配合化学物
Overview
资料来源: 实验室的博士尼尔 · 艾布拉姆斯 — — 美国纽约州立大学环境科学与林业学院
过渡金属随处可见维生素补充剂对电镀。过渡金属也弥补许多涂料中颜料和撰写所有矿物。通常情况下,过渡金属阳离子的形式发现因为他们容易氧化,或失去电子,和被包围的电子给体称为配体。这些配体做不形成离子或金属中心的共价键,而是他们在第三种类型的已知的债券作为坐标共价键。一种配体与金属之间的坐标共价键债券是动态的意思配体是不断交换和重新协调围绕金属中心。金属和配体的身份决定了哪个配体将优先债券在另一个。此外,颜色和磁性质也是形成的配合物的类型。使用各种各样的仪器和工具,分析了形成的配合物。这项实验揭示了为什么这么多配合物是可能和使用化学光谱法 (颜色和化学) 的方法来帮助识别形成的配合物的类型。
Principles
协调配合
协调配合物具有至少一种金属复杂,其中包含一个金属中心和包围电子基团的配体。这就是所谓的复杂离子。反离子平衡复杂离子形成分子配合物的电荷。协调配合物溶于水中,金属离子络合物与反离子离解。金属离子和配体表现得像一个多原子离子和不游离。
复杂的离子的几何形状对包括线性,方形的标准价层电子对互斥理论 (VSEPR) 几何平面、 四面体,和八面体。八面体复杂离子是最常见的几何。晶体场理论解释了能量分裂之间d-过渡金属离子和 VSEPR 几何图形的轨道。能级分裂受的形状和方向的d-眶裂片。
配体和光谱化学序列
配体划分,债券或附件的数量,他们可以用一个金属的中心。单个附件被称为单齿 (一个齿)。称为双齿配体,使两个附件 (两齿),和三个附件被称为分叉。配体捐赠电子密度对金属中心形成坐标共价键的债券。配体可能带电的或中性。配体被列为被强或弱根据光谱化学序列:
(弱)我- < Br- < Cl- < SCN- < F- < OH- < 牛2-< ONO- < H2O < NCS- < 乙二胺四乙酸4- < NH3 < en < 没有2- < CN- (强)
分裂的轨道
当六个配体接近金属中心形成八面体的复合体时,五个简并的 d 轨道分裂成三个低能量简并 t2 g轨道和两个更高的能量简并 eg轨道。根据光谱化学系列配体的强度取决于分裂 t2 g和 eg轨道之间的距离。
洪特规则仍然适用,电子填充轨道一在一段时间,但他们填写的分裂 t2 g和 eg轨道大小符合。如果分手可以很小,电子会填满所有轨道单独在配对之前。这最大限度地不成对电子的数量,被称为高自旋。同样,强场原因大 t2 g-eg拆分: 电子配对在 t2 g设置填充高能量 eg轨道之前。这最小化不成对电子的数量,被称为低自旋。对电子驱动器受能源 (或大小) 的轨道分裂相比能量的电子配对。如果配对的能量是高相比将移入 eg轨道能量,电子的高自旋。如果相比移动到更高的 eg轨道的能量,能量的配对较低,电子是低自旋。
电子必须从较低的 t2 g状态移动到更高的 eg状态在金属中心的距离决定复杂吸收的电磁波辐射的能量。如果这种能量是在可见光区域 (400-700 毫微米,1.77 eV-3.1 eV),复杂一般有一种颜色。弱场配体 (我- → OH-) 导致小分裂和配合物吸收低能量的光 (即红色) 的显示颜色是绿色。强场配体 (乙二胺四乙酸 → CN-) 吸收高能光 (即蓝紫光) 和出现红、 黄颜色。配合物与配体的强与弱对光谱化学序列,如氨,是可以采用任何弱或强场的几何。
颜色-配体的关系是名称"光谱化学序列"的理由。成对或不成对电子的数量也引起了顺磁性磁性金属络合物的研究。
当四个配体协调围绕一个金属的中心时,可以导致方形平面或四面体的复杂。在四面体复合体的轨道能量被翻转相比八面体配合物,与能量低于 t2 geg 。这是因为 d 轨道方面协调配体的取向。在平面正方形的配合物,有几个不同的轨道能量,与 dyz和 dxz正在退化和最低的能量 (低于 dz2,),然后 dxy,和最后最高能量 dx2 y2轨道。
结构和颜色
因为在轨道分裂距离随配体强度,相同的金属中心协调配合可以有各种颜色根据配体的协调。例如,水溶液的镍 (H2O)62 +颜色为浅绿色,但镍 (NH3)62 +是深蓝色。颜色来自能量变化 t2 g-eg轨道之间。NH3是强场配体,推轨道进一步彼此以及取代从金属中心的 H2O 配体。颜色和配合在这个实验中,我们将进一步探索配体的影响。
