Method Article
Free Radicals in Chemical Biology: from Chemical Behavior to Biomarker Development
In This Article
Summary
Radical-based biomimetic chemistry has been applied to building-up libraries necessary for biomarker development.
Abstract
The involvement of free radicals in life sciences has constantly increased with time and has been connected to several physiological and pathological processes. This subject embraces diverse scientific areas, spanning from physical, biological and bioorganic chemistry to biology and medicine, with applications to the amelioration of quality of life, health and aging. Multidisciplinary skills are required for the full investigation of the many facets of radical processes in the biological environment and chemical knowledge plays a crucial role in unveiling basic processes and mechanisms. We developed a chemical biology approach able to connect free radical chemical reactivity with biological processes, providing information on the mechanistic pathways and products. The core of this approach is the design of biomimetic models to study biomolecule behavior (lipids, nucleic acids and proteins) in aqueous systems, obtaining insights of the reaction pathways as well as building up molecular libraries of the free radical reaction products. This context can be successfully used for biomarker discovery and examples are provided with two classes of compounds: mono-trans isomers of cholesteryl esters, which are synthesized and used as references for detection in human plasma, and purine 5',8-cyclo-2'-deoxyribonucleosides, prepared and used as reference in the protocol for detection of such lesions in DNA samples, after ionizing radiations or obtained from different health conditions.
Introduction
The reactivity of free radicals revealed its enormous importance for many biological events, including aging and inflammation. Nowadays, it is more and more evident that the clarification of each chemical step involved in this reactivity is needed, in order to understand the underlying mechanisms and envisage effective strategies for the control of free radicals and repair of the damage. The contribution from chemical studies is fundamental, but the direct study in the biological environment can be difficult, since the superimposition of different processes complicates and perturbs the examination of the results and the related conclusions. Therefore, the strategy of modeling free radical reactions under biologically related conditions has become a fundamental step in the research of chemical mechanisms in biology.
In the last decade our group developed models of free radical processes under biomimetic conditions. In particular we envisaged biologically relevant transformations of unsaturated fatty acids, nucleosides, and sulfur-containing amino acids and put them in the track to be evaluated and validated as biomarkers of health status.1-4
Our general approach consists of three modules:
- Organic synthesis, which provides access to appropriately modified biomolecules. The synthetic plan can be also designed in order to simulate the free radical processes occurring in the biological environment, thus applying conditions able to simulate the biological process of interest, which is the principle of biomimetic radical chemistry. In biomimetic models the reaction medium is water or a heterogeneous medium due to the coexistence of hydrophilic and hydrophobic compartments. The advantage to work with biomimetic models is to have a simplified environment with known reaction partners, where reactivity and products can be examined in more details, possibly discovering novel chemical pathways. From this step mechanistic and kinetic information can be gathered;
- Purification, analysis and characterization of the products, providing structural and chemical information of the modified biomolecules in order to organize molecular libraries and facilitate recognition.in the more complex biological environment. Protocols are established also in view of resolving complex mixtures of compounds such as those derived from biological specimens;
- Biomarker development using biological samples, derived either by in vitro experiments using cell cultures, and in vivo studies from animals and humans. The protocols developed in biomimetic models are then applied to the complex analysis of samples, to envisage the free radical transformations under different conditions. Molecular libraries developed by synthesis are of enormous help to life science discoveries. The information gathered on the free radical transformations give the opportunity to figure out repair and prevention strategies. Databases of the results can be used for a careful evaluation by multivariate analysis of the biomarker significance as well as possible associated factors.
We chose two classes of relevant biomarkers to accredit this approach: cholesteryl esters and purine 5',8-cyclo-2'-deoxyribonucleosides.
Protocol
1. Synthesis of Mono-trans Isomers of Cholesteryl Esters
- Dissolve cholesteryl esters (linoleate or arachidonate cholesteryl ester) in 2-propanol (15 mM) . For a better solubilization, the samples were sonicated for 15 min under argon.
- Transfer the solution to a quartz photochemical reactor, add 2-mercaptoethanol in 2-propanol (to reach 7 mM concentration, from a 2 M stock solution of the thiol). Flush the reaction mixture with argon for 20 min in order to eliminate the presence of oxygen in the solution.
- Irradiate the reaction mixture by UV light using a 5.5W low-pressure mercury lamp at 22±2 °C for 4 min. Monitor by analytical Ag-TLC (silver-thin layer chromatography) to evidence the formation of the mono-trans cholesteryl esters (see Figure 1 for the chemical formulas). The TLC staining is carried out by pouring the plate in the cerium ammonium molibdate (CAM) solution, and the spots appear on heating the plate. Quench the reaction at an early stage, in order to recover the starting material, that can be reused to perform other rounds of isomerization, obtaining an increase of the overall yield.
- Collect the reaction mixture in a round-bottom flask, washing the apparatus with a few ml of 2-propanol. Remove solvent and purify the mono-trans isomers of cholesteryl esters by Ag-TLC as described in the literature.5 Use hexane-diethyl ether (9:1 v/v) as the eluent for mono-trans cholesteryl linoleate isomers, whereas use hexane-diethyl ether-acetic acid (9:1:0,1 v/v) for mono-trans cholesteryl arachidonate isomers.
2. Isolation of Cholesteryl Esters Fraction from Human Serum
- Dilute 1 ml of human serum (obtained by centrifugation of blood) with 1 ml of brine and pour the solution into a separatory funnel under a stream of argon in order to avoid artifacts (e.g. oxidation adducts). Add 10 ml chloroform-methanol (2:1 v/v) thrice. Shake the separatory funnel very slowly in order to limit the formation of the emulsion due to the presence of albumin.
- Wash the organic layers once with brine (10 ml), then collect in an Erlenmeyer flask under an argon flow, dry over anhydrous sodium sulphate. Remove volatiles by rotary evaporator to afford a yellow oil (total plasma lipid fraction).
- Take up the crude with 1 ml of chloroform-methanol (2:1 v/v), load onto a preparative TLC under a stream of argon and use a mixture of hexane-diethyl ether (9:1 v/v) as the eluent. Scrape off the silica portion containing the cholesteryl ester fraction and pour into a vial. Then, extract silica (3 × 5 ml) with chloroform-methanol (2:1 v/v), collect the organic layers and remove the solvent after evaporation to afford the pure fraction of cholesteryl esters (usually ~1.5 mg).
- Keep cholesteryl esters in a dark vial covered by aluminum foil under Argon and store at -20 °C. Cholesteryl esters are sensitive to light and oxygen.
3. Characterization of Mono-trans Cholesteryl Esters by Raman Spectroscopy
- Extract cholesteryl esters from human serum (≥0.7 mg) as described in section 2, dissolve in a small amount of carbon tetrachloride (≤10 μl) and place in a vial. CCl4 is selected as the solvent because its Raman signal does not overlap with the region of interest of cholesteryl ester analysis.
- Transfer the solution to the sample holder through a disposable glass pipette, then carefully remove the solvent by a slow stream of argon. Once a uniform oily film is formed on the internal wall of the sample holder, place the latter into the instrument for measurement.
- Fourier Transformed Raman spectrum of cholesteryl esters is directly obtained on the lipid extracts without any derivatization reaction. The laser power on the sample is <100 mW to avoid sample damage. The total number of scans for each spectrum is ≥800 to minimize the background noise.
- The 1,700-1,630 cm-1 range of the Raman spectrum is analysed since it reflects the contribution from the C=C double bonds present in the molecule (C=C stretching mode). A curve fitting analysis is performed on this region, thus allowing the distinguishing of the superimposed contributions from cis and trans double bonds in the fatty alkyl chain and double bond in the ring B of cholesterol (see Figure 2).
- To fit correctly the vibrational bands, some parameters should be fixed or constrained within reasonable limits. The component peak profiles are described as a linear combination of Lorentzian and Gaussian functions, whereas the half-height bandwidths are best determined by the computer optimisation routine. The number and position of the component peaks are obtained by using the fourth derivative spectra, which allow to detect also some weak component bands. Since the signal to noise ratio which deteriorates upon differentiation of the signal can preclude the use of the fourth derivative, smoothed fourth derivatives with a thirteen-point Savitsky-Golay function, are of advantage; thus, a right compromise between resolution and signal to noise ratio is obtained. Best curve fitting are obtained at the lowest possible c2 values.
- The presence of trans isomers is revealed by the component at 1671±1 cm-1, in addition to, at least, the two components, slightly shifted towards lower frequencies, due to the fatty acid chain and cholesterol C=C double bonds. Representative Raman spectrum of plasma cholesteryl ester is shown in Figure 9. In the inset the comparison of a specific region with cholesteryl linoleate and mono-trans cholesteryl linoleic acid isomers is shown.
4. Derivatization to Fatty Acid Methyl Esters (FAME) and Analysis by Gas Chromatography
- Dissolve cholesteryl esters (~1.5 mg) with 0.5 ml of a 1 M solution of sodium hydroxide in benzene-methanol (2:3 v/v) and place in a dark vial covered by aluminum foil under argon.
- Leave the mixture stirring at room temperature and monitor by TLC using a mixture of hexane-diethyl ether (9:1 v/v) as the eluent. Reaction time is usually 30 min but a careful reaction monitoring is required. In fact, once the corresponding methyl esters are formed, the reaction must be quenched in order to avoid the formation of degradation and side products. TLC showed the quantitative formation of methyl esters.
- Quench the reaction mixture by adding 1 ml of brine and extract methyl esters with n-hexane (3 × 2 ml). Collect the organic layers into a vial and remove the solvent by rotary evaporation to afford the methyl ester fraction, which is then used for GC analysis without further purification.
- Dissolve the methyl esters in the minimum quantity of n-hexane (usually 1 mg in 50 μl) and inject in the GC equipped with a 60 m × 0.25 mm × 0.25 μm (50%-cyanopropyl)-methylpolysiloxane column. Use a flame ionization detector (FID) with the following oven program: temperature start at 165 °C, hold for 3 min, followed by increase of 1 °C/min up to 195 °C, hold for 40 min, followed by a second increase of 10 °C/min up to 240 °C, and hold for 10 min. A constant pressure mode (29 psi) is chosen. Helium or hydrogen can be used as the carrier gas. Methyl esters are identified by comparison with the retention times of authentic samples. Figure 10 shows a representative GC analysis of FAME obtained from plasma cholesteryl esters.
