A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Establishment of a Robust and Reproducible Model of Radiation-Induced Skin and Muscle Fibrosis
* These authors contributed equally
In This Article
Summary
Here we present a protocol to induce radiation-induced skin fibrosis in the hind limb of mice and perform post-irradiation measurements of chronic impairment via limb excursion and gait index analyses to evaluate the functional outcome. The model elucidates radiation-related skin fibrosis mechanisms and is useful in subclinical therapeutic studies.
Abstract
Radiation-induced skin fibrosis (RISF) can result from a plethora of scenarios including cancer therapy, accidental exposure, or acts of terrorism. Radioactive beams can penetrate through the skin and affect the structures in their path including skin, muscles, and internal organs. Skin is the first structure to get exposed to radiation and is susceptible to develop chronic fibrosis, which is challenging to treat. Currently, limited treatment options show moderate efficacy in mitigating radiation-related skin fibrosis. A key factor hindering the development of effective countermeasures is the absence of a convenient and robust model that could allow for translation of the experimental findings to humans. Here, a robust and reproducible murine hind limb skin fibrosis model has been established for prophylactic and therapeutic evaluation of possible agents for functional and molecular recovery.
The right hind limb was irradiated using a single dose of 40 (Gray) Gy to induce skin fibrosis. Subjects developed edema and dermatitis in the early stages proceeded by visible skin constriction. Irradiated limbs showed a significantly reduced limb range of motion in the following weeks. In late stages, acute side effects subsided, yet chronic fibrosis persisted. A gait index was performed as an additional functional assay, which demonstrated the development of functional impairment. These non-invasive methods demonstrated reliable measurements for tracing fibrosis progression, which is supported by histological analyses. The radiation dose, application, and post-irradiation analyses employed in this model offer a vigorous and reproducible method for studying radiation-induced skin fibrosis and testing the efficacy of therapeutical agents.
Introduction
The skin is the largest organ of the body, covering and protecting the body from hazards. It has three distinct layers: epidermis, dermis, and hypodermis. Each layer has its unique functions: the epidermis prevents dehydration and microbial invasion; the dermis has a rich network of cells, and an extracellular matrix that provides tensile strength and elasticity1; the dermal layer contains the sensory receptors, hair follicles, glands, and vessels for lymphatic and capillary networks. The hypodermis or subcutaneous tissue, with its abundance of adipose tissue, contours the body and distributes mechanical stress2,3,4.
Radiation, generated as a result of accidents, war, terrorism, or therapeutical applications, penetrate through the body in a linear progressive nature, leading the skin to be the first organ to come in contact. The threat of such incidents has intensified due to the increased use of radioactive materials in industries, medical facilities, and military installations5. Clinically, radiation damage to the skin is characterized by cutaneous radiation syndrome (CRS), one of four sub-syndromes of acute radiation syndrome (ARS). The response of the skin to ionizing radiation has important implications for treatment and protection from further damage6. Concomitant injuries such as burns and trauma further complicate the clinical outcome when combined with radiation injuries7. The extent of skin exposure to radiation correlates to a point-of-no-return threshold, from which the impairment of other organs results in single or multiple organ failure, and ultimately leads to patient death8,9. Cutaneous radiation injury is comprised of an acute and a chronic phase. Acute radiation injury clinically manifests as erythema, skin edema, dermatitis, blistering, epidermal denudation, dry or wet desquamation, ulceration, and changes in the hair and nails. The chronic phase is manifested as dermal atrophy, fibrosis, chronic ulceration, and telangiectasias10,11. In general, acute effects are predominately manifested in the epidermis, while chronic effects are most prominent in the dermis. Acute reaction to radiation exposure leads to a marked decrease in mitotic activity within 12 h of exposure, followed by hyperemia, cell enlargement, vacuolization, nuclear pyknosis, and fragmentation4,12.
