표준 추가 방법
Overview
출처: 폴 바우어 박사연구소 - 퍼듀 대학교
표준 첨가 방법은 양적 분석 방법이며, 관심 샘플에 매트릭스 효과를 초래하는 여러 구성 요소가 있을 때 자주 사용되며, 여기서 추가 구성 요소는 분석 흡수성 신호를 줄이거나 향상시킬 수 있습니다. 이로 인해 분석 결과에 심각한 오류가 발생합니다.
표준 추가는 일반적으로 매트릭스가 모든 솔루션에 동등하게 영향을 미친다고 가정하기 때문에 측정에서 매트릭스 효과를 제거하는 데 사용됩니다. 또한 추출 공정에서 수행되는 화학상 분리를 보정하는 데 사용됩니다.
이 방법은 알 수 없는 용액의 실험(이 경우 형광) 강도를 판독한 다음, 공지된 표준의 다양한 양으로 미지의 강도를 측정함으로써 수행된다. 데이터는 형광 강도대 추가된 표준의 양으로 플로팅됩니다(표준이 추가되지 않은 알 수 없음 자체는 y축에 플롯됩니다). 최소 사각형 선은 도 1에도시된 바와 같이 알 수 없는 농도의 음수로 x축을 교차합니다.

그림 1. 표준 추가 방법의 그래픽 표현.
Procedure
1. 시약 준비
- 100 ppm 표준 Al3+ 용액: 알루미늄 질산염(Al(NO3)3• 9H2O)를DI 물로 1L 체피 플라스크에 용해하십시오.
- 8HQ 용액은 1M 아세트산(2% wt/vol): 100mL 체피 플라스크에 8-하이드록시퀴놀린 2.0 g를 추가합니다.
- 조심스럽게 100 mL 플라스크에 5.74 mL 빙하 아세트 산을 추가 한 다음 DI 물로 마크에 희석하십시오. 이를 통해 8-하이드록시퀴놀린이 수단계에서 용해될 수 있습니다.
- 1 M NH4+/ NH3 버퍼 (pH ~8): 100 mL 병에 암모늄 아세테이트 (NH4OAc)의 20 g를 추가합니다.
- 이 100mL 병에 수산화 암모늄 의 7mL을 추가하고 DI 물로 마크에 희석하십시오. 이렇게 하면 결합될 때 8HQ 용액에서 산
Results
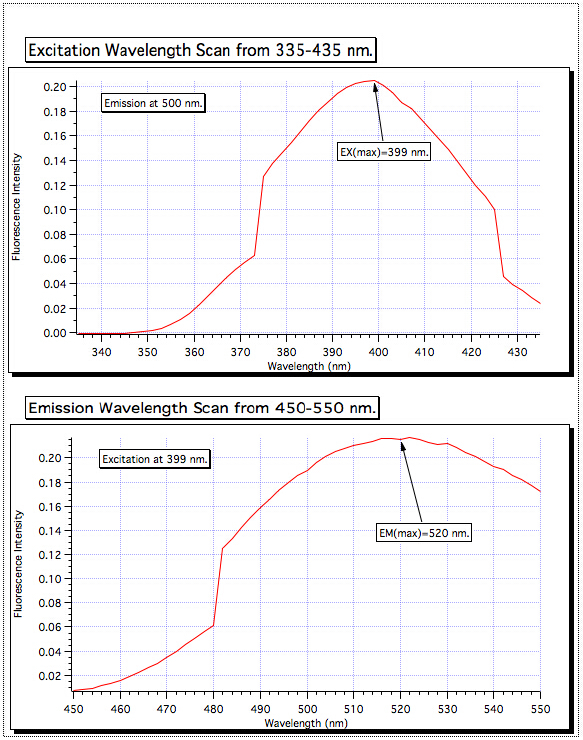
335-435에서 난회 파장의 스캔은 399 nm에서 가장 높은 흡수를 보였기 때문에 그 값에 대해 흥분 단색으로토르가 설정되었습니다. 이어서 방출 스캔은 450-550 nm에서 수행되었고, 가장 강한 신호는 520 nm에 있는 것으로 나타났다. 이들은 모든 샘플에 사용되는 파장입니다.