5. Synthesis of (5'R)- and (5'S)-5',8-cyclo-2'-deoxyadenosine
- Dissolve 33 mg of 8-bromo-2'-deoxyadenosine (8-Br-dAdo) in acetonitrile (100 ml) in order to reach 1 mM concentration. Regulate gas flow (Ar or N2) in the photoreactor and fill the apparatus with the solution of 8-Br-dAdo.
- Prepare a septum with the appropriate size for the photoreactor's input and pass a needle connected to the inert gas line through the center of it. Close the system with the septum. Flush the inert gas for 30 min.
- Irradiate by UV light using a 125W medium-pressure mercury lamp at 22±2 °C for 15 min, collect the solution into a 250 ml round-bottomed flask. Wash the photoreactor with 10 ml acetonitrile and collect washings in the same flask.
- Quench the crude reaction mixture with 1 M NH4OH solution and evaporate the solvent in the rotary evaporator.
- Run high performance liquid chromatographic analysis (HPLC-UV) with analytical column (see instrumentation section) under known conditions , and compare the profile with reference compounds.
- Run high performance liquid chromatographic analysis (HPLC-UV) with an analytical column (see instrumentation section) by using the following solvents and gradients: 2 mM ammonium formate as solvent A and acetonitrile as solvent B. Set the flow rate to 1 ml/min and the gradient from 0% solvent B to 0.3% solvent B to be reached in 2.2 min. Then 0.8% solvent B in in 4.0 min, then 1% solvent B in 4.8 min. Remain at 1% solvent B for 9 min and then go to 8% solvent B in 6 min. After go to 10 % solvent B in 4 min and to 30% solvent B in 5 min and remain at 30% solvent b for other 5 min. Collect the chromatographic peaks corresponding to the two diastereomeric products in two different vials.
- Measure the absorbance of the two collected fractions in the UV spectrophotometer and calculate the exact concentration based on the Lamber-Beer law (A = εlC, where A is the absorbance, ε the extinction coefficient and l the length of the cell). Use the extinction coefficient ε of 2'-deoxyadenosine (dAdo) (15,400 M-1 cm-1 at 260 nm).
6. Synthesis of (5'R)- and (5'S)-5',8-cyclo-2'-deoxyguanosine
- Dissolve 35 mg of 8-bromo-2'-deoxyguanosine (8-Br-dGuo) and 30 mg of sodium iodide (NaI) in distilled water (100 ml) in order to reach 1mM and 2mM concentration, respectively. Regulate inert gas (Ar or N2) flow connected to the photoreactor and fill the photoreactor with the reaction solution.
- Degass the reaction mixture as described in procedure 5 (step b).
- Irradiate by UV light using a 125W medium-pressure mercury lamp at 22±2 °C for 30 min, collect the solution into a 250 ml round-bottomed flask. Wash the photoreactor with 10 ml of water and collect washings in the same flask.
- Quench the crude reaction mixture with 1 M NH4OH solution and evaporate the solvent under vacuum.
- Perform analysis as described in Procedure 5 (steps e to g). Use the extinction coefficient ε of 2'-deoxyguanosine (dGuo) (11,700 M-1 cm-1 at 260 nm).
7. Synthesis of Isotopic Labeled Purine (5'R)- and (5'S)-5',8-cyclonucleosides
- Prepare 2 ml of 1mM aqueous solution (distilled water) of the 15N isotopic labeled purine nucleosides ([15N5]dAdo or [15N5]dGuo) in a glass vial (4 ml) with a septum cap.
- Connect the vial with the septum cap and connect to the gas line (N2O for saturation of the solution) through a needle in the septum that reaches the bottom of the vial. Another short needle in the septum cap works as the gas exit.
- Flush the solution with N2O for 30 min. The gas flow is regulated to be very low.
- The exit needle is first taken out and after 1' the long one follows, in order to have a small pressure inside the vial.
- Put the solution in the apparatus of gamma radiolysis for 8 hr (calculation on the basis of a dose rate ca. 4.5 Gy/min).
- After reaction quench the crude with 1 M NH4OH solution.
- Run high performance liquid chromatographic analysis (HPLC-UV) under known conditions,6 and compare the chromatogram with the standard references.
8. Gamma Radiolysis of DNA Aqueous Solutions
- Prepare 1 ml of 0.5 mg/ml solution (distilled water) of calf thymus DNA and put in a glass vial (2 ml) with a septum cap.
- Degass the reaction as described in Procedure 7 (steps b-d).
- Put the solution in the apparatus of gamma radiolysis for 30 min (calculation on the basis of dose/rate ca. 4.5 Gy/min).
9. Enzymatic Digestion of DNA
- To an Eppendorf tube containing 80 μg of DNA, add 8 U of nuclease P1, 0.01 U of phosphodiesterase II, 20 nmol of EHNA in 20 μl of 300 mM sodium acetate (pH 5.6) and 10 mM zinc chloride. Incubate the mixture at 37°C for 48 hr.
- Next add a mixture of 8 U of alkaline phosphatase, 0.02 U of phosphodiesterase I, in 40 μl of 0.5 M Tris-HCl buffer (pH 8.9) to mixture digestion. The sample is incubated at 37°C for 2 hr.
- The mixture is neutralized by addition of 10% formic acid.