Radiation doses exceeding 40 Gy result in moist desquamation and loss of epidermis, leading to an increased susceptibility to infections13. In addition, skin exposure to radiation induces cytokine production, triggering an inflammatory immune response in the dermal layer. Prominent inflammatory mediators include interleukins (IL-1, IL-3, IL-5, IL-6, and IL-8) and tumor necrosis factor-α (TNFα)14. Failure in the resolution of inflammation can eventually result in fibrosis development at the site of radiation injury15. Additional physical wounds or thermal injuries further aggravate this fibrotic response, extending through the muscle layer16. Transforming growth factor-β (TGFβ) is the key cytokine in fibrosis development17. Currently, very few treatment options show promising results, and the majority might have challenges with patient compliance. Further research exploring the cellular and molecular responses of the skin to different radiation doses will improve the understanding of the radiation-induced skin pathophysiology and enhance the development of new therapies.
To facilitate the clinical translation of research outcomes in preclinical models in alleviating radiation-induced injury to the skin and soft tissues, designing highly relevant experimental models of therapeutic interventions following irradiation is crucial. Both in vitro and in vivo models of radiation-induced injury have been described, including cell culture models of irradiated endothelial cells18,19, fibroblasts20, or keratinocytes19 and in vivo rodent, swine, and non-human primate animal models. Rodent models are widely used in radiation research due to their similarities in response to radiation injury with humans and their flexibility of genetic manipulation21. Radiation dose requirements are higher in rodents than in humans when seeking similar outcomes: desquamation, fibrosis, and necrosis16,22. Description of scoring criteria to measure the response to radiation has further enhanced the adoption of rodent models of radiation skin injury21,23.
Current research in the preclinical setting focuses on understanding the mechanisms of radiation-induced skin injury and developing therapeutical options. Thus, establishing a robust and reproducible preclinical model to create the radiation insult with high clinical translatability is essential. This work describes a murine model of skin fibrosis with optimized radiation dose and delivery technique. Our model, which combines functional, histological, and molecular measurements, can be used to effectively study the mechanism of fibrosis development and investigate new therapeutical options.
Protocol
Ethical animal use was approved by the Institutional Animal Care and Use Committee (IACUC), which acts in compliance with the Animal Welfare Act. Animals were housed in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) approved facility and treated according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
1. Anesthesia
- Place mice in the box of a small animal anesthesia system (Figure 1). Deliver 4% isoflurane to the box and wait for 5-10 min at which time lower the isoflurane to 2-3%.
- Confirm the anesthesia depth via toe pinch. Apply eye lubricant to prevent drying. Move the subject to the nose cone with 2% isoflurane flow.
- Use the above technique to anesthetize each mouse for shaving and limb measurement assay. Use an intraperitoneal pentobarbital injection dose of 1.25 mg/kg to anesthetize mice for irradiation.
2. Skin area preparation
- Plan to shave the mice 2-3 days before irradiating. Trim the hair using a clipper (Figure 2A).
- Apply depilatory cream and wait for 1-2 min (Figure 2B). Wipe the cream with dry gauze and rinse the skin with a phosphate buffer saline (PBS)-soaked gauze (Figure 2C).
3. Irradiation procedure
- Anesthetize each mouse with an intraperitoneal injection administered 5 min before irradiation. Position the limb in the radiation field (25 cm x 25 cm) and secure it with surgical tape (Figure 3A-C).
- Restrict the body using surgical tape (Figure 3D). Place a 1 cm thick bolus to prevent/minimize deep penetration of radiations (Figure 3E).
- Calculate the applicator and cutout factors to deliver 40 Gy to the mouse skin. For the experiment here, use a linear accelerator to generate a 6 MeV electron beam for 3740 monitor units to induce irradiation burns. In this setting, a 25 cm x 25 cm radiation field at 1,000 MU/min dose with 100 cm source to surface distance (SSD), which is the distance from the irradiation source to the top of the bolus to deliver the 40 Gy. Irradiation time will vary according to linear accelerator used, beam energy and field size. A radiation or medical physicist should be consulted to calculate the monitor units necessary to deliver 40Gy on the linear accelerator.
4. Visual monitoring of fibrosis development
- Use a handheld digital camera to document the progression of fibrosis. Use the macro setting to capture detailed photos.
- Anesethetize mice by inhalation of isoflurane.
- Take pictures by positioning the lens closer to the skin and press capture. Try to keep the photos as consistent as possible. Keep the subject under direct supervision until sufficient consciousness to maintain sternal recumbency is regained.
5. Measurement of leg excursion as a functional outcome of fibrosis
- Starting from third week after irradiation, plan to have measurements each week for up to 6 weeks.
- Initiate and maintain the anesthesia as described above. Prepare a field in front of the nose cone and fix a protractor with tape in the center (Figure 4A).
- Transfer the mouse to the field and gently place its nose on the cone. Position the right knee to the protractor's center (Figure 4B).
- Keep the knee fixed using the left hand and use the right hand to dorsiflex the foot with the index and pollex fingers (Figure 4C,D).
- Note the degree of extension by reading the value indicated by the toes. Perform the same procedure on the contralateral non-irradiated leg.
6. Measurement of gait functional index
- 3D-print the rodent walking track to create a 40 cm walking path suspended at a height of 15 cm with a transparent floor (Figure 5). Place a video recorder underneath the track and start filming. The camera is adjusted to record a video at the highest possible resolution and at a minimum frame rate of 60 frames per second.
- Open one end of the track and transfer the mouse inside. Let the animal walk freely on the track. Capture the animal walking as smoothly as possible at least three times.
- Check the quality of the video before recording the next mouse.
- Transfer the videos to a computer with a video player, screenshot application, an image processing program, and spreadsheet software. Watch the recordings to capture three different, clear footprints using the screenshot function (Figure 6).
- Open the image processing program for toe spread measurements. Select File from the upper panel and click Open to find and display the image to analyze.
- Select Straight Line Tool from the second row of the upper panel (Figure 7_1). Using this tool, mark the wall width and click Analyze > Set Scale, then enter the exact value for a known distance to calibrate the scale (Figure 7_2-5).
- Use the Straight Line Tool to mark the footprint for different measurements (Figure 6C, D; 1: foot length, 2: outer toe spread, 3: inner toe spread), select Analyze > Measure, and record the length value (Figure 8).
- Perform the analyses for both the irradiated and non-irradiated limbs. Use the following equation published previously to assess the functionality24
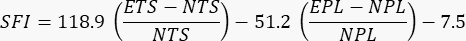
where SFI = sciatic functionality index, E = experimental or injured paw, N = normal or uninjured paw, TS = toe spread, and PL = print length.
7. Euthanasia
- Transfer the subject to a CO2 line-connected box. Start the CO2 infusion to reach 30%-70% concentration within the chamber. For a 5 L chamber, the gas infusion must be between 1.5-3.5 L/min.
- Wait for the animal to cease respiration for 5-10 min. Euthanize by cervical dislocation via holding the head in place from the skull base and firmly pulling the tail25.
8. Histology and downstream analyses 17
- Pull the irradiated hind limb to prepare it for excision. Select a 2 cm x 1 cm area to the posterior plane of the extremital skin.
- Use pair of sharp scissors to carefully collect the skin sample. Cut the tissue in half from the long axis to obtain two separate pieces.
- Fix one tissue piece in 10% formalin buffered saline. Process the fixed tissue for preparing histological sections on slides.
- Transfer the second piece to a box filled with dry ice to preserve the proteins and RNA. Then, quickly transfer the tissue samples to a -80 °C freezer and keep them frozen until further processing.
- Stain the slides for hematoxylin and eosin (H&E) stain and Masson's Trichrome stain. Visualize the stained slides under a microscope and take images using 10x magnification. Measure the epithelium thickness using image processing software as explained above for the gait functional assay.
9. Statistics
- Present the data as mean ± standard deviation. Evaluate the results using analysis of variance (ANOVA) followed by Bonferroni's multi-comparison test or Student t-test.
Results
Establishment and optimization of the current irradiation protocol resulted in a consistent and reproducible induction of fibrosis in mice. Right limbs of the mice were positioned and secured within the radiation field on the day of irradiation, and 40 Gy of radiation was administered.
The development of functional impairment in skin was monitored by capturing images every week, post-irradiation. Photos showed that the optimized protocol created fibrosis by day 40 with 95% confidence. An examp...
Discussion
Skin injury is a likely outcome of accidental or medical treatment-related exposure to radiation. Nuclear reactors possess an accidental breach risk due to human error or natural disasters like Chernobyl and Fukushima26,27. Therapeutical dosing for cancer treatment is the most common exposure, which uses fractionated repeated dose regimens that risk causing radiation-related fibrosis in the treated areas. This common chronic adverse reaction can be prevalent in u...
Disclosures
The authors have no competing financial interests or other conflicts of interest related to this work.
Acknowledgements
This work is funded by research grants from the Department of Defense W81XWH-19-PRMRP-DA, NIAID/NIH Grant 5R21AI153971-02, and PSF/MTF Grant 603902.
Materials
| Name | Company | Catalog Number | Comments |
| 10% Formalin | Fischer Scientific | 23-427098 | |
| Bolus | Orfit | 8333.SO1/R | |
| Clipper | Kent Scientific Corp. | CL8787-KIT | |
| CO2 | Various | ||
| CO2 Chamber | E-Z Systems Inc. | E-22000 | |
| Depilatory Cream | Church & Dwight Co., Inc. | Nair | |
| Digital Camera | Wolfang | GA100 | |
| Eppendrof Tubes | Eppendorf | 22364111 | |
| Eye Lubricant | Dechra | Puralube Ophthalmic Ointment | |
| Gauze | Covidien | 682252 | |
| Image Processing Program | NIH | Image J | |
| Isoflurane | Dechra | USP Inhalation Anesthetic | |
| Linear Accelaerator | Varian Medical Systems, Inc. | 23EX | |
| PBS | Cytiva | SH30256.LS | |
| Pentobarbital | Akorn Pharmaceuticals | Nembutal | |
| Protractor | Westcott | 550-1120 | |
| Small Animal Anesthesia System | E-Z Systems Inc. | EZ-SA800 | Single animal system |
| Spreadsheet Software | Microsoft | Excel | |
| Surgical Scissors | Medline | MDS0834111 | |
| Surgical Tape | 3M | 1538-1 | |
| Tape | 3M | H-1113 |
References
- Breitkreutz, D., Mirancea, N., Nischt, R. Basement membranes in skin: unique matrix structures with diverse functions. Histochemistry and Cell Biology. 132 (1), 1-10 (2009).
- Kim, J. -. S., et al. Comparison of skin injury induced by β-and γ-irradiation in the minipig model. Journal of Radiation Protection and Research. 42 (4), 189-196 (2017).
- Kolarsick, P. A., Kolarsick, M. A., Goodwin, C. Anatomy and physiology of the skin. Journal of the Dermatology Nurses' Association. 3 (4), 203-213 (2011).
- von Essen, C. F. Radiation tolerance of the skin. Acta Radiologica: Therapy, Physics, Biology. 8 (4), 311-330 (1969).
- Dainiak, N., et al. Literature review and global consensus on management of acute radiation syndrome affecting nonhematopoietic organ systems. Disaster Medicine and Public Health Preparedness. 5 (3), 183-201 (2011).
- Hopewell, J. The skin: its structure and response to ionizing radiation. International Journal of Radiation Biology. 57 (4), 751-773 (1990).
- Flynn, D. F., Goans, R. E. Nuclear terrorism: triage and medical management of radiation and combined-injury casualties. Surgical Clinics. 86 (3), 601-636 (2006).
- Peter, R. Cutaneous radiation syndrome in multi-organ failure. The British Journal of Radiology. 78 (1), 180-184 (2005).
- Meineke, V. The role of damage to the cutaneous system in radiation-induced multi-organ failure. The British Journal of Radiology. 78 (1), 95-99 (2005).
- Berger, M., Christensen, D., Lowry, P., Jones, O., Wiley, A. Medical management of radiation injuries: current approaches. Occupational Medicine. 56 (3), 162-172 (2006).
- Ralf, U. P., Petra, G. Management of cutaneous radiation injuries: diagnostic and therapeutic principles of the cutaneous radiation syndrome. Military Medicine. 167, 110-112 (2002).
- Bray, F. N., Simmons, B. J., Wolfson, A. H., Nouri, K. Acute and chronic cutaneous reactions to ionizing radiation therapy. Dermatology and Therapy. 6 (2), 185-206 (2016).
- Mendelsohn, F. A., Divino, C. M., Reis, E. D., Kerstein, M. D. Wound care after radiation therapy. Advances in Skin & Wound. 15 (5), 216-224 (2002).
- Peter, R. U. . Radiation Treatment and Radiation Reactions in Dermatology. , 185-188 (2015).
- Ejaz, A., Greenberger, J. S., Rubin, P. J. Understanding the mechanism of radiation induced fibrosis and therapy options. Pharmacology & Therapeutics. 204, 107399 (2019).
- Williams, J. P., et al. Animal models for medical countermeasures to radiation exposure. Radiation research. 173 (4), 557-578 (2010).
- Ejaz, A., Epperly, M. W., Hou, W., Greenberger, J. S., Rubin, J. P. Adipose-derived stem cell therapy ameliorates ionizing irradiation fibrosis via hepatocyte growth factor-mediated transforming growth factor-beta downregulation and recruitment of bone marrow cells. Stem Cells. 37 (6), 791-802 (2019).
- Haubner, F., et al. Effects of external radiation in a co-culture model of endothelial cells and adipose-derived stem cells. Radiation Oncology. 8 (1), 66 (2013).
- Ebrahimian, T. G., et al. Cell therapy based on adipose tissue-derived stromal cells promotes physiological and pathological wound healing. Arteriosclerosis, Thrombosis, and Vascular Biology. 29 (4), 503-510 (2009).
- Haubner, F., et al. A co-culture model of fibroblasts and adipose tissue-derived stem cells reveals new insights into impaired wound healing after radiotherapy. International Journal of Molecular Sciences. 16 (11), 25947-25958 (2015).
- Urano, M., Kenton, L. A., Kahn, J. The effect of hyperthermia on the early and late appearing mouse foot reactions and on the radiation carcinogenesis: effect on the early and late appearing reactions. International Journal of Radiation Oncology Biology Physics. 15 (1), 159-166 (1988).
- Law, M., Thomlinson, R. The pathogenesis of necrosis after radiotherapy. The British Journal of Radiology. 47 (562), 740 (1974).
- Abe, Y., Urano, M. Fraction size-dependent acute skin reaction of mice after multiple twice-a-day doses. International Journal of Radiation Oncology Biology Physics. 18 (2), 359-364 (1990).
- Inserra, M. M., Bloch, D. A., Terris, D. J. Functional indices for sciatic, peroneal, and posterior tibial nerve lesions in the mouse. Microsurgery. 18 (2), 119-124 (1998).
- Suckow, C. P. S. a. M. A., Weichbrod, R. H., Thompson, G. A., Norton, J. N. . Management of Animal Care and Use Programs in Research, Education, and Testing. , (2018).
- Yamashita, S., Suzuki, S., Suzuki, S., Shimura, H., Saenko, V. Lessons from Fukushima: latest findings of thyroid cancer after the Fukushima nuclear power plant accident. Thyroid. 28 (1), 11-22 (2018).
- Cardis, E., et al. Cancer consequences of the Chernobyl accident: 20 years on. Journal of Radiological Protection. 26 (2), 127-140 (2006).
- Williams, N. R., et al. Radiation-induced fibrosis in breast cancer: A protocol for an observational cross-sectional pilot study for personalised risk estimation and objective assessment. International Journal of Surgery Protocols. 14, 9-13 (2019).
- Meineke, V., Fliedner, T. Radiation-induced multi-organ involvement and failure: challenges for radiation accident medical management and future research. The British Journal of Radiology. 78 (1), 196-200 (2005).
- Stone, H. B. Leg contracture in mice: an assay of normal tissue response. International Journal of Radiation Oncology Biology Physics. 10 (7), 1053-1061 (1984).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved