혈액 채취 II
Overview
출처: 케이 스튜어트, RVT, RLATG, CMAR; 발레리 A. 슈뢰더, RVT, RLATG. 노틀담 대학교, IN
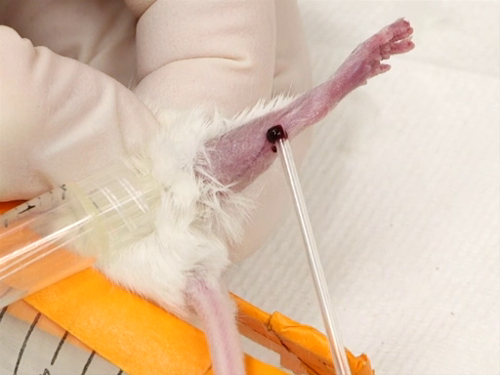
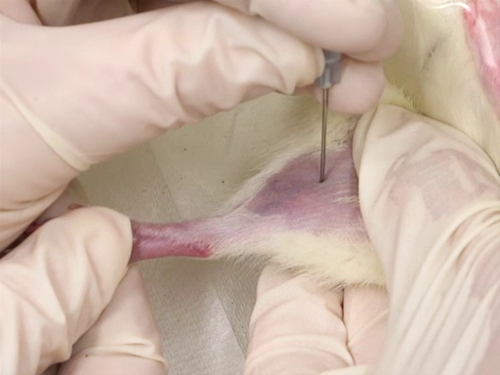
분석을 위해 마우스와 쥐로부터 혈액의 수집은 다양한 방법을 통해 수행 될 수있다. 수집의 각 방법은 필요한 구속의 유형, 절차의 침습성 및 일반 마취의 필요성에 변화가 있습니다. 1 역사적으로, 복고풍 궤도 부비동 구멍의 사용은 사용되었습니다, 하지만 논쟁없이. 잠재적 인 조직 손상, 또는 심지어 실명과 관련된 논쟁은, 복고풍 궤도 출혈에 의해 발생, 마우스에서 얼굴과 하복부 정맥 출혈 방법의 개발로 이어졌다. 쥐와 쥐 모두에서 수페누스 정맥에서 혈액 수집은 개발 된 또 다른 기술이다. 이러한 절차는 마취를 필요로하지 않으며 따라서 마취제의 사용이 혈액 결과 또는 기타 데이터를 혼동 할 수있는 경우에 적합합니다.
Procedure
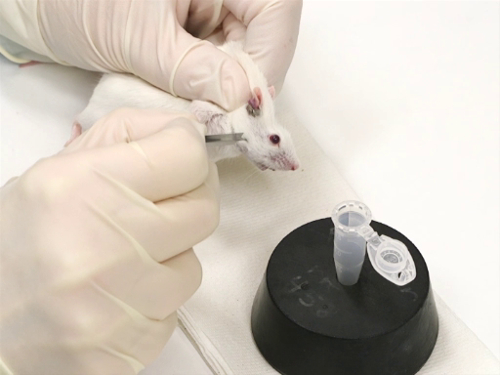
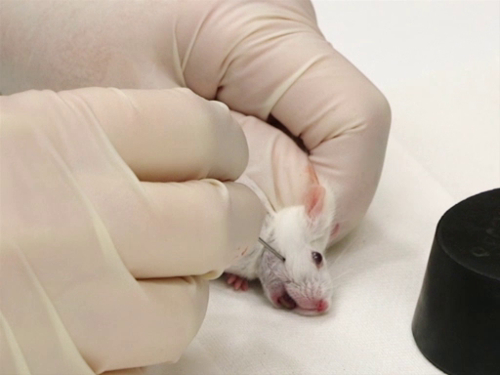
1. 마우스에서 출혈 얼굴 정맥
- 설비
- 혈액의 수집은 혈액 튜브 또는 엡펜도르프 튜브에 무료 캐치에서입니다. 어떤 경우에는 혈액을 헤마토크튜브로 직접 수집하는 것이 바람직합니다.
- 골든로드 랜싯은 나이와 성별에 따라 동물에게 적합한 크기에 따라 선택됩니다.
- 랜싯은 마우스의 나이/크기에 따라 다음과 같이 선택됩니다.
4mm 랜싯: 3-4주 된 마우스 (15 그램 체중 미만)
5mm 랜싯: 10주 미만의 여성 마우스(체중 ~20그램 미만)
5mm 랜싯: 6주 미만의 남성 마우스(~20그램 체중 미만)
5mm 랜싯: 단일 드롭 샘플용(혈액 얼룩용)
5.5mm 랜싯: 10주 이상 여성 마우스(체중 ~20그램 이상)
5.5mm 랜싯: 6주 이상 남성 마우스(체중 ~20그램 이상)
5.5mm 랜싯: 대형 샘플
- 랜싯은 마우스의 나이/크기에 따라 다음과 같이 선택됩니다.
- 구속
- 마우스는 스크러핑 기술을 사용하여 제지됩니다.<
Application and Summary
혈액 수집 방법은 시료의 분석에 변화를 일으킬 수 있습니다. 샘플 수집을 수행하는 기술자의 기술 수준은 샘플의 품질과 동물의 복지에 영향을 미칩니다. 마취제의 사용은 샘플 품질에도 영향을 줄 수 있습니다. 여기에 설명된 방법은 모두 마취를 사용하지 않고 수행되므로 변수가 제거되었습니다. 또한 이러한 모든 기술은 동물에 대한 최소한의 불편함으로 시리얼 샘플링에 사용할 수 있습니다...
References
- Fernandez, I., Arantza, P., Del Teso, N., Perez, V. and Rodriguez-Cuesta, J. 2010. Clinical biochemistry parameters in C57BL/6J mice after blood collection from the submandibular vein and retroorbital plexus. Journal of American Association of Laboratory Animal Science. 49:2. 202-210.
- Joslin, O.T. 2009. Blood collection techniques in exotic small animals. Journal of Exotic Pet Medicine. 18:2. 117-139.
- Hem, A., Smith, A.J. and Solberg, P. 1998. Saphenous vein puncture for blood sampling of the mouse, rat, hamster, gerbil, guinea pig, ferret and mink. Laboratory Animals. 32. 364-368.
- Diehl, K-H., Hull, R., Morton, D., Pfister, R. Rabemampianina, Y., Smith, D., Vidal, J-M., and van de Vorstenbosch, C. 2001. A good practical guide to the administration of substances and removal of blood, including routes and volumes. Journal of Applied Toxicology. 21. 15-23.
Tags
건너뛰기...
이 컬렉션의 비디오:

Now Playing
혈액 채취 II
Lab Animal Research
73.7K Views

설치류 취급 및 통제 기술
Lab Animal Research
175.4K Views

실험실 기본 관리
Lab Animal Research
28.1K Views

사육 및 이유의 기초
Lab Animal Research
35.8K Views

설치류 식별 I
Lab Animal Research
55.0K Views

설치류 식별 II
Lab Animal Research
25.7K Views

화합물 투여 I
Lab Animal Research
101.1K Views

화합물 투여 II
Lab Animal Research
35.1K Views

화합물 투여 III
Lab Animal Research
31.6K Views

화합물 투여 IV
Lab Animal Research
52.0K Views

혈액 채취 I
Lab Animal Research
172.4K Views

마취 유도 및 유지
Lab Animal Research
50.9K Views

설치류 수술에 대한 고려 사항
Lab Animal Research
22.5K Views

진단 부검 및 조직 수확
Lab Animal Research
58.3K Views

멸균 조직 수확
Lab Animal Research
34.9K Views
Copyright © 2025 MyJoVE Corporation. 판권 소유