Complexes de coordination
Vue d'ensemble
Source : Laboratoire du Dr Neal Abrams — SUNY College of Environmental Science and Forestry
Métaux de transition sont partout de suppléments vitaminiques pour galvanoplastie. Aussi, métaux de transition forment des pigments dans les nombreuses peintures et composer tous les minéraux. En règle générale, les métaux de transition se trouvent sous la forme cationique puisqu’ils facilement oxydent, ou perdent des électrons et sont entourés par les donneurs d’électrons appelés ligands. Ces ligands ne pas forme ionique ou covalent liaisons avec le centre métallique, plutôt qu’ils prennent sur un troisième type de liaison connue comme covalentes coordonnées. La coordonnée-covalente entre un ligand et d’un métal est dynamique, ce qui signifie que les ligands sont continuellement échanger et re-coordination autour du centre métallique. Les identités des fois le métal et le ligand dicte les ligands adhère préférentiellement sur l’autre. En outre, couleur et propriétés magnétiques sont également en raison des types de complexes qui sont forment. Les composés de coordination qui se forment sont analysés en utilisant une variété d’instruments et d’outils. Cette expérience examine pourquoi tant de complexes sont possibles et utilise une méthode de spectrochimiques (couleur ou chimiques) pour aider à identifier le type de complexe qui se forme.
Procédure
1. nickel Complexes et couleurs
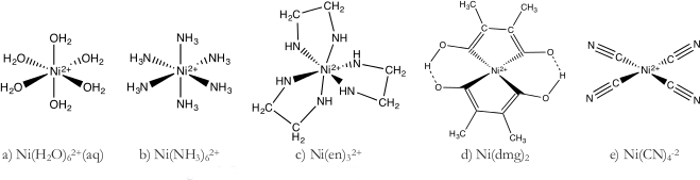
- Ni (H2O)62 + complexe (Figure 1 a)
- Préparer une solution de 1 M de Ni (H2O)62 + en dissolvant NiSO4 dans la quantité appropriée d’eau.
- Plus loin, diluer la solution de Ni (H2O)2 +6en ajoutant 70 mL de la solution 1 M à 1 000 mL d’eau désionisée.
- Diviser le6de Ni (H2O)2 + parmi sept béchers
Applications et Résumé
De pigments aux gens, métaux de transition sont trouvent dans tous les domaines de la chimie, la biologie, géologie et génie. Comprendre le comportement des métaux de transition sous différents États chimiques peut être aussi simple que la surveillance de couleur ou de comportement magnétique. Presque tous métaux de transition 3d (4ème ligne) est essentielle pour une fonction physiologique et, dans tous les cas, ces métaux est tenus par des ligands de forme complexes de coordination. Par exemple, le...
References
- Shakhashiri, B. Z.; G. E. Dirreen, G. E; Juergens, F. Color, Solubility, and Complex Ion Equilibria of Nickel (II) Species in Aqueous Solution. J. Chem. Ed. 52 (12), 900-901 (1980).
Tags
Passer à...
Vidéos de cette collection:

Now Playing
Complexes de coordination
General Chemistry
91.7K Vues

Présentation et utilisation de la verrerie de laboratoire courante
General Chemistry
658.3K Vues

Solutions et Concentrations
General Chemistry
275.1K Vues

Déterminer la densité d'un solide et d'un liquide
General Chemistry
556.8K Vues

Déterminer la concentration massique d'une solution aqueuse
General Chemistry
383.8K Vues

Déterminer la formule empirique
General Chemistry
183.7K Vues

Détermination des règles de solubilité des composés ioniques
General Chemistry
141.6K Vues

Utiliser un pH-mètre
General Chemistry
346.7K Vues

Introduction au titrage
General Chemistry
425.4K Vues

Loi des gaz parfaits
General Chemistry
79.0K Vues

Détermination spectrophotométrique d'une constante d'équilibre
General Chemistry
158.7K Vues

Principe de Le Châtelier
General Chemistry
265.8K Vues

Identification d'un composé inconnu grâce à la loi de la cryométrie
General Chemistry
160.8K Vues

Détermination de la loi de vitesse et de l'ordre de la réaction
General Chemistry
196.3K Vues

Utilisation de la calorimétrie différentielle à balayage pour mesurer les changements d'enthalpie
General Chemistry
44.7K Vues