Method Article
Minimally Invasive Endoscopic Intracerebral Hemorrhage Evacuation
In This Article
Summary
This paper details the surgical protocol for minimally invasive endoscopic intracerebral hemorrhage evacuation using the SCUBA technique.
Abstract
Intracerebral hemorrhage (ICH) is a subtype of stroke with high mortality and poor functional outcomes, largely because there are no evidence-based treatment options for this devastating disease process. In the past decade, a number of minimally invasive surgeries have emerged to address this issue, one of which is endoscopic evacuation. Stereotactic ICH Underwater Blood Aspiration (SCUBA) is a novel endoscopic evacuation technique performed in a fluid-filled cavity using an aspiration system to provide an additional degree of freedom during the procedure. The SCUBA procedure utilizes a suction device, endoscope, and sheath and is divided into two phases. The first phase involves maximal aspiration and minimal irrigation to decrease clot burden. The second phase involves increasing irrigation for visibility, decreasing aspiration strength for targeted aspiration without disturbing the cavity wall, and cauterizing any bleeding vessels. Using the endoscope and aspiration wand, this technique aims to maximize hematoma evacuation while minimizing collateral damage to the surrounding brain. Advantages of the SCUBA technique include the use of a low-profile endoscopic sheath minimizing brain disruption and improved visualization with a fluid-filled cavity rather than an air-filled one.
Introduction
Intracerebral hemorrhage is a bleed that occurs in the brain parenchyma and is the most devastating subtype of stroke in terms of mortality and disability. The worldwide incidence of ICH is approximately 24.6 per 100,000 individuals, with 40,000 to 67,000 cases occurring every year in the US1,2. Intracerebral hemorrhage is a medical emergency requiring fast diagnosis and management. Historically, outcomes have been bleak, with mortality rates of 40% at 1 month, 51-54% at 1 year, and 71% at 5 years3,4,5,6. A key reason for such a poor prognosis is that there are no evidence-based treatments for this disease process. Previous clinical trials (STICH I and II) did not demonstrate improved outcomes for surgery compared to conservative medical management7,8. A proposed hypothesis for the failure of craniotomies is that any benefit gained from evacuating the clot is outweighed by the extensive brain trauma inflicted by the invasive nature of the procedure. As a result, in the past decade, a number of minimally invasive techniques have been developed to attempt to solve this problem, each with advantages and disadvantages. The techniques can be grouped into two categories: stereotactic aspiration with thrombolysis and active evacuation. The former involves aspirating the clot through a burr hole, administering a thrombolytic agent, and draining the residual clot through a catheter over the period of several days. This technique is currently being tested in the MISTIE clinical trial and is used by clinicians in China with the YL-1 craniopuncture needle9,10. Active evacuation, on the other hand, involves aspirating the entire clot in a single procedure without the need for a draining catheter.
A number of clinical trials are also underway for this technique, including ENRICH, which utilizes the NICO BrainPath system for endoport-assisted trans-sulcal evacuation; the INVEST trial11, which is a single arm feasibility study using the Penumbra Apollo or Artemis Systems for endoscopic evacuation; and the MIND trial, which is a multicenter randomized clinical trial evaluating endoscopic evacuation using the Artemis device. Endoscopic evacuation is a promising technique because it has the lowest profile working channel to minimize brain trauma12. This paper outlines a specific endoscopic technique described as Stereotactic ICH Underwater Blood Aspiration (SCUBA)13. The first phase focuses on debulking the hematoma using maximal aspiration while working within the end of the sheath. The second phase utilizes a high irrigation rate to aspirate residual clots and cauterize any bleeding vessels in a highly targeted manner.
There are three devices used in the SCUBA procedure: a sheath (6.33 mm), endoscope, and aspiration system. The aspiration system consists of a surgical wand (2.6 mm) designed to fit inside the working channel of an endoscope, which is inserted into the sheath. The wand is capable of aspiration and, with the press of a button on the handle, morcellation. The morcellation component of the device is a spinning bident at the tip of the suction tube that spins upon activation. Suction is activated by covering the hole at the thumb on the handle, and the bident is activated by pressing firmly on the button. Suction activation in this regard is similar to common neurosurgical suction instruments.
