric titration to quantify the carboxyl group contents of samples resulted from esterification by the dicarboxylic acid.
- Add CNC or CNF suspension with 50 mg (OD) of CNCs or CNFs into 120 mL of 1 mM NaCl solution. Titrate the mixture by adding approximately 0.2 mL of 2 mM NaOH solution at 30 s intervals.
- Measure the conductivity using a conductance meter. Find the inflection point (the lowest point) on the conductivity curve during the course of adding NaOH.
- Calculate the amount of carboxyl groups (mmol/L) based on the consumed NaOH relative to the inflection point using the following equation in which c is the concentration of NaOH solution (mol/L), v is the volume of added NaOH solution (mL), m is the mass of CNCs or CNFs in OD weight (g).
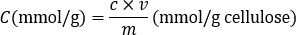
8. CNC and CNF Thermal Stability Determination
- Conduct thermal degradation measurements of the CNC and CNF samples by thermogravimetric analysis (TGA).
- Purge the furnace using a high purity nitrogen flow at 20 mL/min to prevent any unwanted oxidative decomposition. Dry the samples at 50 °C for 4 h before testing. Use a sample size of 5 mg in dry weight.
- Record the weight of the sample as the furnace temperature is increased from ambient to 600 °C at a heating rate of 10 °C/min.
- Normalize the measured weight loss by the initial weight.
- Conduct separate thermal stability tests of CNC and CNF samples in an oven at 105 °C. Record the color change of the samples after 4 and 24 h by conventional photography.
9. X-ray Diffraction
- Press freeze-dried CNC or CNF samples at 180 MPa to make pellets as described previously. 5 Conduct wide angle X-ray diffraction measurements of the pellet using Cu-Kα radiation on an X-ray diffractometer in the 2θ range of 10 - 38° in steps of 0.02°.
- Calculate the crystallinity index (CrI) of a pellet using the Segal method 19 (without base line subtraction).
Wyniki
Typical AFM images of the CNC and CNF from BEP and UMHP along with corresponding SEM images of the feed acid hydrolyzed fibers are shown in Figures 1 and 2. The images clearly show the substantial reductions in fiber length by acid hydrolysis with minimal change in fiber diameters (comparing Figure 1a with 1b, and 2a with 2b). The shortened fiber length was also reflected by the measured cellulose degree of polymerizatio...
Dyskusje
The thicker CNC diameters of the CNC samples from maleic acid hydrolysis resulted in a moderate average aspect ratio 7.24 and 8.53, for the CNCs from BEP and UMHP, respectively, despite their long lengths as discussed above. The CNFs had a longer length and a thinner diameter, which resulted in a large aspect ratio of 13.9 and 19.0, for the CNCs from BEP and UMHP, respectively, both greater than their respective CNCs. It is possible to use severe mechanical fibrillation to reduce CNF diameter to improve the aspect ratio ...
Ujawnienia
Chen and Zhu are co-inventors of a US patent application using dicarboxylic acids for CNC and CNF production.
Podziękowania
This work was conducted while Bian, Chen, and Wang were visiting Ph.D. students at the US Forest Service, Forest Products Laboratory (FPL), Madison, WI, and on official government time of Zhu. This work was partially supported by the USDA Agriculture and Food Research Initiative (AFRI) Competitive Grant (No. 2011-67009-20056), the Chinese State Forestry Administration (Project No. 2015-4-54), the National Natural Science Foundation of China (Project No. 31470599), Guangzhou Elite Project of China, and China Scholarship Fund. Funding from these programs made the visiting appointments of Bian, Chen, and Wang at FPL possible.
Materiały
| Name | Company | Catalog Number | Comments |
| Bleached eucalypus pulp | Aracruz Cellulose | ||
| Unbleached mixed hardwood kraft pulp | International Paper | ||
| Maleic acid | Sigma-Aldrich | M0375-1KG/CAS110-16-7 | Powder; assay: 99.0% (HPLC) |
| Glycerol | Sigma-Aldrich | G5516-4L/CAS56-81-5 | |
| Sodium hydroxide | Fisher Scientific | S318-500/CAS1310-73-2, 497-19-8 | Certified ACS |
| Sodium chloride | Mallinckrodt | 7581-12/CAS7647-14-5 | Crystal,AR |
| Cupriethylenediamine solution | GFS Chemicals | E32103-1L/CAS14552-35-3 | 1 M, for determination of solution viscosity of pulps |
| Acetone | Fisher Scientific | A18-500/CAS67-64-1 | Certified ACS |
| Accu-TestTM Vials for COD Testing | Bioscience,Inc. | 01-215-28 | COD testing for 20 to 900 mg/L standard range concentration |
| Heating plate | IKA | Mode: C-MAD HS7 digital | |
| Magnetic stir bar | ACE Glass | ||
| Pyrex three-neck round-bottom flask | Sigma-Aldrich | CLS4965B500-1EA | |
| Dialysis tubing cellulose membrane | Sigma-Aldrich | D9402-100FT | Typical molecular weight cut-off = 14,000 kDa |
| Disposable aluminum dishes | Sigma-Aldrich | Z154857-1PAK | Circles, 60 mm |
| Disintegrator | Testing Machines Inc.(TMI) | ||
| Microfluidizer | Microfluidics Corporation | ||
| Sonicator | Qsonica LLC. | Mode: 3510R-MT, 50-60 Hz, 180 W | |
| Zeta potential analyzer | Brookhaven Instruments Corporation | ||
| FTIR | PerkinElmer | ||
| Conductometric titrator | Yellow Springs Instrument (YSI) | ||
| TGA analyzer | PerkinElmer | ||
| X-ray diffractometer | Bruker Corporation | ||
| AFM imging | AFM Workshop | ||
| SEM imaging | Carl Zeiss |
Odniesienia
- Giese, M., Blusch, L. K., Khan, M. K., MacLachlan, M. J. Functional Materials from Cellulose-Derived Liquid-Crystal Templates. Angew Chem Int Ed. 54 (10), 2888-2910 (2015).
- Zhu, H., et al. Wood-Derived Materials for Green Electronics, Biological Devices, and Energy Applications . Chem. Rev. , (2016).
- Wang, Q. Q., et al. Approaching zero cellulose loss in cellulose nanocrystal (CNC) production: recovery and characterization of cellulosic solid residues (CSR) and CND. Cellulose. 19 (6), 2033-2047 (2012).
- Hamad, W. Y., Hu, T. Q. Structure-process-yield interrelations in nanocrystalline cellulose extraction. Can J Chem Eng. 88 (3), 392-402 (2010).
- Chen, L. H., et al. Tailoring the yield and characteristics of wood cellulose nanocrystals (CNC) using concentrated acid hydrolysis. Cellulose. 22 (3), 1753-1762 (2015).
- Mukherjee, S. M., Woods, H. J. X-ray and electron microscope studies of the degradation of cellulose by sulphuric acid. Biochim Biophys Acta. 10 (4), 499-511 (1953).
- Camarero Espinosa, S., Kuhnt, T., Foster, E. J., Weder, C. Isolation of thermally stable cellulose nanocrystals by phosphoric acid hydrolysis. Biomacromolecules. 14 (4), 1223-1230 (2013).
- Yu, H. Y., et al. Facile extraction of thermally stable cellulose nanocrystals with a high yield of 93% through hydrochloric acid hydrolysis under hydrothermal conditions. J Mater Chem, A. 1 (12), 3938-3944 (2013).
- Leung, A. C. W., et al. Characteristics and properties of carboxylated cellulose nanocrystals prepared from a novel one-step procedure. Small. 7 (3), 302-305 (2011).
- Saito, T., Isogai, A. TEMPO-mediated oxidation of native cellulose. The effect of oxidation conditions on chemical and crystal structures of the water-insoluble fractions. Biomacromolecules. 5 (5), 1983-1989 (2004).
- Yang, H., Chen, D. Z., van de Ven, T. G. M. Preparation and characterization of sterically stabilized nanocrystalline cellulose obtained by periodate oxidation of cellulose fibers. Cellulose. 22 (3), 1743-1752 (2015).
- Huang, Y. B., Fu, Y. Hydrolysis of cellulose to glucose by solid acid catalysts. Green Chem. 15 (5), 1095-1111 (2013).
- Shimizu, K. I., Satsuma, A. Toward a rational control of solid acid catalysis for green synthesis and biomass conversion. Energy & Environ Sci. 4 (9), 3140-3153 (2011).
- Wang, Q. Q., Zhu, J. Y., Considine, J. M. Strong and optically transparent films prepared using cellulosic solid residue (CSR) recovered from cellulose nanocrystals (CNC) production waste stream. ACS Appl Mater Interfaces. 5 (7), 2527-2534 (2013).
- Chen, L. H., Zhu, J. Y., Baez, C., Kitin, P., Elder, T. Highly thermal-stable and functional cellulose nanocrystals and nanofibrils produced using fully recyclable organic acids. Green Chem. 18, 3835-3843 (2016).
- Fischer, E., Speier, A. Darstellungder der Ester. Chemische Berichte. 28 (3), 3252-3258 (1895).
- Allen, T. C., Cuculo, J. A. Cellulose derivatives containing carboxylic acid groups. J Polym Sci: Macromol Rev. 7 (1), 189-262 (1973).
- Wang, Q. Q., Zhao, X. B., Zhu, J. Y. Kinetics of strong acid hydrolysis of a bleached kraft pulp for producing cellulose nanocrystals (CNCs). Ind Eng Chem Res. 53 (27), 11007-11014 (2014).
- Segal, L., Creely, J. J., Martin, A. E., Conrad, C. M. An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J. 29 (10), 786-794 (1959).
Przedruki i uprawnienia
Zapytaj o uprawnienia na użycie tekstu lub obrazów z tego artykułu JoVE
Zapytaj o uprawnieniaThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. Wszelkie prawa zastrzeżone