液相反応: しょ糖
概要
ソース: ケリー ・ m ・ ドゥーリーとマイケル g. ベントン、工業化学科、ルイジアナ州立大学、バトン ルージュ, ルイジアナ
バッチと連続流反応器は、触媒反応で使用されます。固体触媒および連続的な流れを使用して、パックされたベッドは、最も一般的な構成です。充填層のような原子炉は、"流れ"をプラグインとして広範なリサイクル ストリームがない場合は、通常モデル化します。他の最も一般的な連続炉、撹拌槽、完全に混合すると仮定されます。1充填層反応器の有病率の理由の一つは、ほとんどの攪拌槽の設計とは異なりリアクター容積の比率に大きい壁領域はより急速な熱伝達を促進すること、です。ほぼすべての原子炉の熱の追加または撤退場所を取る望ましい反作用の温度を制御する必要があります。
簡単な 1st注文以上に複雑、2nd順序、教科書など反応触媒反応の速度論が多い。反応速度は物質移動の率によっても影響されます - 反応はどの反応が表面または製品を削除する - レートに供給されると熱伝達率より速く起こることができません。これらの理由から、実験、大規模な設備を設計する前に反応速度を決定するために必要なほとんど。この実験では、このような実験を行う方法および反応速度式と速度定数を見つけることによってそれらを解釈する方法を探る。
この実験では、しょ糖の動態を決定する充填層反応器の使用について説明します。この反応は、液相反応物質と製品固体触媒によって特徴付けられるそれらの典型的です。
ショ糖 → グルコース (ブドウ糖) + fructose(1)
充填層反応器は、滞留時間に関連し、バッチ反応器経過時間に似ています、宇宙の時間を制御する流量で運営されます。固体酸触媒は、他のイオンの存在のためプロトンを交換することによって準備最初。原子炉は反応の流れを所望の温度 (等温操作) に加熱されます。温度が平衡、製品サンプリングが開始されます。サンプルは、旋光計、旋光を測定によって分析されます。混合物の旋光は反応ショ糖および明白な速度定数に関して、反応の順序を決定する使用して標準的な動態解析におけるショ糖の変換に関連付けることができます。-ない軸方向混合 (プラグ流) 対いくつか軸方向混合 (シリーズの撹拌槽) - 流体力学の動態に及ぼす影響をも分析します。
手順
触媒のプロパティ: サイズ = 20-40 メッシュ重量 = 223 g。含水率 = 30 wt %;。見掛け (かさ) 密度 = 1.01 g/ミリリットルです。酸部位濃度 = 4.6 モル酸サイト/g 乾燥重量;面積 50 m2/g; =macroporosity (猫の孔隙量/排出量) = 0.34;平均孔径 = 80 nm。ユニットの P & ID ダイアグラムを図 2に示します。この実験だけベッド #1 有機物タンク、ポンプ、浮子式流量計が使用されます。サンプルは上のドレインで収集されます。T505 は温度コント ローラーです。
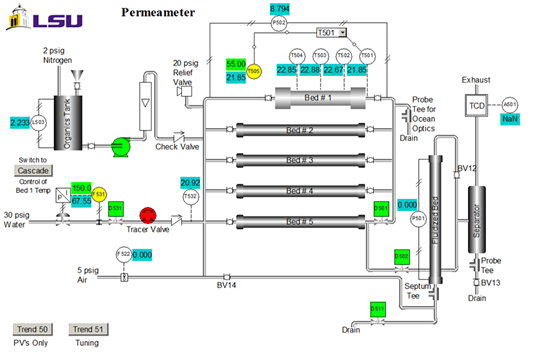
図 2.装置 (制御システム インターフェイス) の P & I
結果
申請書と概要
タグ
スキップ先...
このコレクションのビデオ:

Now Playing
液相反応: しょ糖
Chemical Engineering
9.7K 閲覧数

フィンドチューブ熱交換器の熱伝達効率のテスト
Chemical Engineering
18.0K 閲覧数

トレイを使用して対流および伝導性の熱伝達を調査するドライヤー
Chemical Engineering
44.0K 閲覧数

プロピレング リコール溶液の粘度
Chemical Engineering
33.2K 閲覧数

シリカ アルミナ粉末のポロシ メーター
Chemical Engineering
9.7K 閲覧数

押し出しによる力法律モデルのデモ
Chemical Engineering
10.3K 閲覧数

ガス吸収
Chemical Engineering
36.9K 閲覧数

気液平衡
Chemical Engineering
89.5K 閲覧数

トレイ蒸留効率に還流比の影響
Chemical Engineering
77.9K 閲覧数

液-液抽出の効率
Chemical Engineering
48.6K 閲覧数

化学修飾によるサリチル酸の結晶化
Chemical Engineering
24.3K 閲覧数

充填層反応器内の単相および流れ
Chemical Engineering
19.0K 閲覧数

ポリジメチルシロキサンを付加重合の動力学
Chemical Engineering
16.4K 閲覧数

エチレンの触媒反応: 水素化
Chemical Engineering
30.5K 閲覧数

スピンと寒さ
Chemical Engineering
7.4K 閲覧数
Copyright © 2023 MyJoVE Corporation. All rights reserved
