11.10 : Epossidazione di Sharpless
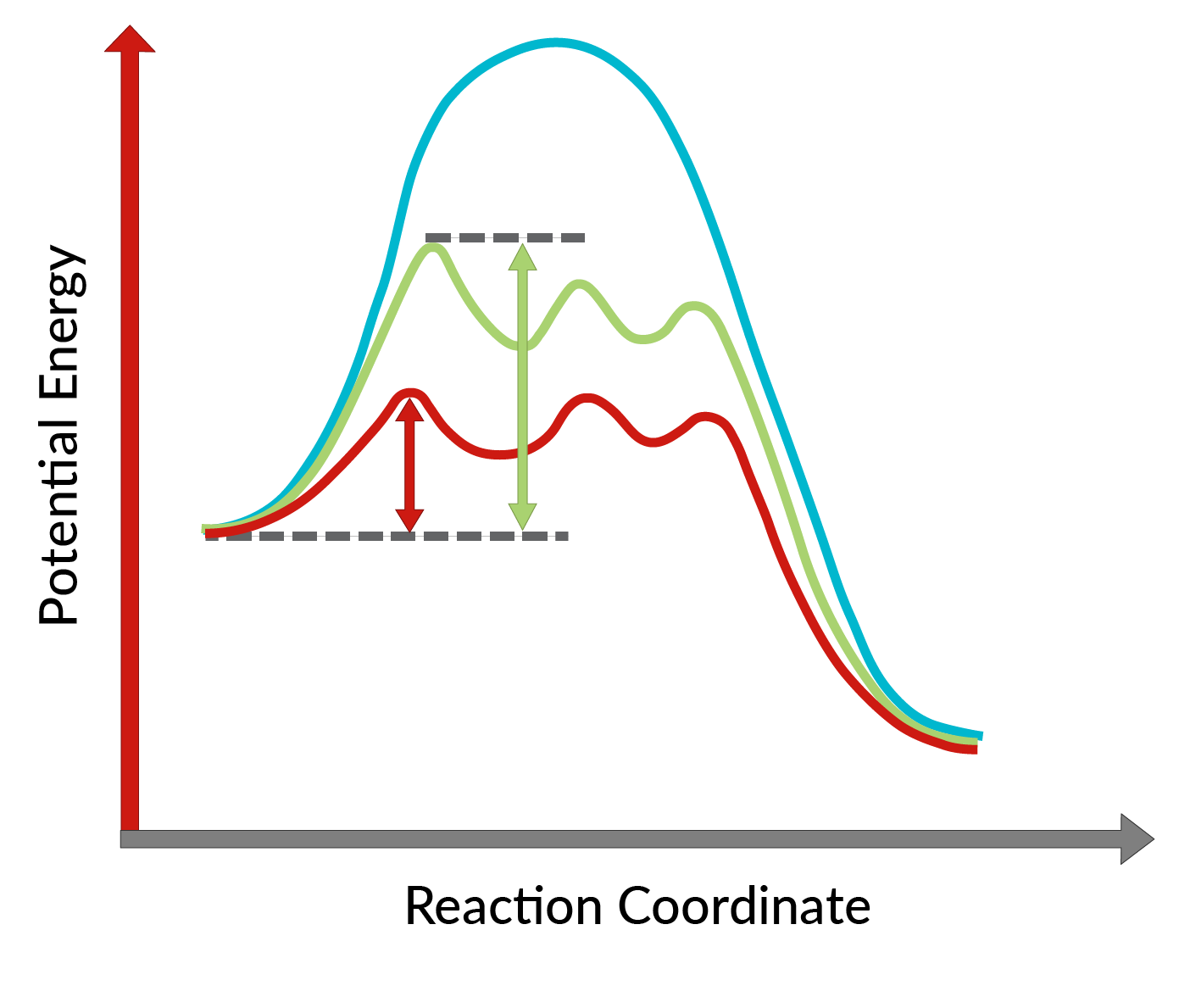
La conversione degli alcoli allilici in epossidi utilizzando il catalizzatore chirale è stata scoperta da K. Barry Sharpless ed è nota come epossidazione di Sharpless. L'utilizzo di un catalizzatore chirale consente la formazione di un enantiomero del prodotto in eccesso. Questo catalizzatore chirale è principalmente un complesso chirale di tetraisopropossido di titanio ed estere tartrato (stereoisomero specifico). Lo stereoisomero utilizzato nel catalizzatore chirale determina la formazione dell'enantiomero del prodotto. In altre parole, l'uso del L-(+)-dietil tartrato porta ad enantiomeri con l'anello epossidico sotto il piano, mentre con il D-(−)-dietil tartrato, ad enantiomeri con l'anello epossidico sopra il piano. L'elevata enantioselettività della reazione può essere spiegata considerando le energie di attivazione richieste affinché la reazione proceda nella direzione in avanti in presenza del catalizzatore chirale. Come mostrato in Figura 1, rispetto alla reazione non catalizzata (curva blu), l'energia di attivazione della reazione diminuisce drasticamente con l'aggiunta del catalizzatore chirale (curve rossa e verde). Inoltre, l'energia di attivazione per la formazione di un enantiomero (curva rossa) si abbassa maggiormente rispetto a quella di un altro enantiomero (curva verde), portando alla formazione di un enantiomero in eccesso. Pertanto, la reazione di epossidazione di Sharpless può essere utilizzata per la sintesi degli enantiomeri desiderati del prodotto.
La stereochimica del prodotto formato, quando qualsiasi alcol allilico è sottoposto a epossidazione di Sharpless, può essere prevista semplicemente orientando la molecola di alcol allilico su un piano con i gruppi idrossilici rivolti verso l'angolo inferiore destro, come mostrato nella Figura 2. Su questa struttura planare, Il D-(−)-dietil tartrato trasporta l'ossigeno dalla faccia superiore dell'alchene, rendendo possibile la formazione dell'epossido dall'alto del piano, mentre l'L-(+)-dietil tartrato trasporta l'ossigeno dalla faccia inferiore dell'alchene, installando in questo modo l'anello epossidico da sotto il piano.
Dal capitolo 11:

Now Playing
11.10 : Epossidazione di Sharpless
Eteri, epossidi e solfuri
3.8K Visualizzazioni

11.1 : Struttura e nomenclatura degli eteri
Eteri, epossidi e solfuri
11.2K Visualizzazioni

11.2 : Proprietà fisiche degli eteri
Eteri, epossidi e solfuri
6.9K Visualizzazioni

11.3 : Dagli alcoli agli eteri: disidratazione degli alcoli e sintesi di Williamson degli eteri
Eteri, epossidi e solfuri
10.2K Visualizzazioni

11.4 : Eteri da alcheni: addizione alcolica e alcossimercurazione-demercurazione
Eteri, epossidi e solfuri
7.7K Visualizzazioni

11.5 : Da eteri ad alogenuri alchilici: scissione acido-catalizzata
Eteri, epossidi e solfuri
5.6K Visualizzazioni

11.6 : Autossidazione di eteri a perossidi e idroperossidi
Eteri, epossidi e solfuri
7.4K Visualizzazioni

11.7 : Gli eteri corona
Eteri, epossidi e solfuri
5.1K Visualizzazioni

11.8 : Struttura e nomenclatura degli epossidi
Eteri, epossidi e solfuri
6.3K Visualizzazioni

11.9 : Preparazione degli epossidi
Eteri, epossidi e solfuri
7.4K Visualizzazioni

11.11 : Apertura dell'anello degli epossidi acido-catalizzata
Eteri, epossidi e solfuri
7.1K Visualizzazioni

11.12 : Apertura dell'anello degli epossidi base-catalizzata
Eteri, epossidi e solfuri
8.3K Visualizzazioni

11.13 : Struttura e nomenclatura di tioli e solfuri
Eteri, epossidi e solfuri
4.6K Visualizzazioni

11.14 : Preparazione e reazioni dei tioli
Eteri, epossidi e solfuri
6.0K Visualizzazioni

11.15 : Preparazione e reazioni dei solfuri
Eteri, epossidi e solfuri
4.7K Visualizzazioni