免疫荧光显微镜:石蜡内嵌组织部分的免疫荧光染色
Overview
资料来源:托马斯·查菲1,托马斯·格里菲斯2,3,4,凯瑟琳·施韦特费格1,3,4
1明尼苏达大学明尼阿波利斯分校实验室医学和病理学系,MN 55455
2明尼苏达大学泌尿科,明尼阿波利斯,MN 55455
3共济会癌症中心,明尼苏达大学明尼阿波利斯分校,MN 55455
4明尼苏达大学明尼阿波利斯分校免疫学中心,MN 55455
组织部分的病理分析可用于更好地了解正常组织结构,并有助于我们了解疾病机制。组织活检,无论是从患者还是从实验性体内模型,通常通过固定在甲醛或甲醛和嵌入石蜡中来保存。这允许长期存储和组织被分割。使用微缩图,将组织切成薄(5 μm)部分,并且这些截面粘附在玻璃玻片上。组织部分可以沾染抗体,从而检测组织部分的特定蛋白质。与荧光素(也称为氟铬)结合的抗体染色 - 当激光激发时以特定波长发出光的化合物 - 称为免疫荧光。检测部分内蛋白质的能力可以提供信息,如细胞类型异质性在组织内,激活特定信号通路,和生物标志物的表达。根据所使用的荧光光和可用于分析的显微镜类型,可以使用多种颜色,从而可以多路分析目标。
以下协议概述了石蜡嵌入式组织部分的免疫荧光染色所涉及的基本步骤。需要注意的是,该协议将不包括关于组织固定、石蜡嵌入过程或组织切片的任何细节。一旦组织被分割并放置在玻璃幻灯片上,它们就会通过一系列分级乙醇(EtOH)孵育物进行补水。这些部分用阻塞试剂孵育,以减少抗体与组织部分的非特异性结合。然后,用可能或可能不会直接标记荧光剂的原抗体孵育这些部分。如果原抗体没有直接标记,则用标有荧光剂的次级抗体孵育这些部分。不同的抗体可能需要不同的染色条件,因此包括优化抗体的建议。洗涤后去除所有未结合的抗体后,将幻灯片与含有DAPI的介质安装,以荧光标记细胞核。安装介质干燥后,可以使用显微镜使用激光对幻灯片进行成像,激光可以检测不同的荧光道。
Procedure
1. 设置
- 典型的染色协议涉及以下步骤:
- 使用一系列分级乙醇对幻灯片上的组织部分进行重新保湿。
- 用阻塞缓冲液孵育组织部分,这将有助于阻断抗体与组织的非特异性结合,并减少背景荧光。
- 去除阻塞缓冲液,在原抗体中孵育该部分,此时抗体将结合其肽靶点。
- 去除原抗体,在洗涤缓冲液中广泛清洗各部分。
- 如果原抗体没有直接标记有荧光团,并且需要二级抗体,则用二级抗体孵育部分,以便与原抗体结合。
- 将次级抗体从滑轨上洗掉。
- 在安装介质中安装幻灯片,使其在荧光显微镜可视化之前干燥。
- 需要以下物品:滑动支架(玻璃或塑料)、罐、移液器、纸笔、潮湿室、盖玻片以及带 DAPI 的安装介质。
- 二甲苯用于幻灯片的补液。二甲苯是危险的,应与适当的PPE一起使用,包括手套和实验室外套。
- 缓冲剂、溶液和试剂的配方
-
分级乙醇
乙醇 - 160 mL 200
-
分级乙醇
Results
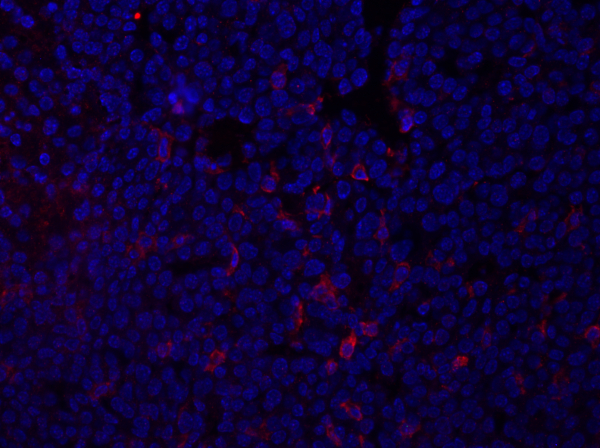
图1:乳腺肿瘤部分的F4/80染色。固定后,小鼠乳腺肿瘤被分割,并沾染抗F4/80,并使用含有DAPI的安装介质安装。染色由细胞表面F4/80红色染色显示。请点击此处查看此图的较大版本。
Application and Summary
References
- Im K, Mareninov S., Diaz MFP, Yong WH. An Introduction to Performing Immunofluorescence Staining. Yong W. (eds) Biobanking. Methods in Molecular Biology. 1897, Humana Press, New York, NY (2019)
- Ramos-Vara JA. Principles and Methods of Immunohistochemistry. Gautier JC. (eds) Drug Safety Evaluation. Methods in Molecular Biology. 1641, Humana Press, New York, NY (2017)
- Donaldson JG. Immunofluorescence Staining. Current protocols in Cell Biology. 69 (1):1 4.3.1-4.3.7. (2015)
Tags
跳至...
此集合中的视频:

Now Playing
免疫荧光显微镜:石蜡内嵌组织部分的免疫荧光染色
Immunology
53.8K Views

流细胞学和荧光活化细胞分拣(FACS):血性B淋巴细胞的分离
Immunology
92.9K Views

磁性活细胞分拣 (MACS): 胸腺 T 淋巴细胞的分离
Immunology
22.9K Views

ELISA Asas :间接、三明治和竞争
Immunology
238.3K Views

ELISPOT 分析:检测 IFN-- 分泌性细胞
Immunology
28.5K Views

免疫组织化学和免疫细胞化学:通过光显微镜进行组织成像
Immunology
78.9K Views

抗体生成:使用杂交瘤生产单克隆抗体
Immunology
43.5K Views

共聚焦荧光显微镜:确定小鼠纤维细胞中蛋白质定位的技术
Immunology
43.2K Views

基于免疫沉淀的技术:使用阿加玫瑰珠纯化内源性蛋白质
Immunology
87.7K Views

细胞周期分析:使用CFSE染色和流细胞测定法评估刺激后CD4和CD8 T细胞增殖
Immunology
24.2K Views

收养细胞转移:将供体小鼠孢子细胞引入宿主小鼠,并通过FACS评估成功
Immunology
22.3K Views

细胞死亡测定:细胞毒性能力的铬释放测定
Immunology
151.4K Views
版权所属 © 2025 MyJoVE 公司版权所有,本公司不涉及任何医疗业务和医疗服务。