Procedure
1.镍配合物的颜色
- 倪 (H2O)62 +复杂 (图 1a)
- 法制备镍 (H2O)62 + 1 M 的溶液溶解美国4在适当体积的水。
- 进一步稀释镍 (H2O)62 +解决方案 70 毫升的 1 M 溶液加入去离子水温 1000 毫升左右。
- 划分的镍 (H2O)62 + 7 400 毫升烧杯中。
- 水性镍溶液对颜色为浅绿色,因为水是一种弱场配体。
- 吸光度光谱表明,红色的波长被吸收,证明与之相反,绿色,被观察。
- 倪 (NH3)62 +复杂 (图 1b)
- 将 5 M 氨性溶液添加到一个烧杯和搅拌。
- 该解决方案呈现出深蓝色的颜色,指示解决方案吸收的橙色光能量大于红光。
- 吸光度光谱表明黄色波长被吸收,证明与之相反,蓝色,这观察。
- 氨是强场配比水,增加 t2 g和 eg轨道之间的分裂。
- Ni(en)32 +复杂 (图 1 c)
- 将 30%乙二胺 (en) 解决方案添加到的水性镍 (H2O)62 +复杂和搅拌。
- 该解决方案逐步转向从光蓝色紫色作为分子逐步协调围绕最终形成 Ni(en)3 +金属中心的乙二胺。
- 乙二胺是更强的配比水或氨和它是双齿。紫色的颜色指示解决方案是吸收黄灯为高能量比橙色或红色的光。
- 吸光度光谱表明黄色波长被吸收,证明与之相反,紫色,这观察。
- Ni(dmg)22 +复杂 (图 1 d)
- Dimethylgloxine (dmg) 是一个双齿配体,大量的金属螯合物。只有两个 dmg 分子需要每个金属中心因为 Ni(dmg)22 +具有广场平面几何形状。
- 向水复合添加 1%的伤害。
- 固体的粉色/红色沉淀形成,不溶性 Ni(dmg)22 +复杂。
- 可见光透射谱的复杂是不可能的但红颜色指示绿灯被吸收。绿色是更高的能量比黄色、 橙色和红色。
- Ni(CN)42-复杂 (图 1e)
- 氰根离子 (CN-) 是单齿,但非常强场配体,也形成镍 (II) 广场平面配合。
- 添加 1 M KCN 溶液。
- 黄色 Ni(CN)42-复杂形式几乎立即。
- 注: 与氰化物盐工作必须非常小心。加入酸可能导致氰化物气体的形成。
- 氰化物是比任何其他配强配体是 σ-键合的配位体对金属和 π-背板粘接从金属到配体。黄色的颜色指示解决方案是吸收蓝光,在能量高于绿色、 黄色、 橙色和红色。
- 吸光度光谱表明黄色波长被吸收,证明与之相反,紫色,这观察。
2.配体强度
- 光谱化学序列,根据一些配体是比别人更强场对应于分裂的中心金属离子的 d 轨道的大小。
- 强场配体取代弱场配体在溶液中。
- 硫酸镍水溶液外观呈浅绿色因为镍 (H2O)62 +复杂的形式。
- 按顺序解的乙二胺、 氨氮、 氰化物、 丁二酮肟,向解决方案中添加含镍边搅拌。
- 在每个加法以后, 以前的颜色消失,出现新的颜色。
- 颜色的变化表明新的配合物的配体力量的推动下形成。这些是可以量化的每个反应的平衡常数:
倪 (H2O)62 +(aq) + 6 NH3 (aq) → 镍 (NH3)62 + (aq) + 6 H2OK情商= 1.2 x 109
倪 (NH3)62 + (aq) + 3 en(aq) → Ni(en)32 + (aq) + 6 NH3 (aq) K情商= 1.1 x 109
Ni(en)32 + (aq) + 2 Hdmg(aq) → Ni(dmg)2 (s) + 3 en(aq) ++ (aq) K 2 H情商= 1.35 x 105
Ni(dmg)2 (s) + 4 CN- (aq)-→ Ni(CN)4-2 (aq) + 2 dmg- (aq) K情商= 6.3 x 107 - 在每个反应的平衡常数是很大 (> 1),表明反应驱动的所有产品。
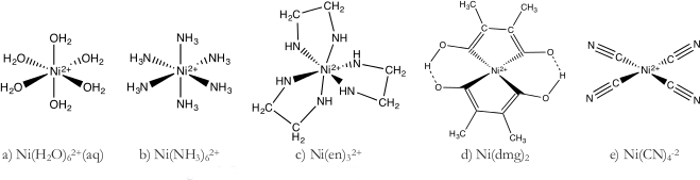
图 1。镍 (II) 配合物的电子结构。
Application and Summary
从人的颜料,过渡金属人遍布的化学、 生物学、 地质学和工程领域。理解的不同化学状态下过渡金属行为可以作为监测颜色或磁行为一样简单。几乎每个 3d(4 行)过渡金属是重要的生理功能,在所有情况下,这些金属因有义务配体形成配合物。例如,铁对于所有脊椎动物的氧气运输至关重要。血红蛋白,复杂的蛋白,包含四个血红素亚基与 Fe2 +在每个中心。在血红蛋白,Fe2 +是螯合四环和组氨酸残基,使它方形金字塔 (五边)。当氧气存在时,亚基成为八面体。O2被认为是一种强场配体,导致大 d 轨道 t2 g-eg分裂,使其成为低自旋。它要求促进电子 eg状态,所以蓝色光被吸收使氧 (动脉) 血液呈亮红色相对高能光。相比之下,缺氧 (静脉) 的血液有较小的 d 轨道劈裂和低能量红光被吸收,使氧的血液出现黑暗,略带紫色红色。在同样的尊重,一氧化碳,CO,是一种强场配体和将取代氧气。它给血液更明亮的红色外观,由于强场分裂。CO 在血液中的 O2的优惠绑定通常是致命的。
配位化学的另一个应用程序是在油漆和颜料。虽然许多颜料是简单的金属氧化物,如普鲁士蓝和酞菁蓝还有配合其颜色来自分裂在 d 轨道 (图 2)。普鲁士蓝,铁是由六个氰化物的配体,创建高自旋铁 (III) 铁氰化复杂,Fe(CN)63-包围。另一种化合物,酞菁蓝,是一个平面正方形的复杂与铜 (II) 离子在包围四酞菁分子的中心。
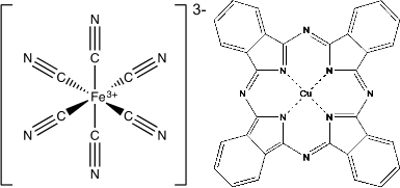
图 2。普鲁士蓝,以铁为中心协调复杂和酞菁蓝,一个铜中心协调复杂。
配位化合物有周围配体与金属离子中心和反离子平衡电荷。配体可以是单齿或螯合与二四附件站点。配体还通过光谱化学序列,分类配体来拆分金属 d 轨道的相对强度进行了分类。颜色和磁性受金属和配位体。大 d 轨道分裂需要大的能量,为促进电子融入更高能量的轨道和吸收高能光 (短波长)。这些是低自旋配合物,并有不成对的电子的最大数目。相比之下,小 d 轨道分裂被称为弱场和吸收低能量的光,以及具有最大数量的未成对电子。电荷和身份金属离子以及绑定的配体配位化合物在定义观察的颜色和磁性。
References
- Shakhashiri, B. Z.; G. E. Dirreen, G. E; Juergens, F. Color, Solubility, and Complex Ion Equilibria of Nickel (II) Species in Aqueous Solution. J. Chem. Ed. 52 (12), 900-901 (1980).
Tags
跳至...
此集合中的视频:

Now Playing
配合化学物
General Chemistry
91.7K Views

常见的实验室玻璃器皿和用途
General Chemistry
658.3K Views

溶液和浓度
General Chemistry
275.1K Views

固体和液体的密度测定
General Chemistry
556.8K Views

确定在水溶液中的质量百分组成
General Chemistry
383.8K Views

确定经验公式
General Chemistry
183.7K Views

确定离子化合物的溶度积的规则
General Chemistry
141.6K Views

使用 pH 计
General Chemistry
346.7K Views

滴定法简介
General Chemistry
425.4K Views

理想气体定律
General Chemistry
79.0K Views

平衡常数的分光光度法测定
General Chemistry
158.7K Views

Le Châtelier 原则
General Chemistry
265.8K Views

凝固点降低,以确定一种未知的化合物
General Chemistry
160.8K Views

确定率法律和秩序的反应
General Chemistry
196.3K Views

用焓差扫描量热法测量的变化
General Chemistry
44.7K Views
版权所属 © 2025 MyJoVE 公司版权所有,本公司不涉及任何医疗业务和医疗服务。