Protocol
Prior to performing this protocol, the required institutional approval and patient consent were obtained. All procedures were approved by Mount Sinai Hospital.
1. Inclusion Criteria
- Include patients who meet all of the following criteria: age > 18 years old, baseline modified Rankin Scale (mRS) < 4, presenting Glasgow Coma Scale (GCS) > 4, presenting NIH Stroke Scale (NIHSS) ≥ 6, symptom onset < 24 hours prior to initial CT scan (minimally invasive ICH evacuation can be performed within 72 hours of ictus), supratentorial location of ICH, ICH volume > 20 cm3, stability in ICH volume measured on two CT scans 6 hours apart, systolic blood pressure controlled to < 160 mmHg for at least 6 hours prior to surgery.
2. Exclusion Criteria
- Exclude patients who meet one or more of the following criteria: CT scan demonstrates expanding hemorrhage; spot sign on CTA imaging; underlying, unsecured lesion (e.g., arteriovenous malformation, aneurysm, tumor); hemorrhagic conversion of an acute ischemic stroke; infratentorial location of ICH; large intraventricular hemorrhage requiring treatment as a result of mass effect or shift; extension of hemorrhage into the midbrain; absolute requirement for long-term anticoagulation; coagulopathy; platelet count < 100,000 cells/mm3; INR > 1.4; elevated activated partial thromboplastin time (aPTT); presenting GCS < 4, high risk for ischemic stroke; emergent need for surgical decompression; inability to give consent for the procedure; is pregnant, breast-feeding, or displays a positive pregnancy test; evidence of active infection; or any comorbid disease or condition expected to compromise survival.
3. Positioning and Planning
- Administer general anesthesia to the patient using standard techniques.
- Maintain sterile conditions throughout the entire procedure by sterilely preparing the skin and draping the surgical area.
- Plan the trajectory of the evacuation by using preoperative volumetric imaging to draw a line along the long axis of the hematoma to the outer surface of the skull, so that the tip of the sheath will sit 1 to 2 cm from the distal end of the hematoma.
NOTE: Important things to keep in mind when planning the trajectory are to minimize disruption to brain tissue (especially eloquent structures) and avoid any vasculature visible on non-invasive imaging. Trajectory planning is performed on stereotactic navigation software, which varies by institution. A comparison of commonly used navigation systems in ICH evacuation is available in the medical literature14. - Place the patient in the correct anatomical position depending on the location of the hematoma.
NOTE: Most cases (80%) are performed supine, and a minority are performed prone (15%) or supine with the head turned (5%).
4. Opening
- Make a 2 cm linear, horizontal incision along the skin within a natural skin crease.
- Open the skull with a 1- to 1.2 cm-diameter craniectomy using a high-speed drill with a 5 mm cutting burr. Attempt to align the craniectomy with the long axis of the hematoma, but avoid midline structures and eloquent brain territories.
NOTE: A perforator is larger than necessary, especially if the defect is on the forehead. - If the trajectory is not perfectly perpendicular to the skull, drill a cylinder in the bone along the planned trajectory to ensure optimal mobility of the sheath and endoscope within the craniectomy, but be aware that the intersection with the dura will not be perpendicular.
- Use bone wax, gel foam in thrombin, and bipolar cautery to achieve hemostasis.
- Visualize the underlying hematoma using ultrasound to confirm its size and position.
NOTE: Ultrasound quality is highest using the burr hole transducer in a wet field prior to durotomy. - Open the dura in a cruciate fashion and cauterize the dural leaves to within a millimeter of the bone edge.
NOTE: Avoid large veins or arteries. - Incise the pia mater 1 cm with a #11 blade prior to cauterizing. If obtaining a brain biopsy, this is the ideal time. Use tumor forceps or the cup end of a Penfield 1 instrument. Avoid cautery until the biopsy is obtained.
- Cauterize the pial incision and underlying cortex with bipolar cautery.
5. Phase 1 Evacuation
- Insert the introducer sheath along the planned trajectory with a navigation stylet positioned within the sheath. The stylet provides live feedback on the location of the tip.
NOTE: Given the small size of the 1 cm craniectomy, a "trans-sulcal" approach is often not possible; therefore, the pia is incised and entered in a non-vascular space immediately below the craniectomy. - If the clot is particularly fibrous and resistance is encountered, make a slight adjustment to the sheath to reach the target point.
- Remove the introducer and navigation probe once the target point is reached, 1-2 cm from the distal end of the hematoma.
NOTE: Some operators prefer to use stereotactic navigation registered to the endoscope rather than the sheath introducer for continuous navigation. - Note the position of the sheath by marking it at the level of the skin.
NOTE: If the pressure within the hematoma is high, liquid may flow out of the sheath during this step. - Prepare the endoscope by activating the preferred settings including white balance, brightness, filter, and light intensity.
- Attach the irrigation tubing from a 2 L saline bag at shoulder height to the left working port, and set the flow rate to approximately 25% on the endoscope. Open the right port of the endoscope all the way, allowing for egress of irrigation fluid.
- Insert the endoscope into the sheath. Insert the wand inside the working channel of the endoscope and hold the wand with the dominant hand.
NOTE: For this phase of the procedure, maintain the endoscope and wand within the end of the sheath, approximately 0.5 to 2 cm from the end of the sheath. - Use the pointer finger to buffer the distance between endoscope and the wand's handle to maintain constant awareness of the location of the tip of the device within the sheath.
- Set the suction strength of the aspiration system to 100% and set the irrigation flow rate to low (~25%).
- Aspirate any liquid hematoma that presents itself at the end of the sheath while keeping the wand within the distal 1 cm of the sheath.
NOTE: As the hematoma is aspirated, the cavity will collapse inward due to the reduced mass effect. Constant irrigation maintains the structure of the cavity during phase 2 of the evacuation. - If a solid clot is encountered that does not aspirate with suction alone, activate the bident within the wand to digest the clot.
- If a piece of clot is too large or fibrous for suction and adheres to the tip of the wand, withdraw the entire endoscope and wand together with the clot, taking care not to dislodge the clot from the wand.
NOTE: This is called the ADAPT (A Direct Aspiration First Pass Technique) technique, referencing the practice of removing intravascular thrombus during thrombectomy for acute ischemic stroke15. - If the piece of clot is particularly large with a fibrous capsule and the prior two techniques do not work, use the working channel of the endoscope as an additional suction.
- To accomplish this, attach a conventional surgical suction to the second endoscope port (usually the irrigation outflow pathway) with the valve closed but ready for activation. Draw the large piece of clot into the end of the sheath using the wand. Close the irrigation inflow port and open the outflow port to activate maximal surgical suction. The clot is now stuck on the tip of the wand, endoscope, and sheath. Remove the wand, endoscope, sheath, and clot together.
NOTE: This is called the double ADAPT technique.
- To accomplish this, attach a conventional surgical suction to the second endoscope port (usually the irrigation outflow pathway) with the valve closed but ready for activation. Draw the large piece of clot into the end of the sheath using the wand. Close the irrigation inflow port and open the outflow port to activate maximal surgical suction. The clot is now stuck on the tip of the wand, endoscope, and sheath. Remove the wand, endoscope, sheath, and clot together.
- If the clot has a fibrous capsule and is difficult to separate from the brain tissue, use the tip of the sheath as a blunt dissector.
NOTE: This is called the sheath dissection technique. - After aspirating, explore the cavity at the same depth by gently pivoting the sheath laterally until no residual clot remains at that depth.
- Withdraw the sheath 1 cm and repeat the aspiration steps of Phase 1 until the sheath reaches the proximal wall of the cavity.
6. Phase 2 Evacuation
- Decrease the suction of the wand to 25% and increase the irrigation to 100% to improve visibility in the cavity. Explore for residual hematoma and identify bleeding arteries.
NOTE: During this phase, it is critical to minimize suction forces on the pericavity brain tissue. - Aspirate any residual hematoma in a targeted fashion with low aspiration power, taking care to not damage surrounding brain matter which can incur additional bleeding or cause trauma to the cavity wall.
NOTE: Blood products may initially interfere with optimal visualization but the cavity will clear with patient, continuous irrigation. If the cavity does not clear, identify and cauterize the bleeding vessels. - Monitor for any bleeding vessels and address them accordingly with the following steps:
- If small bleeding vessels are challenging to visualize, direct a consistent flow of irrigation towards the vessel by hovering immediately over the bleeding site with the sheath and pulling the scope back from the tip. Once the vessel is better visualized, cauterize the vessel.
NOTE: This is called the sheath hover technique. - Irrigate the cavity until hemostasis is achieved
- Apply pressure using the end of the sheath if pure irrigation does not work.
- Utilize bipolar cautery if the previous two methods do not work.
- Aspirate any residual hematoma along the sides or in the crevices of the cavity.
NOTE: This step becomes easier once the bleeding vessels are addressed, permitting clear visualization.
- If small bleeding vessels are challenging to visualize, direct a consistent flow of irrigation towards the vessel by hovering immediately over the bleeding site with the sheath and pulling the scope back from the tip. Once the vessel is better visualized, cauterize the vessel.
- Ensure that the cavity is cleared of all visible hematomas and bleeding vessels.
NOTE: If there is a thin layer of fresh clot from intraoperative bleeding, beware of causing more bleeding in the attempted aspiration of this thin coating of fresh blood. Aspirate gently or leave the fresh blood in place. Differentiating clots from the cavity wall is a major challenge in the procedure.
7. Assessment and Closure
- Withdraw the endoscope and sheath slowly, with the endoscope at the tip of the sheath to examine the tract walls upon exiting to monitor for additional bleeding.
NOTE: Some operators have advocated for infusing thrombin into the cavity at this point, either by adding thrombin directly to the irrigation fluid or by injecting gelatin mixed with thrombin through the endoscope. This is a reasonable option, but injecting gelatin mixed with thrombin will make ultrasound imaging impossible. - Use burr hole ultrasound to assess for residual hematoma or active bleeding.
NOTE: Ultrasound is useful for settling doubt about any questionable regions as well as detecting any large areas of residual hematoma that may have been missed under direct visualization. - Perform an intraoperative Dyna-CT scan if available to evaluate the degree of evacuation.
NOTE: The goal of the procedure is to obtain at least 80% evacuation. If there is more than 20% of the hematoma remaining, restart Phase 2 of the evacuation before proceeding to the following steps. - Apply hemostatic gel foam in the burr hole over the surface of the brain.
- Cover the craniectomy using a titanium plate and secure it using titanium screws.
- Close the galea and subcutaneous layers using 3-0 polyglactin 910 sutures.
- Close the skin using 4-0 poliglycaprone 25 subcuticular stitches, followed by skin closure surgical tape strips.
Results
The SCUBA evacuation technique has been described in 47 patients undergoing endoscopic ICH evacuation between December 2015 and September 2017. The mean pre-operative ICH volume was reported as 42.6 cm3 (standard deviation = 29.7 cm3; mean post-operative ICH volume = 4.2 cm3, SD 6.6 cm3), resulting in a mean evacuation rate of 88.2% (SD 20.8%) (Table 1). An example of pre-operative and post-operative CT scans is shown in Figure 1. In 23 (48.9%) cases, active bleeding vessels were detected, and in 12 (52.2%) of these cases, bleeding emanated from more than one vessel (Table 2). Bleeding was addressed using irrigation alone in 5 cases (10.6%) and electrocautery in 18 cases (38.3%) (Table 2).Post-operative bleeding was isolated to only a single case (2.1%) in which the routine head CT performed on post-operative day 1 demonstrated that the evacuation cavity had refilled with hemorrhage that appeared to originate from a superficial galeal vessel bleeding into the access tract and cavity (Table 2). This patient's examination did not worsen, and he did not require additional surgery.
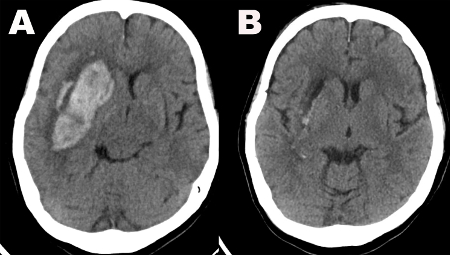
Figure 1: CT scans. (A) Pre-operative CT head image demonstrates a large right basal ganglia hemorrhage. (B) CT head image performed on post-operative day 1 demonstrates near-complete evacuation of the hematoma. Please click here to view a larger version of this figure.
| Variable | Mean | Standard Deviation |
| Preoperative Volume | 42.6 | 29.7 |
| Postoperative Volume | 4.2 | 6.6 |
| Evacuation Percentage | 88.2% | 20.8% |
Table 1: Evacuation details. ICH volumes and evacuation rates for the SCUBA procedure.
| Variable | Number | Percent |
| Total Patients | 47 | - |
| Active Bleeding Identified | 23 | 48.9% |
| Single Vessels | 11 | 23.4% |
| Multiple Vessels | 12 | 25.5% |
| Irrigation | 5 | 10.6% |
| Electrocautery | 18 | 38.3% |
| Postoperative Bleeding | 1 | 2.1% |
Table 2: Operative details. Operative details (specifically bleeding vessels) encountered during the SCUBA procedure.
Discussion
There are several operative best practices to learn and implement during endoscopic ICH evacuation. First and foremost, it is critical to minimize disruption to brain tissue whenever possible. Accomplishing this starts with optimizing the surgical trajectory so that the sheath traverses the shortest possible course while avoiding eloquent structures. For supratentorial ICH, eloquent structures include the supplementary motor area, primary motor and sensory cortices, left superior temporal and angular gyri, and primary visual cortex. In addition, the trajectory should align with the longitudinal axis of the hematoma. The benefits of this strategy include maximizing visualization of the cavity, minimizing torque force on the brain adjacent to the access tract, increasing the likelihood of being able to view the extremes of the cavity, and creating the shortest possible trajectory to the clot, thus minimizing brain trauma.
In addition to minimizing brain tissue disruption, it is also important to minimize distortion of the hematoma cavity. Aspiration in an enclosed cavity can distort elastic brain matter as much as compressive forces with equal damage. To avoid this, suction strength should be at the minimal possible level necessary for effective blood clot aspiration. This is especially important if the tip of the wand is advanced beyond the tip of the sheath. The only time the suction strength should be on high is during Phase 1, when the tip is in direct contact with the clot. The suction strength should decrease as the procedure progresses.
Unfavorable outcomes have been reported when irrigation during an endoscopic intraventricular hematoma evacuation leads to increased intracranial pressure16. The SCUBA procedure avoids this by evacuating the hematoma in Phase 1, which decreases pressure within the cavity, and subsequently irrigating in Phase 2. In Phase 2, the endoscope has a second access port that allows for irrigation outflow, thus avoiding over-distension of the hematoma cavity and elevated intracavitary pressure. In addition, the sheath and tract do not form a watertight seal and irrigation fluid is lost around the sheath.
Achieving and maintaining hemostasis during Phase 2 is a crucial requirement for a successful SCUBA evacuation. It is important to meticulously monitor every wall of the cavity for bleeding vessels and address them accordingly with continuous irrigation or bipolar cautery. Achieving perfect hemostasis ensures that there is minimal risk of postoperative re-bleeding.
Since clear, direct visualization of the residual hematoma within the cavity may not always be possible during the procedure, it is a best practice to check the evacuation with intraoperative imaging after Phase 2. There have been several cases in which direct endoscopic examination suggested that the cavity was clear but residual hematoma was detected on either intraoperative ultrasound or DYNA CT, leading to another pass with the sheath into the cavity and additional hematoma evacuation.
At this early stage in the development of this procedure, there is strong enough evidence to suggest what the lower limit of evacuation percentage of residual clot volume should be. Though there are currently no studies evaluating outcomes for endoscopic procedure evacuation percentages, animal models and the MISTIE trial suggest that increased evacuation is preferred9. In ICH-induced mice, molecules in the blood such as iron had a toxic effect on the surrounding brain tissue, while iron chelators reduced the damage17. The MISTIE II trial found that perihematomal edema volume was smallest when evacuation percentage exceeded 65%, larger when evacuation percentage ranged from 20-65%, and largest when evacuation percentage was less than 20%18. This data also suggest that outcome may improve with higher evacuation percentage, but the study was not powered to assess this feature. The MISTIE phase III, ENRICH, INVEST, and/or MIND trials may shed light on this question.
One area that remains to be resolved is the timeframe of the procedure. Many protocols advocate evacuation within 72 hours and after a 6-hour stability scan to ensure that the hematoma is stable. Many physicians choose this course of action, as a small study from 2004 reported complications, re-bleeding, and poor outcomes in a small series of patients who underwent craniotomy for ultra-early surgery19. More recent studies on minimally invasive endoscopic evacuation have reported good outcomes with ultra-early evacuation20,21. Manuscripts reporting endoscopic evacuation suggest that bleeding is identifiable and controllable in ultra-early evacuations. The ENRICH study protocol requires evacuation within 24 hours of ictus and does not mandate a stability scan. Ultra-early surgery may be an option in the future, but additional studies are necessary to assess the risks and benefits of ultra-early evacuation.
The SCUBA procedure is a minimally invasive intracerebral hemorrhage evacuation technique that involves an endoscope using an aspiration system. Preliminary evidence suggests that the SCUBA technique can be performed safely and reliably results in a high evacuation percentage. Further studies are necessary to assess the impact of this procedure on functional outcomes.
Disclosures
Christopher Kellner received an educational grant from Penumbra for a CME course to teach endoscopic minimally invasive intracerebral hemorrhage evacuation. J Mocco is a co-principal investigator on the INVEST trial, which is a feasibility study to evaluate endoscopic minimally invasive intracerebral hemorrhage evacuation funded by Penumbra. J Mocco is an investor and consultant for Rebound Therapeutics.
Acknowledgements
This research was supported in part by a grant from Arminio and Lucyna Fraga and a grant from Mr. and Mrs. Durkovic.
Materials
| Name | Company | Catalog Number | Comments |
| Artemis Device 2.8mm | Penumbra Inc. | AP28 | Cannula Outer Diameter: 2.8mm. Cannula Length: 27cm. Aspiration Tubing Length: 9.5ft; The Food and Drug Administration (FDA) approved the Apollo System in 2014 for use in intraventricular hemorrhage (IVH) evacuation but its indication now includes ICH and the Artemis System was approved for the same IVH and ICH evacuation in 2017. |
| Artemis Device 2.1mm | Penumbra Inc. | AP21 | Cannula Outer Diameter: 2.1mm. Cannula Length: 26cm. Aspiration Tubing Length: 9.5ft |
| Artemis Device 1.5mm | Penumbra Inc. | AP15 | Cannula Outer Diameter: 1.5mm. Cannula Length: 27cm. Aspiration Tubing Length: 9.5ft |
| MAX Canister | Penumbra Inc. | APCAN2 | |
| Pump MAX 110V | Penumbra Inc. | PMX110 | |
| 19-French Sheath | Aesculap USA | FH604SU | Outer Diameter: 6.33mm |
| Storz Lotta 3-port Endoscope | Karl Stortz | 28164 LA / 28164 LS | Outer Diameter: 6.1mm. Two ports for irrigation/suction (1.6mm). One working channel (2.9mm) |
| Medtronic AxiEM | Medtronic | UC201403939 | An advantage of the Medtronic AxiEM system is it does not require pinning or line-of-site navigation. |
| High-speed drill with 5-mm cutting burr | Medtronic | 9BA60 | |
| Bone Wax | Ethicon | W31 | |
| Hemostatic Gel Foam with Thrombin | J&J Healthcare | 2994 | |
| Bipolar Cautery | State of the Art | 401102 | |
| Aloka burr hole ultrasound transducer | Aloka | UST-52114P | |
| 11-blade | Bard Parker | 372611 | |
| Penfield 1 instrument | Sklar Corp | 47-2255 | |
| AxiEM stylet | Medtronic | 9735428 | |
| Titanium plate | Depuy Synthes | 04503023/04503024 | |
| Titanium screws | Depuy Synthes | 0450310301/0450310401 | |
| DYNA CT on the Artis Q | Siemens Healthineers | A91AX-01343-33C1-7600 | |
| 3-0 Vicryl sutures | Ethicon | J416 | |
| 4-0 monocryl subcuticular stitches | Ethicon | Y426 | |
| Steri-Strips | 3M | R1547 |
References
- van Asch, C. J. J., Luitse, M. J. A., Rinkel, G. J. E., vander Tweel, I., Algra, A., Klijn, C. J. M. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurology. 9 (2), 167-176 (2010).
- Qureshi, A. I., Mendelow, A. D., Hanley, D. F. Intracerebral haemorrhage. Lancet. 373 (9675), 1632-1644 (2009).
- Aguilar, M. I., Freeman, W. D. Spontaneous intracerebral hemorrhage. Seminars in Neurology. 30 (5), 555-564 (2010).
- Broderick, J., et al. Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Circulation. 116 (16), 391-413 (2007).
- Caceres, J. A., Goldstein, J. N. Intracranial hemorrhage. Emergency Medicine Clinics of North America. 30 (3), 771-794 (2012).
- Poon, M. T. C., Fonville, A. F., Al-Shahi Salman, R. Long-term prognosis after intracerebral haemorrhage: systematic review and meta-analysis. Journal of Neurology, Neurosurgery, and Psychiatry. 85 (6), 660-667 (2014).
- Mendelow, A. D., Gregson, B. A., Rowan, E. N., Murray, G. D., Gholkar, A., Mitchell, P. M. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): a randomised trial. Lancet. 382 (9890), 397-408 (2013).
- Mendelow, A. D., et al. Early surgery versus. initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet Neurology. 365 (9457), 387-397 (2005).
- Hanley, D. F., et al. Safety and efficacy of minimally invasive surgery plus alteplase in intracerebral haemorrhage evacuation (MISTIE): a randomised, controlled, open-label, phase 2 trial. Lancet Neurology. 15 (12), 1228-1237 (2016).
- Wang, W. -. Z., et al. Minimally invasive craniopuncture therapy vs. conservative treatment for spontaneous intracerebral hemorrhage: results from a randomized clinical trial in China. International Journal of Stroke. 4 (1), 11-16 (2009).
- Fiorella, D., Arthur, A. S., Mocco, J. D. The INVEST Trial: A Randomized, Controlled Trial to Investigate the Safety and Efficacy of Image-Guided Minimally Invasive Endoscopic Surgery With Apollo vs ..... Neurosurgery. , (2016).
- Fiorella, D., Arthur, A., Bain, M., Mocco, J. Minimally Invasive Surgery for Intracerebral and Intraventricular Hemorrhage: Rationale, Review of Existing Data and Emerging Technologies. Stroke. 47 (5), 1399-1406 (2016).
- Kellner, C. P., et al. The Stereotactic Intracerebral Hemorrhage Underwater Blood Aspiration (SCUBA) technique for minimally invasive endoscopic intracerebral hemorrhage evacuation. Journal of NeuroInterventional Surgery. , (2018).
- Chartrain, A. G., et al. A review and comparison of three neuronavigation systems for minimally invasive intracerebral hemorrhage evacuation. Journal of NeuroInterventional Surgery. , (2017).
- Turk, A. S., et al. ADAPT FAST study: a direct aspiration first pass technique for acute stroke thrombectomy. Journal of NeuroInterventional Surgery. 6 (4), 260-264 (2014).
- Trnovec, S., Halatsch, M. -. E., Putz, M., Behnke-Mursch, J., Mursch, K. Irrigation can cause prolonged intracranial pressure elevations during endoscopic treatment of intraventricular haematomas. British Journal of Neurosurgery. 26 (2), 247-251 (2012).
- Wu, H., Wu, T., Xu, X., Wang, J., Wang, J. Iron toxicity in mice with collagenase-induced intracerebral hemorrhage. Journal of Cerebral Blood Flow & Metabolism. 31 (5), 1243-1250 (2011).
- Mould, W. A., et al. Minimally invasive surgery plus recombinant tissue-type plasminogen activator for intracerebral hemorrhage evacuation decreases perihematomal edema. Stroke. 44 (3), 627-634 (2013).
- Morgenstern, L. B., Demchuk, A. M., Kim, D. H., Frankowski, R. F., Grotta, J. C. Rebleeding leads to poor outcome in ultra-early craniotomy for intracerebral hemorrhage. Neurology. 56 (10), 1294-1299 (2001).
- Nishihara, T., et al. Newly developed endoscopic instruments for the removal of intracerebral hematoma. Neurocritical Care. 2 (1), 67-74 (2005).
- Nagasaka, T., Inao, S., Ikeda, H., Tsugeno, M., Okamoto, T. Inflation-deflation method for endoscopic evacuation of intracerebral haematoma. Acta Neurochirurgica. 150 (7), 685-690 (2008).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved