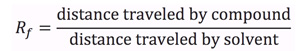Performing 1D Thin Layer Chromatography
Overview
Source: Laboratory of Dr. Yuri Bolshan — University of Ontario Institute of Technology
Thin layer chromatography (TLC) is a chromatographic method used to separate mixtures of non-volatile compounds. A TLC plate consists of a thin layer of adsorbent material (the stationary phase) fixed to an appropriate solid support such as plastic, aluminum, or glass1. The sample(s) and reference compound(s) are dissolved in an appropriate solvent and applied near the bottom edge of the TLC plate in small spots. The TLC plate is developed by immersing the bottom edge in the developing solvent consisting of an appropriate mobile phase. Capillary action allows the mobile phase to move up the adsorbent layer. As the solvent moves up the TLC plate, it carries with it the components of each spot and separates them based on their physical interactions with the mobile and stationary phases.
Procedure
1. TLC Plates
- Common adsorbents for TLC are silica gel, alumina, and cellulose. TLC plates are commercially available with a variety of properties. Choose a TLC plate and cut it down to an appropriate size (approximately 5 cm x 5 cm is sufficient for most applications). For glass-backed TLC plates, score the glass using a ruler and a glass cutter, then carefully break along the line.
2. Spotting
- Dissolve the sample in a suitable solvent to make an approximately 1
Results
An example of a typical TLC plate is shown in Figure 1. An unknown compound 'A' may be compared to known standards 'B' through 'E'. Determination of the Rf value for each component is achieved by spotting of each respective compound, developing the TLC plate, and visualization. The Rf of unknown compound 'A' is calculated by measuring the spot height (y) and dividing by the solvent height (z). Comparing this
Application and Summary
TLC has a number of practical applications in the laboratory. TLC may be used to identify unknown compounds and unknown components of mixtures via comparison with standards. TLC is commonly used to monitor the course of a chemical reaction, and to assess the purity of the product through the comparison of relative amounts of reactants, products, and by-products on successive chromatograms over time. TLC can also be used to determine if a substance purified by other methods (such as recrystallization or distillation) stil
Tags
Skip to...
Videos from this collection:

Now Playing
Performing 1D Thin Layer Chromatography
Organic Chemistry
290.2K Views

Introduction to Catalysis
Organic Chemistry
34.7K Views

Assembly of a Reflux System for Heated Chemical Reactions
Organic Chemistry
168.5K Views

Conducting Reactions Below Room Temperature
Organic Chemistry
70.7K Views

Schlenk Lines Transfer of Solvents
Organic Chemistry
41.7K Views

Degassing Liquids with Freeze-Pump-Thaw Cycling
Organic Chemistry
56.4K Views

Preparing Anhydrous Reagents and Equipment
Organic Chemistry
79.4K Views

Purifying Compounds by Recrystallization
Organic Chemistry
710.5K Views

Separation of Mixtures via Precipitation
Organic Chemistry
158.1K Views

Solid-Liquid Extraction
Organic Chemistry
238.2K Views

Rotary Evaporation to Remove Solvent
Organic Chemistry
213.0K Views

Fractional Distillation
Organic Chemistry
334.9K Views

Growing Crystals for X-ray Diffraction Analysis
Organic Chemistry
32.9K Views

Column Chromatography
Organic Chemistry
361.2K Views

Nuclear Magnetic Resonance (NMR) Spectroscopy
Organic Chemistry
249.1K Views
Copyright © 2025 MyJoVE Corporation. All rights reserved