Method Article
Isolation of Precursor B-cell Subsets from Umbilical Cord Blood
In This Article
Summary
Here we describe a protocol for isolating subsets of precursor B-cells from umbilical cord blood. A sufficient quantity and quality of nucleic acids may be extracted from the cells and used in subsequent assays utilizing DNA or RNA.
Abstract
Umbilical cord blood is highly enriched for hematopoietic progenitor cells at different lineage commitment stages. We have developed a protocol for isolating precursor B-cells at four different stages of differentiation. Because genes are expressed and epigenetic modifications occur in a tissue specific manner, it is vital to discriminate between tissues and cell types in order to be able to identify alterations in the genome and the epigenome that may lead to the development of disease. This method can be adapted to any type of cell present in umbilical cord blood at any stage of differentiation.
This method comprises 4 main steps. First, mononuclear cells are separated by density centrifugation. Second, B-cells are enriched using biotin conjugated antibodies that recognize and remove non B-cells from the mononuclear cells. Third the B-cells are fluorescently labeled with cell surface protein antibodies specific to individual stages of B-cell development. Finally, the fluorescently labeled cells are sorted and individual populations are recovered. The recovered cells are of sufficient quantity and quality to be utilized in downstream nucleic acid assays.
Introduction
In order to identify aberrations that are present in disease, it is vitally important that we use healthy tissues or cells that correspond to the tissue or cell type affected by the disease. One reason for this is that epigenetic variation among tissue types is responsible for regulating gene expression and is critical for cellular differentiation during normal human development1,2. A second reason is that aberrant tissue specific gene regulation may have dire consequences on normal development and is known to contribute to a multitude of disease states including cancer. Therefore, a better understanding of a disease that involves hematopoietic cells requires knowledge of healthy hematopoietic cells.
The development of hematopoietic cells in the bone marrow proceeds through a systematic order of events characterized by changes in the expression of cell surface markers3. Studies involving adult participants have shown that bone marrow usually contains a low number of precursor B-cells4,5; whereas studies involving pediatric participants have shown that the percentage of precursor B-cells is relatively high in individuals less than 5 years of age6. Umbilical cord blood is used as a source of hematopoietic stem cells in the treatment of blood related disorders and malignancies, is readily available via cord blood banks and is enriched for immature B and T cells7 which are the target cells of multiple disorders including leukemia and lymphomas.
Precursor B-cells in the bone marrow have been extensively phenotyped8,9 and can be defined by the presence of specific cell surface markers that can be used to sort these cells into distinct subsets. Normal B-cell differentiation proceeds through a series of stages in the bone marrow beginning with the earliest pro-B cells and culminating in immature or naïve B-cells. According to van Zelm and colleagues10, pro-B cells are characterized by the presence of CD34 and in the transition to stage 2 (Pre-BI) CD19 is acquired. Stage 3 (Pre-BII) cells no longer express CD34 and begin to express cytoplasmic IgM. Finally, a defining characteristic of stage 4 (immature B-cells) is the expression of surface IgM. The sorting strategy described in this protocol was first described by Caldwell and colleagues6 and includes the use of only 3 cell surface markers which greatly reduces the complexity and the cost of performing cell sorting experiments. In their work, a relationship between CD45 and the stages of B-cell differentiation was established. They observed that B-cells in the bone marrow display variable levels of expression of CD45. Specifically, cells that expressed high levels of CD45 corresponded to cells that expressed surface IgM (immature B-cells), those that expressed an intermediate level of CD45 corresponded to cells that expressed cytoplasmic IgM (pre-BII cells), and those that expressed low levels of CD45 corresponded to cells that did not express cytoplasmic IgM (pre-BI cells). This protocol uses the strategy developed by Caldwell and colleagues6 to isolate subsets of precursor B-cells from umbilical cord blood (Figure 1) which can be used in downstream assays requiring high quality nucleic acids such as the methylated-CpG island recovery assay (MIRA)11 and quantitative real time PCR assays. The method employs an initial separation using magnetic beads to deplete all non-B cells from umbilical cord blood and requires staining with only 3 antibodies (CD34, CD19 and CD45). The cells that are recovered represent 4 stages of B-cell differentiation: 1) CD34+; CD19+ (late pro-B and early pre-BI); 2) CD34-; CD19+; CD45low (late pre-BI); 3) CD34-; CD19+; CD45med (pre-BII); and 4) CD34-; CD19+; CD45high (immature B-cells).
Protocol
1. Isolation of Mononuclear Cells from Umbilical Cord Blood
- Prepare EDTA-PBS buffer- add 5 ml bovine serum albumin (BSA) stock solution to 95 ml rinsing buffer (1:20 dilution). Degas the buffer and keep the buffer on ice. IMPORTANT: Failure to degas the buffer may result in less than optimal results when isolating CD19+ B-cells because bubbles may block the isolation column.
- Prepare 50 ml conical tubes for density gradient centrifugation. Determine the number of tubes required for processing the cord blood (1 tube can process 8 ml blood) and add 15 ml of Ficoll-Paque PLUS to each tube.
- Dilute 8 ml of cord blood with 24 ml DPBS (1X) and carefully layer the diluted cord blood mixture on top of the Ficoll-Paque PLUS in each of the 50 ml conical tubes. Do not mix the blood and Ficoll-Paque PLUS. IMPORTANT: To avoid mixing of the cord blood and Ficoll-Paque PLUS, hold the tube at a 45 degree angle and layer the blood mixture slowly.
- Centrifuge at 400 x g for 40 min at 20 °C. Mononuclear cells (MNC) will remain at the plasma-Ficoll-Paque PLUS interface whereas granulocytes and erythrocytes sediment due to higher density at the osmotic pressure of Ficoll-Paque PLUS. Label seven 5 ml round-bottom tubes to be used in Part 3 with the sample ID, date and the following:
| Tube | Label | Purpose |
| 1 | Unstained | Unstained cells for normalization during flow cytometry |
| 2 | 7AAD | To determine viability during flow cytometry |
| 3 | +++ | Contains the cells that will be sorted |
| 4 | 34+ | To collect CD34+/CD19+ (late pro-B - early pre-BI) cells |
| 5 | 45low | To collect CD34-/CD19+/CD45low (pre-BI) cells |
| 6 | 45med | To collect CD34-/CD19+/CD45med (pre-BII) cells |
| 7 | 45high | To collect CD34-/CD19+/ CD45high (immature B) cells |
Coat tubes 4, 5, 6, and 7 with 2% FBS and place all tubes on ice.
- Aspirate the upper plasma layer carefully and avoid contact with the mononuclear cell layer. Using a 10 ml glass pipette, carefully transfer the mononuclear cell layer to a new 50 ml conical tube. Combine the mononuclear cells from three tubes together into a single 50 ml tube.
- Fill the tube with PBS, mix gently and centrifuge at 300 x g for 10 min at 20 °C. Carefully aspirate the supernatant without disturbing the cell pellet. Repeat 1x. After the first wash, resuspend the pellets and transfer to a single 50 ml conical tube.
- Gently resuspend the cell pellet in 200 μl of PBS. Remove 1 μl of the cell suspension, add it to 1 ml of PBS in a 1.5 ml microcentrifuge tube and set aside for counting (count cells after placing the 50 ml cell suspension in the centrifuge). Fill the tube with PBS and centrifuge at 200 x g for 15 min to remove platelets. Remove the supernatant completely without disturbing the cell pellet.
2. Modified B-cell Isolation from Mononuclear Cells Using MACS Separation
- Resuspend the pellet from step 1.7 with 160 μl cold (4 °C) EDTA-PBS buffer.
- Add 40 μl B-CLL biotin antibody cocktail. Pipette to mix well and incubate for 10 min at 4 °C.
- Wash cells - add 1 ml cold (4 °C) EDTA-PBS buffer per 10 million cells (cell number determined in step 1.7) and centrifuge at 300 x g for 10 min. Aspirate the supernatant completely and then resuspend the cell pellet in 320 μl cold (4 °C) EDTA-PBS buffer.
- Add 80 μl anti-biotin MicroBeads, mix well and incubate for 15 min at 4 °C.
- Wash cells - add 1 ml cold (4 °C) EDTA-PBS buffer per 10 million cells and centrifuge at 300 x g for 10 min. Resuspend up to 100 million cells in 500 μl of cold (4 °C) EDTA-PBS buffer. Scale buffer volume according to cell number.
- Prepare MACS Columns and MACS Separators during centrifugation (Step 2.5). Place an LS column in the magnetic field of the MACS separator, add 3 ml of cold (4 °C) EDTA-PBS buffer onto the column and allow the buffer to drip through the column. Use a receptacle to catch the flow through. Discard the flow through.
- Place a 50 ml conical tube below the column and pipette the cell suspension onto the column. Allow the unlabeled cells to drip through the column and into the 50 ml conical tube. IMPORTANT: These are the cells that will be used for antibody labeling and cell sorting. Do not discard the flow through. To recover all of the cells from step 2.5, add an additional 1 ml of cold (4 °C) EDTA-PBS buffer to the walls of the conical tube. Mix gently and pipette the remaining cell suspension onto the column. Repeat 1x. Proceed to step 2.8 using the unlabeled cells collected in the conical tube after passing through the column.
- Fill the conical tube containing unlabeled cells with PBS and centrifuge at 300 x g for 10 min. Carefully remove the supernatant without disturbing the cell pellet. Add 200 μl of EDTA-PBS buffer and gently resuspend the cells.
3. Antibody Labeling and Preparation for Cell Sorting
- Aspirate 1 μl of the cell suspension and place into tube labeled "unstained" (Step 1.4). Add 500 μl EDTA-PBS buffer and store the tube on ice.
- Add 20 μl of each antibody (CD19, CD34 and CD45) per million cells into the remaining cell suspension. Mix well and place the tube in the dark for 30 min at RT. CD antibodies are light sensitive, complete steps 2-4 with the lights turned off.
- After 30 min of incubation with antibodies, aspirate 40 μl of the cell suspension and place it into the tube labeled "7AAD". Add 1 ml PBS into the tube and centrifuge at 500 x g for 2 min. Remove the supernatant and resuspend the cells in 100 μl of binding buffer. Add 7 μl of 7AAD (7-Aminoactinomycin D) and incubate in the dark for 10 min at RT. During the 10 min incubation complete step 3.4.
- Add PBS to the top of the tube containing the remaining cells labeled with CD antibodies. Centrifuge the tube at 500 x g for 3 min. Remove the supernatant, add 500 μl EDTA-PBS buffer and resuspend the cell pellet. Transfer the entire volume of the cell suspension into the tube labeled "+++". To recover all of the cell suspension add an additional 1 ml EDTA-PBS buffer to the 50 ml conical tube and transfer into the tube labeled "+++".
- After incubation (Step 3.3), add 300 μl binding buffer into 7AAD tube. The "unstained", "7AAD" and "+++" samples and the "34 +", "45low", "45med" and "45high" tubes are ready for flow cytometry.
4. Cell Sorting Using the MoFlo XDP Flow Cytometer
- Set-up MoFlo for sorting: Align lasers; stabilize droplet stream; determine drop delay.
- Prepare single color compensation controls using Invitrogen AbC Mouse bead kit (or similar) according to manufacturer's instructions. Run single color controls, adjusting the voltages of fluorescence channels for optimum separation of positive and negative populations. Set compensation coefficients and apply compensated parameter to collection protocol. Re-establish compensation coefficients each time a new lot of antibody is obtained.
- Create protocol including plots as shown in Figure 2.
- Run unstained sample, set voltage and gain for forward and side scatter, and identify negative fluorescence populations. Because DNA recovery is the goal of the sort, the threshold should be set low (≤1%) so that DNA-containing debris does not contaminate the recovered samples. *Ensure that the Aerosol Evacuation System is running at all times that live human cells are being run on the MoFlo.
- Run a small aliquot of the +++ sample (~ 50,000 events) to set the gating strategy (Figure 2). Because B-cell progenitors do not scatter identically to mature B-cells, backgate the ungated CD19+/CD34+ population onto the SSC vs FSC plot to ensure the lymphocyte gate includes potential Pro-B cells. From this sample also set the negative fluorescence population for 7AAD.
- Run the 7AAD sample to determine sample viability. Only sort cells from a sample with high viability at the outset (≥95%) as dead cells will stain indiscriminately and may contaminate sort populations.
- Set up sort decisions to collect four populations: CD19+/CD34+; CD19+/CD34-/CD45low; CD19+/CD34-/CD45med; and CD19+/CD34-/CD45high. Include the lymphocyte and doublet discrimination gates in the sort decisions of all populations.
- To prevent clogs in the sorting tip, filter +++ sample through a 40 μm cell strainer immediately before sorting and re-filter if any aggregation occurs in sample during sort.
- Sort the cells into collection tubes (Step 1.4) coated with 2% FBS in PBS in a cooled or ice-packed tube holder.
- Immediately after sort is completed, extract DNA.
Results
Between sample variation plays a role in the success of the cell sort (Table 1). The samples with good success rates have low levels of contaminating debris (Figure 3A) and the samples with poor success rates have high levels of contaminating debris (Figure 3B). Between sample variation can be somewhat controlled if the cord blood sample was collected within 24 hr of shipment (O/N first priority).
The flow sorted cells from each precursor B-cell subset are of sufficient quantity and quality to perform nucleic acid isolations. The DNA isolated is of high quality and can be used in downstream analyses. We routinely utilize this DNA in MIRA11 to enrich for methylated DNA (Figure 4).
| Cord Blood Facility Data | Poor Sort | Good Sort |
| Working Blood Volume with Anticoagulant (ml) | 110 | 104 |
| White Blood Cells [103/ μl] | 8.68 | 11.19 |
| Lymphocytes [103/ μl] | 3.88 | 3.76 |
| Lymphocyte (%) | 44.70 | 33.6 |
| Red Blood Cells [106/ μl] | 3.65 | 3.05 |
| In-house Data | ||
| Cell Number after Ficoll-Paque | 206 M | 309 M |
| Cell Number after MACS Separation | 22 M | 16.5 M |
| Total Events Count (MoFlo XDP) | 30.57 M | 19.97 M |
| Viability (%) | 98.00 | 98.00 |
| Cells in Lymphocyte Gate (%) | 17.00 | 50.00 |
| CD19+/CD34+ | ||
| Total Cell Number | 10959 | 48316 |
| % of Total Events | 0.04 | 0.24 |
| Efficiency | 88% | 88% |
| CD19+/CD34-/CD45low | ||
| Total Cell Number | 16619 | 26941 |
| % of Total Events | 0.05 | 0.13 |
| Efficiency | 89% | 87% |
| CD19+/CD34-/CD45med | ||
| Total Cell Number | 469745 | 507540 |
| % of Total Events | 1.54 | 2.54 |
| Efficiency | 89% | 89% |
| CD19+/CD34-/CD45high | ||
| Total Cell Number | 1896062 | 2047142 |
| % of Total Events | 6.2 | 10.25 |
| Efficiency | 91% | 90% |
Table 1. Representative cell sort statistics. Rows 1-5 are counts provided by the cord blood facility. The remaining rows are data collected in our laboratory. The poor sort contained high levels of debris and a low percentage of cells in the lymphocyte gate.
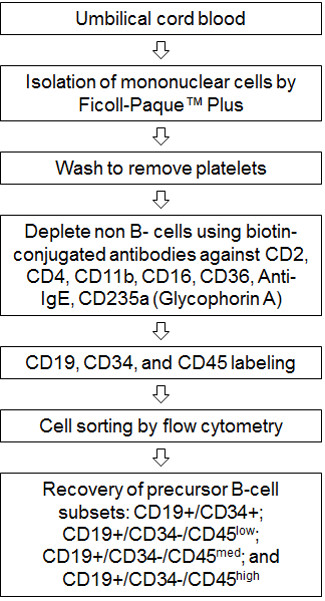
Figure 1. Flow chart of procedure. Umbilical cord blood is processed and mononuclear cells are isolated using Ficoll-paque plus. An additional wash step is included to remove contaminating platelets. B-cells are isolated from mononuclear cells using the MACS human B-cell isolation kit (B-CLL). This step uses biotin-conjugated monoclonal antibodies (CD2, CD4, CD11b, CD36, Anti-IgE and CD235a) to remove all non-B cells. The recovered B-cells are labeled with CD19-APC, CD34-PE and CD45-FITC antibodies then sorted using the MoFlo XDP flow cytometer to recover precursor B-cell subsets: CD19+/CD34+; CD19+/CD34-/CD45low; CD19+/CD34-/CD45med; and CD19+/CD34-/CD45high.
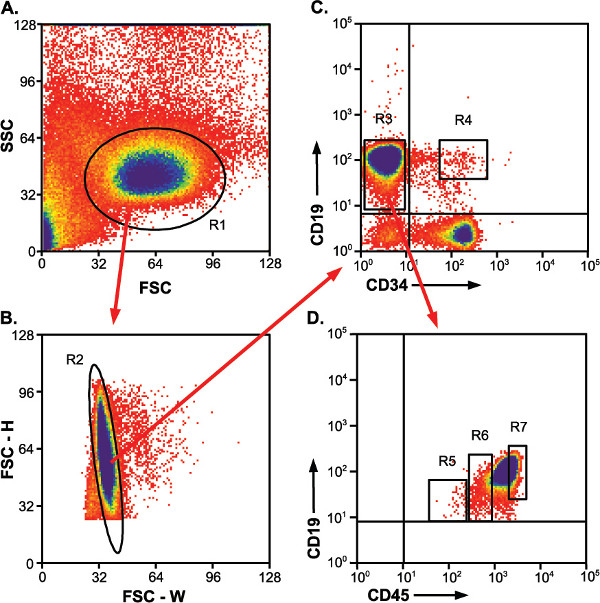
Figure 2. Gating strategy for identifying and sorting populations. Red arrows indicate the gate that is applied to subsequent plot. R4, R5, R6 and R7 gates indicate sorted populations. a) Forward vs. side scatter plot showing enriched lymphocyte population. R1, lymphocyte gate. b) Height vs. width of forward scatter plot for identifying single cells versus doublets. R2, single cell gate. c) CD19-APC positive cells fall into CD34-PE negative and positive populations. R3, CD19+/CD34- gate; R4, CD19+/CD34+ gate indicating desired sort population. d) CD19+/CD34- cells fall into three somewhat distinct CD45-FITC populations. R5 and R6, CD19+/CD34-/CD45low and CD19+/CD34-/CD45med populations, respectively. R7, because of high cell numbers in the CD19+/CD34-/CD45high population, this sort gate encompasses only a portion of the population. All cells of this population do not need to be collected for downstream analyses.
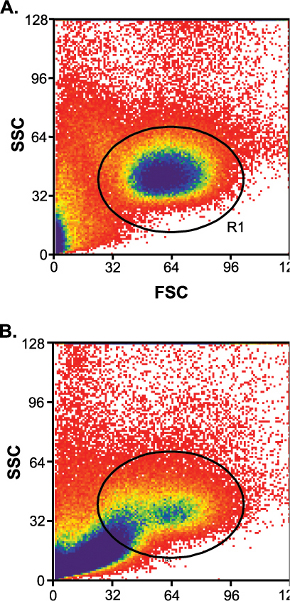
Figure 3. a) Low levels of contaminating debris after column enrichment. R1, lymphocyte gate. b) High levels of contaminating debris after column enrichment.
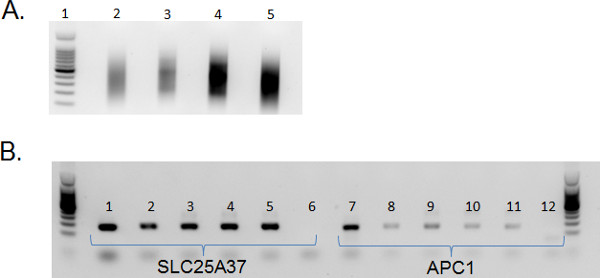
Figure 4. Enrichment of methylated DNA from subsets of precursor B-cells. a) Sonicated DNA isolated from precursor B-cell subsets is of high quality. For library construction DNA is sonicated to an average of 200-600 bp. Lane 1: Promega 100 bp ladder (catalog #G2101); Lane 2: Total DNA isolated from CD19+/CD34+ cells; Lane 3: Total DNA isolated from CD19+/CD34-/CD45low cells; Lane 4: 100 ng DNA isolated from CD19+/CD34-/CD45med cells; Lane 5: 100 ng DNA isolated from CD19+/CD34-/CD45high cells. DNA was sonicated using the Diagenode Bioruptor on High for a total of 9 min (30 sec ON; 30 sec OFF). DNA was column purified and visualized on a 1% agarose gel with SYBR green nucleic acid gel stain. b) After MIRA using the ActivMotif MethylCollector Ultra kit, PCR with SLC25A37 (1-6) and APC1 (7-12) is performed to confirm enrichment of methylated DNA. 1&7: Sonicated genomic DNA (no MIRA); 2&8: MIRA-CD19+/CD34+ DNA; 3&9: MIRA-CD19+/CD34-/CD45low DNA; 4&10: MIRA-CD19+/CD34-/CD45med DNA; 5&11: MIRA-CD19+/CD34-/CD45high DNA; 6&12: Water control. Higher amplification of SLC25A37 confirms the enrichment of methylated DNA in the subsets of precursor B-cells.
Discussion
The factor with the greatest impact on the success of the protocol is the presence of contaminating debris. If requesting blood from a cord blood bank it is important to have the blood shipped as soon after collection as possible. In addition, samples that are classified as lymphocytosis contain higher numbers of lymphocytes; however, these samples do not have adequate numbers of precursor B-cells and should not be used. To increase the probability of obtaining sufficient numbers of cells for each of the precursor subsets we recommend beginning with at least 85 ml of cord blood.
It is important to note that unlike adult peripheral blood separations, when fractionating umbilical cord blood the mononuclear layer is often contaminated with red blood cells12. This protocol includes an extra wash step to remove contaminating platelets. Protocols describing mononuclear separations from cord blood suggest including a lysis step to remove unwanted red cells. We do not recommend this step because it produces contaminating debris and has a negative impact on the success of the cell sort.
In order to reduce the number of cells that have to be distinguished by flow cytometry it is necessary to perform a B-cell enrichment prior to cell sorting. The protocol provided by Miltenyi Biotec, that accompanies the B-Cell Isolation Kit (B-CLL), has been optimized for peripheral blood and recommends using 10 μl B-CLL biotin antibody cocktail per 10 million cells. This step uses antibodies against CD2 (T cells; NK cells), CD4 (T cells), CD11b (granulocytes; monocytes; macrophages), CD16 (NK cells; macrophages; mast cells), CD36 (platelets), CD235a (erythroid cells) to deplete non-B cells from the umbilical cord blood. It is important to use the kit described and not the B-cell isolation kit II because the former kit contains an antibody against CD43 which is present on pro-B cells. During our optimization of this protocol, we found that a total of 40 μl of B-CLL biotin antibody cocktail is sufficient to produce a positive cell sort result. In addition we found that using as little as 5 μl more of the B-CLL biotin antibody cocktail had adverse effects on the cell sort. Therefore, we highly recommend using 40 μl of B-CLL biotin antibody cocktail for total mononuclear cell numbers between 175-250 million. If starting with lower or higher numbers of cells it may be necessary to scale the reagents accordingly.
There are numerous conflicting publications describing the markers that may be used to distinguish sub-populations of B-cells. Most of the discrepancy can be explained by the fact that differentiation is an on-going process and that the presence or absence of cell surface markers occurs in gradual manner instead of in an all or nothing manner. In this protocol CD34-/CD19+and the intensity level of CD45+ (low, medium, high) expression was used to distinguish pre-BI, pre-BII and immature B-cells with an increase in the level of CD45 expression corresponding to the progression of differentiation6. Pro-B cells have been described by some to be CD19+/CD34+ 13 while others have shown that pro-B cells are CD19-/CD34+ and pre-BI cells are CD19+/CD34+ 9,10. Based on these discrepancies we have designated the CD19+/CD34+cells as late pro-B cells/early pre-BI cells. It is important to note that this strategy was developed to isolate subsets of precursor B-cells that correspond to the cells affected by the disease acute lymphoblastic leukemia. However, there are multiple sorting strategies that may be employed depending on the cell subtype of interest.
Antibody-fluorochrome combinations should be selected based on the capabilities of the available cell sorter. As a general rule, the brightest fluorochrome in your panel should be used to label the least populous antigen and vice versa. In detecting double-stained populations, especially rare events such as these, doublet discrimination (Figure 2B) is a vitally important part of the gating strategy. This ensures that double-positive events are truly single cells stained with both antibodies and not merely two single-stained cells adhered to one another.
High speed, 4-way cell sorting necessarily creates a controlled aerosol environment. Therefore, live human cell sorting should be performed with great care. Published safety and decontamination guidelines are available and should be reviewed and implemented before the sort14. Approval from Institutional Biosafety Committees or their equivalents may be required prior to sorting. For proper decontamination after sorting, bleach should be added to the waste container to a final concentration of 10%. Similarly, all samples tubing as well as all surfaces in the immediate area should be thoroughly decontaminated with a freshly made 10% bleach solution.
In summary, this protocol provides a strategy for obtaining rare populations of precursor B-cells and may be modified to isolate any rare population present in cord blood including hematopoietic stem cells and immature T-cells. Recently, immature B cells have been identified in the peripheral blood of individuals with advanced HIV15. Therefore, the utility of this method extends beyond the study of blood cancers. Finally, we have not yet performed RNA isolations on the flow sorted cells; however, this method should be adaptable with the caveat that during cell sorting the cells should be sorted directly into trizol or an equivalent RNA compatible solution such as RLT from the RNeasy kit available through Qiagen.
Disclosures
Authors have nothing to disclose.
Acknowledgements
This work was supported by the National Institutes of Health (NCI R00 CA132784) to K.T.
Materials
| Name | Company | Catalog Number | Comments |
| DPBS (1X) | Gibco by Life Technologies | 14190-144 | |
| Ficoll-Paque PLUS | GE Healthcare Bio-Sciences AB | 17-1440-03 | |
| LS Column | MACS Miltenyi Biotec | 130-042-401 | |
| MACS Multi Stand | MACS Miltenyi Biotec | 130-042-303 | |
| MidiMACS Separator | MACS Miltenyi Biotec | 130-042-302 | |
| B-Cell Isolation Kit (B CLL) | MACS Miltenyi Biotec | 130-093-660 | |
| Fetal Bovine Serum | ATLANTA Biologicals | S11195 | |
| Microcentrifuge tube (1.5 ml) | MIDSCI | SS1500 | |
| BD Pharmingen 7-AAD (7-Aminoactinomycin D) | BD Biosciences | 559763 | |
| DB Pharmingen APC Mouse Anti-Human CD19 | BD Biosciences | 555415 | |
| DB Pharmingen PE Mouse Anti-Human CD34 | BD Biosciences | 560941 | |
| CD45 FITC | BD Biosciences | 347463 | |
| autoMACS Rinsing Solution | MACS Miltenyi Biotec | 130-091-222 | |
| MACS BSA Stock Solution | MACS Miltenyi Biotec | 130-091-376 | |
| BD Falcon 5 ml Polystyrene Round-Bottom Tube | BD Biosciences | 352058 | |
| BD Falcon 50 ml Tube | BD Biosciences | 352098 | |
| PuraFlow Sheath Fluid, 8X | Beckman Coulter | CY30230 | |
| FlowCheck Alignment beads | Beckman Coulter | 6605359 | |
| Ultra Rainbow Alignment beads | Spherotech | URFP-30-2 | |
| ViroSafe Aerosol Evacuation Filter | Beckman Coulter | ML01330 | |
| ABC Mouse bead kit | Invitrogen | A-10344 | |
| 40 μm cell strainer | Fisher Scientific | 22363547 | |
| Fisher Scientific Hemocytometer | Fisher Scientific | 267110 | |
| Microscope | |||
| accuSpin Model 3R Benchtop Centrifuge | Fisher Scientific | 13-100-516 | |
| MoFlo XDP flow Cytometer | Beckman Coulter | ML99030 | |
| Aerosol Evacuation Unit | Beckman Coulter |
References
- Song, F., et al. Association of tissue-specific differentially methylated regions (TDMs) with differential gene expression. PNAS. 102 (9), 3336-3341 (2005).
- Ohgane, J., Yagi, S., Shiota, K. Epigenetics: The DNA methylation profile of tissue-dependent and differentially methylated regions in cells. Placenta. 29 (S), 29-35 (2008).
- Brown, G., et al. The sequential determination model of hematopoiesis. Trends Immunol. 28 (10), 442-448 (2007).
- Clark, P., et al. Lymphocyte subsets in normal bone marrow. Blood. 67 (6), 1600-1606 (1986).
- Loken, M. R., et al. Flow cytometric analysis of human bone marrow. II. Normal B lymphocyte development. Blood. 70 (5), 1316-1324 (1987).
- Caldwell, C. W., Poje, E., Helikson, M. A. B-cell precursors in normal pediatric bone marrow. American Journal of Clinical Pathology. 95 (6), 816-823 (1991).
- Tucci, A., et al. Are cord blood B cells functionally mature? Clin. Exp. Immunol. 84 (3), 389-394 (1991).
- Ghia, P., et al. Ordering of human bone marrow B lymphocyte precursors by single-cell polymerase chain reaction analyses of the rearrangement status of the immunoglobulin H and L chain gene loci. J. Exp. Med. 184, 2217-2219 (1996).
- Noordzij, J. G., et al. Composition of precursor B-cell compartment in bone marrow from patients with X-linked agammaglobulinemia compared with healthy children. Pediatric Research. 51 (2), 159-168 (2002).
- van Zelm, M. C., et al. Ig gene rearrangement steps are initiated in early human precursor B cell subsets and correlate with specific transcription factor expression. J. Immunol. 175 (9), 5912-5922 (2005).
- Rauch, T. A., Pfeifer, G. P. The MIRA method for DNA methylation analysis. Methods Mol. Biol. 507, 65-75 (2009).
- Kanof, E. M., et al. Isolation of whole mononuclear cells from peripheral blood and cord blood. Current Protocols in Immunology. , Suppl 19. 7.1.1-7.1.7 (1996).
- LeBien, T. W. Fates of human B-cell precursors. Blood. 96 (1), 9-23 (2000).
- Schmid, I., et al. International society for analytical cytology biosafety standard for sorting of unfixed cells. Cytometry A. 71 (6), 414-437 (2007).
- Malaspina, A., et al. Appearance of immature/transitional B cells in HIV-infected individuals with advanced disease: correlation with increased IL-7. PNAS. 103 (7), 2262-2267 (2006).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved