標準添加法
概要
ソース: 博士ポール亭 - パデュー大学講座
標準の追加のメソッドは、目的の標本にマトリックス効果、どこ追加コンポーネントが減らすか、または試料の吸光度信号を高めるために複数のコンポーネントがある場合にしばしば使用されている定量分析方法です。分析結果に重大なエラー結果します。
標準的な追加は、マトリックスが均等にすべてのソリューションを影響が予想されますので、測定からマトリックスの影響を除去するために使用されます。また、それは化学物質を修正する使用されます相分離抽出プロセスで実行します。
不明なソリューションの実験 (この場合蛍光) 強度を読むし、追加既知の標準の変化量と未知の強度を測定することによって、メソッドが実行されます。データは蛍光強度対.追加標準量としてプロットされます (追加、基準なしの未知の世界自体は y 軸にプロットされる)。二乗ラインは、図 1に示すように、未知の濃度の負の x 軸を交差します。

図 1.標準添加法のグラフィック表現です。
手順
1. 試薬の準備
- 100 ppm 標準 Al3 +ソリューション: DI 水を 1 L メスフラスコにディゾルブ 0.9151 g アルミニウム硝酸塩 (Al (3)3•9H2O)。
- 8HQ ソリューション 1 M 酢酸 (2 %wt/巻): 8-ヒドロキシキノリンの 2.0 g を 100 mL のメスフラスコに追加。
- 慎重に 100 mL フラスコに 5.74 mL の氷酢酸を追加し、DI 水が付いている印に希釈します。これは液相に溶解する 8-オキシキノリンをことができます。
- 1 M NH4+/NH3バッファー (pH ~ 8): 酢酸アンモニウム (NH4OAc) 20 g を 100 mL のボトルに追加。
- この 100 mL ボトルに 30% アンモニア水 7 mL を追加し、DI 水のマークを希薄化します。これにより、8HQ ソリューションを組み合
結果
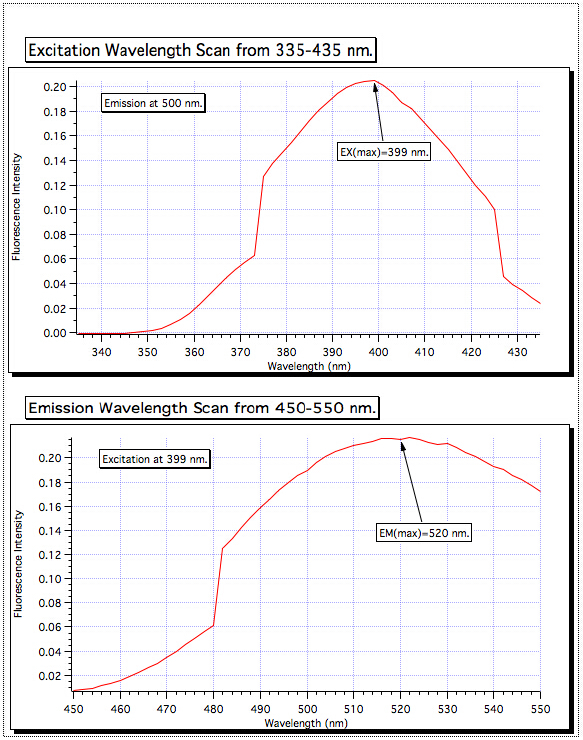
励起波長 335-435 からのスキャンは、399 で最高の吸収を示した nm、ので励起分光器がその値に設定されていました。450-550 nm からエミッション スキャンを行ったし、最も強い信号が 520 でことが判明した nm。これらは、すべてのサンプルに使用される波長です。