- Transfer the sample to an Amicon Ultra-0.5 centrifugal filter device (cutoff 3 kDa), add 200 μl of tridistilled H2O. Place the Ultra-filter into the centrifuge rotor at 14,000 x g for 15 min. Then separate the Ultra filter device from the microcentrifuge tube. Lyophilize the enzyme-free solution in the microcentrifuge tube.
10. DNA Sample Desalinization Prior to Analysis
- Run high performance liquid chromatographic analysis (HPLC-UV) with analytical column (see instrumentation section) following known conditions.6
- Dissolve the lyophilized digested DNA in 20 μl of H2O and inject in HPLC-UV.
- Collect the sample in a vial after the salt elution (first minutes) and right before the elution time of the first nucleoside (5'R-cdGuo), until the end of the chromatographic program.
- The sample is lyophilized and resolubilized in 20 μl water.
11. LC-MS/MS Quantitative Analysis
- Prepare the HPLC-MS/MS (triple quadrupole) before starting the analysis by loading the analytical method and the method for the MS detector (ESI).6
- Add 1 μl of the isotopic labelled compounds mixture in the sample prepared in Procedure 7.
- Inject 20 μl of the spiked digested DNA solution in distilled water.
- The obtained chromatographic data are elaborated on the basis of previous calibration with reference compounds. A representative HPLC run containing adenosine and guanosine 2'-deoxyribonucleosides and their oxidative and cyclo derivatives is shown in Figure 11.
Results
The isomerization process has been described in particular for cholesteryl esters affording the mono-trans isomers of linoleic and arachidonic acids shown in Figure 1 as the first products of this attack, which can occur under conditions of free radical stress in the biological environment.5
In Figure 3 the chemical mechanism involved in the cis-trans double bond isomerization is shown. The radical source is indicated in a generic way to afford S-centered radicals. In the described protocols the radical source is UV light that is able to break homolitically the S-H bond present in the thiol molecule RSH.
Figure 4 summarizes the three steps protocol for the synthesis of the modified lipid class and their detection in human plasma: the synthesis represents a biomimetic free radical process and also provides a one-pot convenient entry to the geometrical isomers, without any contamination by positional isomers followed by purification and isolation protocols.
Several analytical methodologies can be applied for a high sensitive detection of the trans isomer content and characterization of the mono-trans cholesteryl ester library. In particular, Raman spectroscopy can be carried out directly on the cholesteryl ester fraction without derivatization (see Figure 2 and Figure 9).
The second example concerns purine 5',8-cyclo-2'-deoxyribonucleosides, which are DNA lesions created by the attack of free radicals to the position C5' of the sugar moiety and subsequent formation of a covalent bond between the sugar and the base moieties. Four structures can be produced, that is, 5',8-cyclo-2'-deoxyadenosine and 5',8-cyclo-2'-deoxyguanosine, both existing in the 5'R and 5'S diastereomeric forms (Figure 5). In Figure 6 the reaction mechanism is shown, that involves photolysis of 8-bromopurine derivatives 5 to give the corresponding C8 radical 6, which intramolecularly abstracts a hydrogen atom from the C5' position selectively affording the 2'-deoxyadenosin-5'-yl radical 7. Radical 7 undergoes cyclization with a rate constant in the range 105-106 s-1,2 followed by oxidation of the heteroaromatic radical 8 to give the final products 9. The reaction of hydroxyl radicals, generated by radiolysis of water, with 2′-deoxyadenosine and 2′-deoxyguanosine (10) was found to occur ca. 10% by hydrogen abstraction from C5′ position.7
Our chemical biology approach for the libraries of purine 5',8-cyclo-2'-deoxyribonucleosides (including labeled compounds) is illustrated in Figure 7, with the identification of these lesions in oligonucleotides as well as in DNA samples obtained from various sources such as, for example, treated under ionizing radiation conditions, as mimic of radical stress conditions.
In Figure 8 a typical photoreactor equipment is shown. The device allows for irradiation of compounds dissolved in the appropriate solvent. It consists of: i) the reaction chamber equipped with the inlet for the inert gas (on the bottom) and two inlets for the gas exit and for the reagents addition; ii) an internal chamber containing the appropriate mercury lamp connected to a cooling system and electrical power, which is inserted into the reaction chamber through a glass joint.
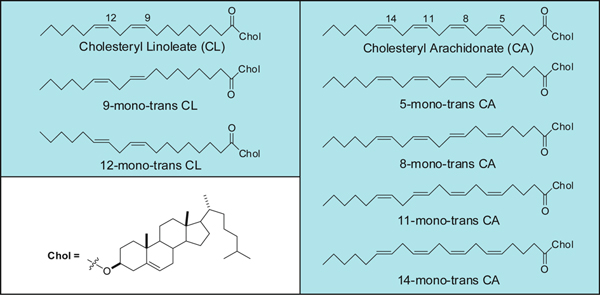
Figure 1. Mono-trans isomers of cholesteryl linoleate and arachidonate.
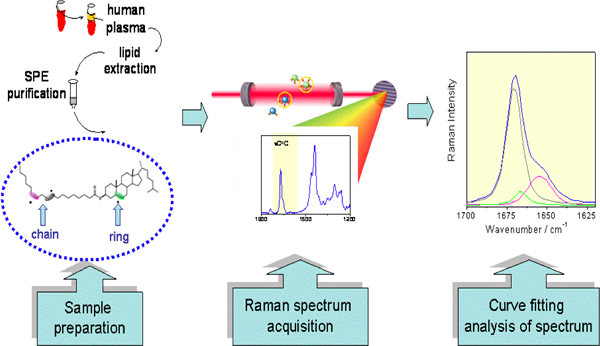
Figure 2. Cholesteryl esters in human serum and direct analysis for mono-trans isomers by Raman spectroscopy.
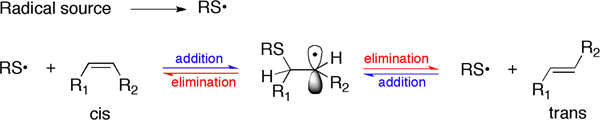
Figure 3. The addition-elimination process that leads to the cis-trans isomerization of double bonds by thiyl radicals.
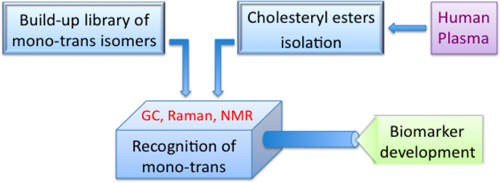
Figure 4. Three steps protocol for development of mono-trans cholesteryl esters as biomarker of free radical stress.
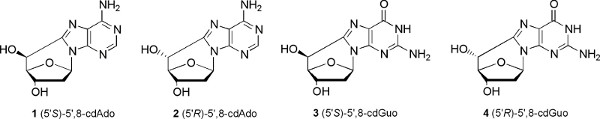
Figure 5. The four purine 5',8-cyclo-2'-deoxyribonucleoside diastereoisomers. Click here to view larger figure.
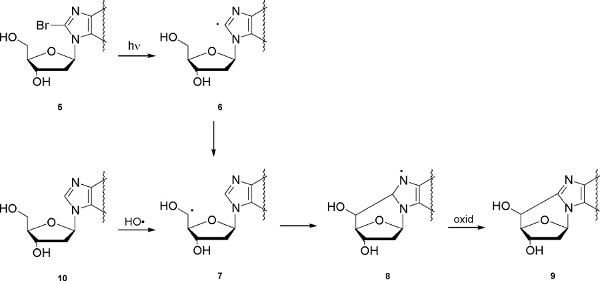
Figure 6. Generation of C5' radicals, either by photolysis of 5 or by reaction of 10 with HO• radicals, and the mechanism of purine 5',8-cyclo-2' deoxyribonucleoside formation. Click here to view larger figure.
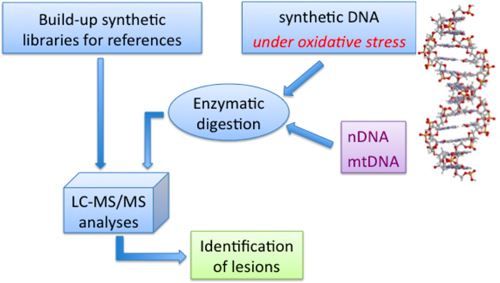
Figure 7. Protocol for identification of some radical-based DNA lesions; mtDNA: mitochondrial DNA, nDNA: nuclear DNA.

Figure 8. Photochemical reactor.
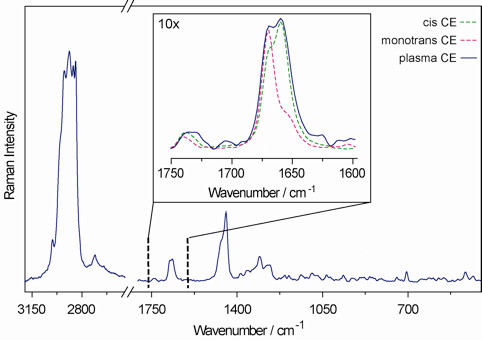
Figure 9. Representative Raman spectrum of plasma cholesteryl ester. In the inset the comparison of a specific region with cholesteryl linoleate and cholesteryl mono-trans linoleic acid isomers.
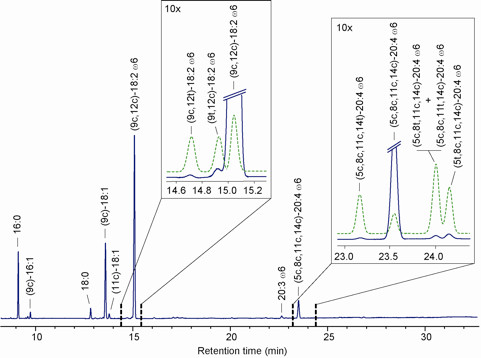
Figure 10. Representative GC analysis of FAME obtained from plasma cholesteryl esters.
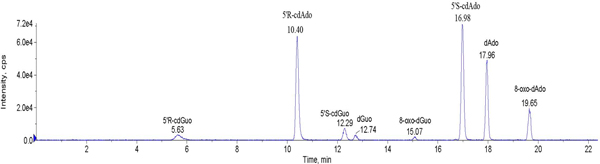
Figure 11. Representative HPLC run containing 2'-deoxyadenosine and 2'deoxyguanosine together with their oxidative and purine 5',8-cyclo-2'-deoxyribo nucleosides. Click here to view larger figure.
Discussion
The conversion of naturally occurring cis unsaturated fatty acids to geometrical trans isomers is a transformation connected with the production of radical stress in the biological environment. Cell membrane lipids, which contain fatty acids, are a relevant biological target for radical stress and we first studied the endogenous cis-trans phospholipids isomerization in cell cultures, animals and humans assessing analytical protocols in each case.8-10 We demonstrated that this transformation can occur by a variety of S-containing compounds, including thiols, thioethers and disulfides, which under different radical stress conditions are able to generate thiyl radicals, i.e. the isomerizing agent (Figure 3). The example shown in this article focuses on the class of cholesteryl esters, which represent a well-known fraction of plasma lipids, strictly involved in lipoprotein metabolism. The ester linkage between fatty acids and cholesterol is biosynthesized by the transfer of fatty acids from the position 2 of the glycerol moiety of phosphatidylcholine to cholesterol, a step catalyzed by the enzyme lecithin cholesterol acyl transferase (LCAT). Therefore, plasma cholesteryl esters are strictly connected with membrane lipid turnover, and contain relatively high proportions of the polyunsaturated fatty acids (PUFA) typically present in phosphatidylcholines, i.e. linoleic and arachidonic acids. Lipoprotein formation is involved in cardiovascular and metabolic diseases. The reactivity of natural cholesteryl esters with free radicals can occur at the double bonds of linoleate and arachidonate residues, which can be transformed in the corresponding trans geometrical isomers (see Figure 1 for the structures). Characterization of the trans cholesteryl ester content in biological samples is interesting for biomarker development. An indirect methodology consists of the transformation of cholesteryl esters isolated from plasma to the corresponding fatty acid methyl esters (FAME) and separation by gas chromatographic protocols. In this case, the calibration of the standard references of cis and trans fatty acid methyl esters is performed, in order to allow the quantitation of the trans content in the samples. Based on the analytical studies carried out on the cholesteryl ester library, we proposed to apply also a method based on Raman spectroscopy, that can be carried out directly on the cholesteryl ester fraction isolated from plasma, without further derivatization to the corresponding FAME (see Figures 2 and 9). It is worth noting that up to now no successful methods are described to separate cis and trans isomers of fatty acid containing lipids by HPLC, as instead described by cholesteryl ester hydroperoxides. So far, the indirect gas chromatographic method is still the best available method so far. By this method the first quantitative evaluation of the mono-trans content derived from cholesteryl esters isolated from plasma of healthy subjects was provided. Using Flame Ionization Detector (FID) the limit of detectability is satisfactory (ppb) and nanomolar quantities of the compounds have been detected.5. With different detection systems this limit can be even lowered. The effect of ionizing radiation on cholesteryl esters is matter of further studies, whether a linear response is obtained relative to the applied dose.
As a second example, we chose modified nucleosides which can be produced by free radical damage of DNA. Hydroxyl radicals (HO• ) are known to be the most harmful Reactive Oxygen Species (ROS) for their ability to cause chemical modifications to DNA. Single or multiple lesions may occur on DNA, that in eukaryotic cells is located in nucleus and mitochondria. Identification and measurement of the main classes of oxidative generated damages to DNA require the appropriate molecular libraries in order to set up the analytical protocols. We focused our interest on the smallest tandem lesions, which are purine 5',8-cyclo-2'-deoxyribonucleosides, having an additional covalent bond between the base and the sugar moieties created by the free radical attack. The compounds are 5',8-cyclo-2'-deoxyadenosine and 5',8-cyclo-2'-deoxyguanosine existing in the 5'R and 5'S diastereomeric forms (Figure 5). Their potential to become free radical stress marker is matter of fundamental research.2 Indeed, when DNA is exposed to HO• radical hydrogen abstraction from C5' position of the sugar is one of the possible events leading to the formation of these tandem lesions. Purine 5′,8-cyclonucleosides can be measured as sum of diastereomers by HPLC-MS/MS in enzymatically digested γ-irradiated DNA samples varying from 1 to 12 lesions /106 nucleosides/Gy going form absence of oxygen to physiological level of oxygen in tissues, the diastereomeric ratio 5'R/5'S being ~4 and ~3 for 5',8-cdAdo and 5',8-cdGuo, respectively (Figure 11).11 It is worth noting that the relationship between radiation dose and 5',8-cdAdo and 5',8-cdAdo lesions detected in cellular DNA is far from being understood. The single experiment based on 2kGy irradiation reported in the experimental cannot be considered conclusive.11 Further experiments of this kind and analytical quantitation of the four lesions are necessary for defining such relationship. The detection of these lesions and the more popular oxidative transformations (such as 8-oxo-2'-deoxyguanosine, 8-oxodGuo) are matter of intense investigations, evidencing the importance of both lesions during oxidative metabolism.6,13 The use of HPLC-MS/MS (triple quadrupole) has a detection limit close to 30fmol for all four lesions. Last improvements are claimed to reach detection limits of attomol levels by the instrument producers. Based on the very recent literature,6 analytical procedures have to include the proper cleanup of the sample and enrichment in order to meet the detection limits of the MS/MS/MS (ion trap) or of the MS/MS (triple quadrupole) used in our case.
Bio-inspired synthetic procedures of compounds 1-4 were developed starting from 8-bromopurine derivatives under or photolysis.7,12 These procedures involve a radical cascade reaction that mimics the DNA damage mechanism of formation of 5',8-cdAdo and 5',8-cdGuo lesions. From biological perspectives, it was found that these lesions accumulate with aging in a tissue-specific manner (liver>kidney>brain), providing evidence that DNA repair mechanisms are inadequate to preserve the genetic material from these lesions.13 Indeed, nucleotide excision repair (NER) is the only pathway currently identified for the repair of these lesions.2
The two classes of compounds shown in Figures 1 and 5 are not commercially available at the moment, however by the synthetic strategies described in the literature it would not be difficult to prepare these compounds for commercial use.
The multidisciplinary approach provided by chemical biology studies not only has an enormous value in the identification of novel mechanisms occurring in the biological environment, but also gives a fundamental contribution to biomarker discovery and diagnostics, ultimately bringing novelty in health care and prevention strategies.14 The chemical contribution is needed for a successful development of molecular medicine, creating integrated platforms and panels for metabolic profiling which are expected to allow for an optimal rationalization of intervention design, either therapeutic and nutritional, reducing uncertainties and failures when they can be predictable.
Disclosures
No conflicts of interest declared.
Acknowledgements
Financial support from the Ministero dell'Istruzione, dell'Universitá della Ricerca (PRIN-2009K3RH7N_002) and Marie Curie Intra-European Fellowship (CYCLOGUO-298555) as well as the sponsorship of COST Action CM0603 on 'Free Radicals in Chemical Biology and COST Action CM1201 on "Biomimetic Radical Chemistry" are gratefully acknowledged.
Materials
| Name | Company | Catalog Number | Comments |
| MATERIALS | |||
| Cholesteryl linoleate ≥98% | Sigma-Aldrich | C0289-100 mg | |
| Cholesteryl arachidonate≥95% | Sigma-Aldrich | C8753-25mg | |
| 2-mercapt–thanol | Sigma-Aldrich | M6250-100 ml | |
| 2-propanol | Sigma-Aldrich | 34965-1L | |
| Methanol 215 SpS | Romil | H409-2,5 L | |
| Ethanol | Sigma-Aldrich | 02860-2.5L | |
| Chloroform SpS | Romil | H135 2,5 L | |
| n-Hexane 95% SpS | Romil | H389 2,5 L | |
| Acetonitrile 230 SpS | Romil | H047 2,5 L | |
| Dichloromethane SpS | Romil | H2022,5 L | |
| Carbon tetrachloride | Sigma-Aldrich | 107344-1L | |
| Sodium iodide | Sigma-Aldrich | 383112-100G | |
| Sodium hydrogen carbonate | Carlo Erba | 478536-500 g | |
| Diethyl ether | Sigma-Aldrich | 309966-1L | |
| NaCl | Sigma-Aldrich | S7653-5KG | |
| NaOH solid | Sigma-Aldrich | 221465-25G | |
| NH4OH sol. 28%-30% | Sigma-Aldrich | 221228-1L-A | |
| Acetic acid | Sigma-Aldrich | 320099-500ML | |
| Ammonium Cerium(IV)sulfate dihydride | Sigma-Aldrich | 221759-100G | |
| Ammonium Molybdate tetrahydrate | Sigma-Aldrich | A7302-100G | |
| Sulfuric Acid 95%-98% | Sigma-Aldrich | 320501-1L | |
| Silver Nitrate | Sigma-Aldrich | 209139-25G | |
| Sodium sulfate anhydrous | Sigma-Aldrich | 238597-500G | |
| Nuclease P-I from penicillium citrinum | Sigma-Aldrich | N8630-1VL | |
| Phosphodiesterase II type I-sa | Sigma-Aldrich | P9041-10UN | |
| Erythro-9-(2-hydroxy-3-nonyl)adenine, hc | Sigma-Aldrich | E114-25MG | |
| Phosphatase alkaline type VII-t from*bov | Sigma-Aldrich | P6774-1KU | |
| Phosphodiesterase I type VI | Sigma-Aldrich | P3134-100MG | |
| Deoxyribonuclease II type IV from*porcin | Sigma-Aldrich | D4138-20KU | |
| Trizma(r) base, biotechnology performanc ce | Sigma-Aldrich | T6066-100G | |
| EDTA | Sigma-Aldrich | E1644-100G | |
| Succinic acid bioxtra | Sigma-Aldrich | S3674-250G | |
| Calcium chloride | Sigma-Aldrich | C5670-100G | |
| Formic acid, 98 % | Sigma-Aldrich | 06440-100ML | |
| Amicon Ultra-0.5 Centrifugal Filter Unit with Ultracel-3 membrane | Millipore | UFC500324 | |
| 8-Bromo-2'-deoxyguanosine | Berry Associates | PR3290-1 g | |
| 8-Bromo-2'-deoxyadenosine | Berry Associates | PR3300-1 g | |
| Sodium iodide | Sigma-Aldrich | 383112-100G | |
| Sodium hydrogen carbonate | Carlo Erba | 478536-500 g | |
| 2'-deoxyguanosine:H2O (U-15N5, 96-98%) | Cambridge Isotope Laboratories, Inc | CILNLM-3899-CA-0.1 | |
| 2'-deoxyadenosine (U-15N5, 98%) 95%+ CHEMICAL PURITY | Cambridge Isotope Laboratories, Inc | CILNLM-3895-0.1 | |
| Nitrous oxide (N2O) | Air Liquide | ||
| Deoxyribonucleic acid from calf thymus | Sigma Aldrich | D4522-5MG | |
| EQUIPMENT | |||
| 60Co-Gammacell | AECL- Canada | 220 | |
| Immersion well reaction medium pressure 125 watts | Photochemical reactors ltd | Model 3010 | |
| Evaporating flask 250 ml | Heidolph | P/N NS 29/32 514-72000-00 | |
| Luna 5 μm C18(2) 100 Å, LC Column 250 x 4.6 mm | Phenomenex | 00A-4252-E0 | |
| Alltima C8 Column 250 x 10 mm 5 μm | Grace | 88081 | Semipreparative |
| SecurityGuard Kit | Phenomenex | KJ0-4282 | Analytical holder kit and accessories |
| Holder for 10.0 mm ID cartridges | Phenomenex | AJ0-7220 | Semipreparative holder |
| 10.0 mm ID cartridges | Phenomenex | AJ0-7221 | |
| High-performance liquid chromatography (HPLC) | Agilent | 1100 | |
| LC/MS/MS | Applied Biosystems | 4000QTRAP System | |
| Tandem mass ESI spectrometer | (Bruker Daltonics) | Esquire 3000 plus | |
| Vial 2-4 ml | SUPELCO | Cod 27516 | |
| Vial 4 ml | SUPELCO | Cod 27517 | |
References
- Ferreri, C., Chatgilialoglu, C. Membrane lipidomics and the geometry of unsaturated fatty acids: from biomimetic models to biological consequences. Methods Molecular Biology. 579, 391-412 (2009).
- Chatgilialoglu, C., Ferreri, C., Terzidis, M. A. Purine 5',8-cyclonucleoside lesions: chemistry and biology. Chemical Society Reviews. 40 (3), 1368-1382 (2011).
- Chatgilialoglu, C., Ferreri, C., et al. Radiation-induced reductive modifications of sulfur-containing amino acids within peptides and proteins. Journal of Proteomics. 74 (11), 2264-2273 (2011).
- Chatgilialoglu, C., Ferreri, C. Trans lipids: the free radical path. Accounts Chemical Research. 38 (6), 441-448 (2005).
- Melchiorre, M., Torreggiani, A., Chatgilialoglu, C., Ferreri, C. Lipid markers of geometrical radical stress: synthesis of mono-trans cholesteryl esters isomers and detection in human plasma. Journal of the American Chemical Society. 133 (38), 15184-15190 (2011).
- Wang, J., Yuan, B., Guerrero, C., Bahde, R., Gupta, S., Wang, Y. Quantification of Oxidative DNA Lesions in Tissues of Long-Evans Cinnamon Rats by Capillary High-Performance Liquid Chromatography-Tandem Mass Spectrometry Coupled with Stable Isotope-Dilution Method. Analytical Chemistry. 83 (6), 2201-2209 (2011).
- Boussicault, F., Kaloudis, P., Caminal, C., Mulazzani, Q. G., Chatgilialoglu, C. The fate of C5' radicals of purine nucleosides under oxidative conditions. Journal of the American Chemical Society. 130 (26), 8377-8385 (2008).
- Ferreri, C., Kratzsch, S., Brede, O., Marciniak, B., Chatgilialoglu, C. Trans lipids formation induced by thiols in human monocytic leukemia cells. Free Radicals Biology & Medicine. 38 (9), 1180-1187 (2005).
- Zambonin, L., Ferreri, C., et al. Occurrence of trans fatty acids in rats fed a trans-free diet: a free radical-mediated formation. Free Radicals Biology & Medicine. 40 (9), 1549-1556 (2006).
- Puca, A. A., Andrew, P., et al. Fatty acids profile of erythrocyte membranes as possible biomarker of longevity. Rejuvenation Research. 11 (1), 63-72 (2008).
- Belmadoui, N., Boussicault, F., et al. Radiation-induced formation of purine 5',8-cyclonucleosides in isolated and cellular DNA: high stereospecificity and modulating effect of oxygen. Organic & Biomolecular Chemistry. 8 (14), 3211-3219 (2010).
- Chatgilialoglu, C., Bazzanini, R., Jimenez, L. B., Miranda, M. A. (5'S)- and (5'R)-5',8-cyclo-2'-deoxyguanosine: mechanistic insights on the 2'-deoxyguanosin-5'-yl radical cyclization. Chemical Research in Toxicology. 20 (12), 1820-1824 (2007).
- Wang, J., Clauson, C. L., Robbins, P. D., Niedernhofer, L. J., Wang, Y. The oxidative DNA lesions 8,5'-cyclopurines accumulate with aging in a tissue-specific manner. Aging Cell. 11 (4), 714-716 (2012).
- Ferreri, C., Chatgilialoglu, C. The role of fatty acid-based functional lipidomics in the development of molecular diagnostic tools. Expert Review of Molecular Diagnostics. 12 (7), 767-780 (2012).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved