Method Article
Implantation of the Syncardia Total Artificial Heart
In This Article
Summary
The purpose of this manuscript is to briefly review indications, management, and outcomes for the total artificial heart. Video operative techniques for device implantation are presented.
Abstract
With advances in technology, the use of mechanical circulatory support devices for end stage heart failure has rapidly increased. The vast majority of such patients are generally well served by left ventricular assist devices (LVADs). However, a subset of patients with late stage biventricular failure or other significant anatomic lesions are not adequately treated by isolated left ventricular mechanical support. Examples of concomitant cardiac pathology that may be better treated by resection and TAH replacement includes: post infarction ventricular septal defect, aortic root aneurysm / dissection, cardiac allograft failure, massive ventricular thrombus, refractory malignant arrhythmias (independent of filling pressures), hypertrophic / restrictive cardiomyopathy, and complex congenital heart disease. Patients often present with cardiogenic shock and multi system organ dysfunction. Excision of both ventricles and orthotopic replacement with a total artificial heart (TAH) is an effective, albeit extreme, therapy for rapid restoration of blood flow and resuscitation. Perioperative management is focused on end organ resuscitation and physical rehabilitation. In addition to the usual concerns of infection, bleeding, and thromboembolism common to all mechanically supported patients, TAH patients face unique risks with regard to renal failure and anemia. Supplementation of the abrupt decrease in brain natriuretic peptide following ventriculectomy appears to have protective renal effects. Anemia following TAH implantation can be profound and persistent. Nonetheless, the anemia is generally well tolerated and transfusion are limited to avoid HLA sensitization. Until recently, TAH patients were confined as inpatients tethered to a 500 lb pneumatic console driver. Recent introduction of a backpack sized portable driver (currently under clinical trial) has enabled patients to be discharged home and even return to work. Despite the profound presentation of these sick patients, there is a 79-87% success in bridge to transplantation.
Introduction
The first human implantation of a total artificial heart (TAH) in 1969 was performed by Denton Cooley in a 47 year old male who was unable to wean from cardiopulmonary bypass following left ventricular aneurysm repair. The experimental device provided hemodynamic support for 64 hr until a donor heart could be found. Although technically successful, device support was complicated by hemolysis and renal failure. The patient would go on to die from overwhelming sepsis 32 hr after transplantation1. A second attempt by Cooley in 1981 had similar unfortunate results2. In 1982, William DeVries performed the widely publicized permanent first implant of the Jarvik-7 TAH into a 61 year old dentist. The patient had a difficult postoperative course marked by recurrent respiratory failure, fracture of the prosthetic mitral valve strut requiring replacement of device, sepsis, stroke, intermittent renal failure, and bleeding related to anticoagulation. He ultimately succumbed after 112 days to pseudomembranous colitis3.
Despite these initial discouraging results, progressive refinement in device design, patient selection, and patient management have led to marked improvement in outcomes. The Jarvik-7 has since evolved into the Syncardia TAH, which remains the only FDA approved TAH in clinical use today. To date, over 1,000 implants have been performed worldwide4.
We present our institutional operative techniques in the context of a video case report.
Protocol
Case Presentation:
The patient is a 60 year old male with hypertrophic cardiomyopathy. He has developed progressive heart failure symptoms over the past 10 years with recent multiple readmissions for decompensated heart failure. He was started on chronic IV milrinone and was listed for heart transplantation. His PMH is notable for atrial arrhythmias s/p ablation, recurrent pulmonary embolism, pulmonary hypertension, left atrial thrombus, and prior stroke. Cardiopulmonary exercise testing demonstrated a low maximal oxygen consumption ( VO2 max 10.6 ml/kg min). Imaging was notable for severe LV hypertrophy with moderate to severely reduced systolic function, and no LV outflow tract obstruction (LVEF 25-30%, LVIDd 5.0 cm, IVSd 1.5 cm, LVPWd 1.5 cm). He had a large left atrial thrombus in the appendage and a possible LV thrombus as well. Hemodynamics (on milrinone 0.5 mcg/kg/min) were notable for: RA 17 mmHg, PCWP 37 mmHg, PA 74/35 mmHg, and Fick CI 1.4 L/min/m2. Due to his restrictive myopathy and small LV cavity, he was considered to be a poor candidate for isolated left ventricular assist device.
1. The Device
- The Syncardia total artificial heart is the only FDA approved TAH in clinical use. It was first introduced in 1982 as the Jarvik-7 heart.
- The device consists of 2 polyurethane ventricle chambers. Each chamber has a maximum stroke volume of ~ 70 ml. Each chamber has 2 tilting disc valves (Medtronic Hall, 27 mm inflow, 25 mm outflow) to direct blood inflow and outflow. Each chamber is pneumatically driven by a separate driveline connected to a driver externally (Figure 1).
2. Indications
- The device displaces a total volume of ~400 ml. General minimum recipient size recommendation include a BSA > 1.7 m2 and a thoracic diameter (AP dimension spine to sternum at the level of the 10th thoracic vertebra) > 10 cm (Figure 2). Placement in smaller patients is possible by displacing the device into the left chest although this increases the risk for left sided pulmonary vein and bronchus compression. A smaller sized device for use in smaller patients is being developed and is expected in the near future.
- TAH support is indicated in patients with late stage severe biventricular failure or other anatomic abnormality that is not optimally treated with isolated left ventricular mechanical support. Such patients often present in extremis with complications related to their malperfusion. Typically patients are classified as INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support) profile 1 (critical cardiogenic shock) or 2 (progressive decline on inotropic support).
- Patients with significant concomitant cardiac pathology. Critically ill patients with end stage heart failure requiring mechanical support may not be optimally treated by the addition of other extensive reparative procedures. This may increase the risk for right ventricular failure post LVAD implantation, which increases the risk for perioperative morbidity and mortality. This includes: acute myocardial infarction with ventricular septal defect; aortic root aneurysm / dissection, cardiac allograft failure, massive ventricular thrombus, refractory malignant arrhythmias (independent of filling pressures), hypertrophic / restrictive cardiomyopathy, and complex congenital heart disease.
3. Preparation of the Device
- The Dacron aortic and pulmonary grafts are sealed with CoSeal Surgical Sealant (Figure 3).
- The grafts and devices are soaked in rifampin.
4. Implantation
- Routine preparation and median sternotomy. The chest and abdomen are prepped in the routine fashion for cardiac surgery and a median sternotomy is performed.
- Division of the left diaphragm and creation of a left upper abdominal preperitoneal pocket. Similar to the implant of an LVAD, the left diaphragm is divided medially and a left upper abdominal preperitoneal pocket is created for the pneumatic drivelines. Two incisions ~5-10 cm below the left costal margin are made and carried through the rectus fascia. Intramuscular tunnels for the pneumatic drivelines are created and maintained with two 1" Penrose drains.
- Cannulation and initiation of cardiopulmonary bypass. The aorta, SVC, and IVC are cannulated. Bicaval cannulation is performed through the atria (vs direct cannulation of the SVC/IVC) to preserve the sites for use during subsequent transplantation. Unnecessary dissection within the pericardium is minimized to preserve tissue planes.
- The aorta is cross clamped. The aortic root and pulmonary artery are divided at the level of the valvar commissures. Excision of the right ventricle is started along the acute margin, 1-2 cm distal to and parallel to the right atrioventricular groove. The interventricular septum is incised opening the left ventricle. The right ventricular incision is continued superiorly into the RV outflow tract. Similarly, the left ventricle incision is extended laterally parallel to the left atrioventricular groove. The LV outflow tract is opened laterally. The remaining interventricular septum is divided and specimen is removed (Figure 4).
- The coronary sinus is oversewn. If a PFO is identified it is similarly oversewn. The left atrial appendage is ligated with a non cutting endoscopic stapler to eliminate a potential source of thrombus (Figures 5 and 6).
- The mitral and tricuspid valve leaflets are excised leaving a several millimeter cuff to the annulus. The ventricular cuffs are trimmed leaving a 1 cm rim of tissue. The cuffs are then oversewn with 2-0 Prolene (MH) for hemostasis and to downsize the orifice to the size of the TAH atrial quick connects. Some centers elect to use a reinforcing strip of felt.
- The cuffs of the TAH atrial quick connects are trimmed to 0.5-1 cm. The cuffs are then inverted and sewn to their respective left and right ventricular cuffs.
- In a similar fashion, the aortic and pulmonary artery graft quick connects are trimmed. The pulmonic graft is left several centimeters longer than the aortic graft to allow room for the aortic graft to pass below. The grafts are sewn to their respective orifices (Figure 7).
- At the time of subsequent re-entry for transplantation, an intense inflammatory response of the pericardium is often seen. Dissection through this tissue at the time of transplantation can be associated with significant blood loss. This appears to be greater than what is typically seen during redo sternotomy. Preclude pericardial membrane is used to line the pericardium and maintain avascular tissue planes. This is started along the left inferolateral pericardium as access to this space will be hindered once the device is in place. Strips of Preclude are also placed around the aortic and pulmonic anastomoses. Some centers use sterilized rubber tourniquets to wrap the anastomoses.
- The drivelines are passed through the previously created tunnels. A 13-15 mm Hegar dilator is used to guide the driveline through the previously placed Penrose drain. A sheet of Preclude membrane is wrapped around the drivelines as well. The original metal connectors of the external console driveline are replaced with plastic connectors that will also connect with the portable discharge driver.
- De-airing nipples on the TAH left and right ventricles are ligated. The left and then right artificial ventricles are flushed and then connected to their respective orifices. The pliable quick connects are grasped with 2 heavy needle drivers and stretched over the hard plastic connectors of the artificial ventricles ensuring a tight connection (Figure 8).
- An aortic root vent is placed. Low rate, low pressure pumping is initiated (Left drive pressure 40, right drive pressure 0, % systole 40, rate 40, vacuum 0) and the lungs are ventilated for de-airing. Flow through the right side is initially passive.
- The cross clamp is removed. Once adequate de-airing is confirmed by TEE, cardiopulmonary bypass can be discontinued relatively quickly. TAH support is increased. Typical initial immediate TAH parameters are: Left drive pressure 180-200 mmHg, right drive pressure 30-60 mmHg, HR 100-120 bpm, and % systole 50. Vacuum is generally left at 0 until the chest is closed (or sealed) to prevent entrainment of air. Once sealed, increases in vacuum (generally to 15 mmHg) will result in increased fill volume and increased cardiac output. Parameters are then titrated for partial ventricular filling and complete ejection.
- Protamine is given and the CPB cannulas are removed. Patients requiring a TAH often present with significant end organ dysfunction including chronic hepatic congestion and may have significant associated underlying coagulopathy. The most common complication following TAH implantation is bleeding. We recommend a low threshold for mediastinal packing and delayed sternal closure.
- Once the chest is ready to be closed, the remainder of the pericardium along the right and superiorly is lined with Preclude. The configuration of the TAH does not maintain the normal oblong shape. Contraction of the pericardium about the device will limit the space available for subsequent transplantation. A saline breast implant is used to maintain the apical pericardial space for subsequent transplant. A smooth saline breast implant is placed at the apex and filled with 200-250 ml of saline (Figure 9).
- The chest is closed in the routine fashion. Although imgagin by intraoperative TEE is limited, it remains a useful tool at the end of surgery to assess for compression of the right and left atrial venous return by the device during closure. This is more common on the right (both the cava and right sided pulmonary veins) and can generally be treated with displacement of the device inferiorly and to the left. This can be secured with a heavy suture placed around the costal margin and tied to the artificial right ventricle. A strip of silastic is left beneath the sternum to protect the pump during re-entry for transplantation.
Results
From April 2006 through July 2012, 66 patients were implanted with a TAH at Virginia Commonwealth University Medical Center. Patients were critically ill: 18% were on other mechanical support (ECMO, LVAD, or BiVAD), 58% were on an intra-aortic balloon pump, 58% were on inotropic medications, 17% were mechanically ventilated, and 17% were on hemodialysis. Patients were supported for a total of 7863 days. Median duration of support was 87.5 days (range 1 to 602 days). 10 patients were discharged home on a portable discharge driver (as part of a clinical trial, not FDA approved). 50 (76%) patients were successfully bridged to transplantation, 7 (11%) remained on the device awaiting transplantation, and 9 (14%) died while on the device (Figure 10). 3 of the deaths occurred in the 1st week following implantation and were related to progressive multisystem organ failure. The other 6 deaths occurred from 32 to 169 days post implant (3 sepsis / MSOF, 1 mediastinal bleeding, 1 intracranial hemorrhage, and 1 hypertensive crisis). The most frequent perioperative adverse event was bleeding requiring mediastinal re-exploration in 30%.
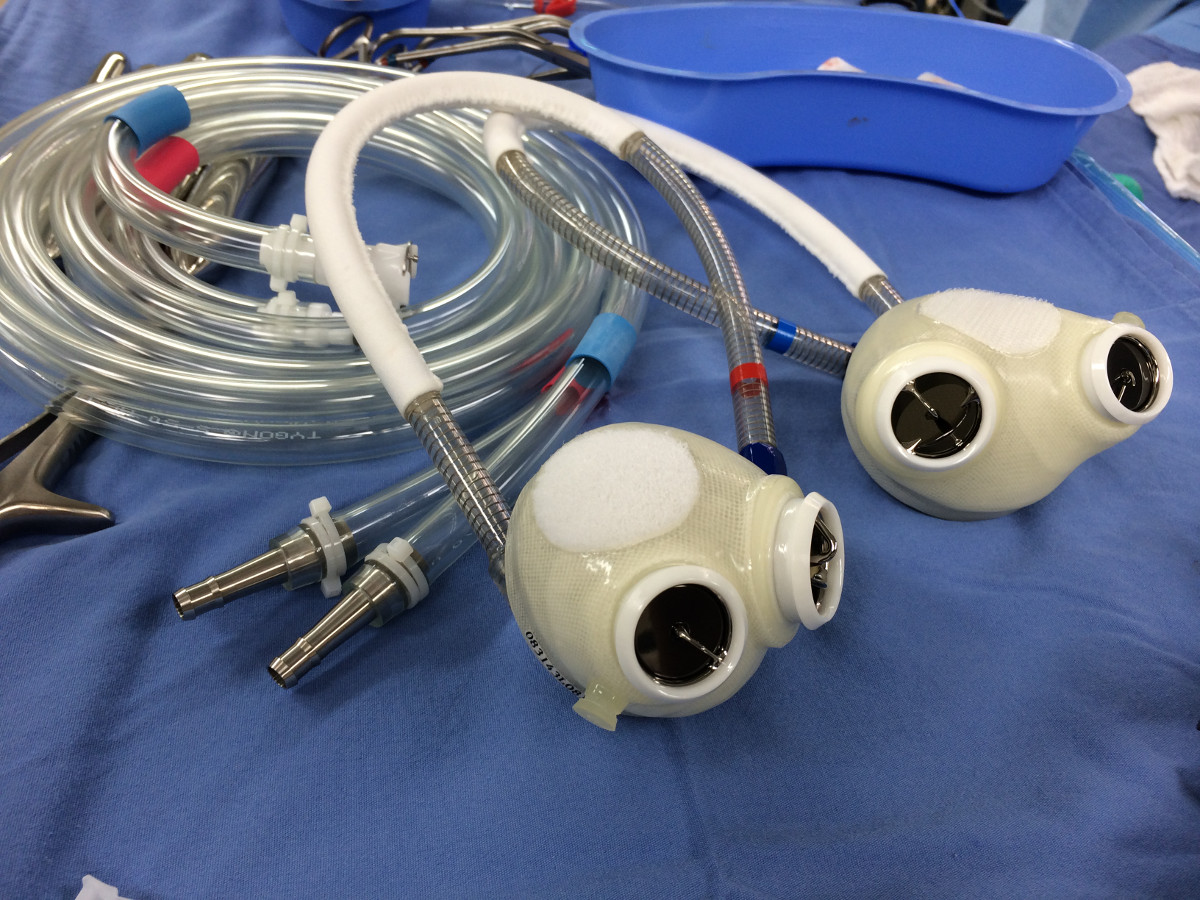
Figure 1. The Syncardia total artificial heart.
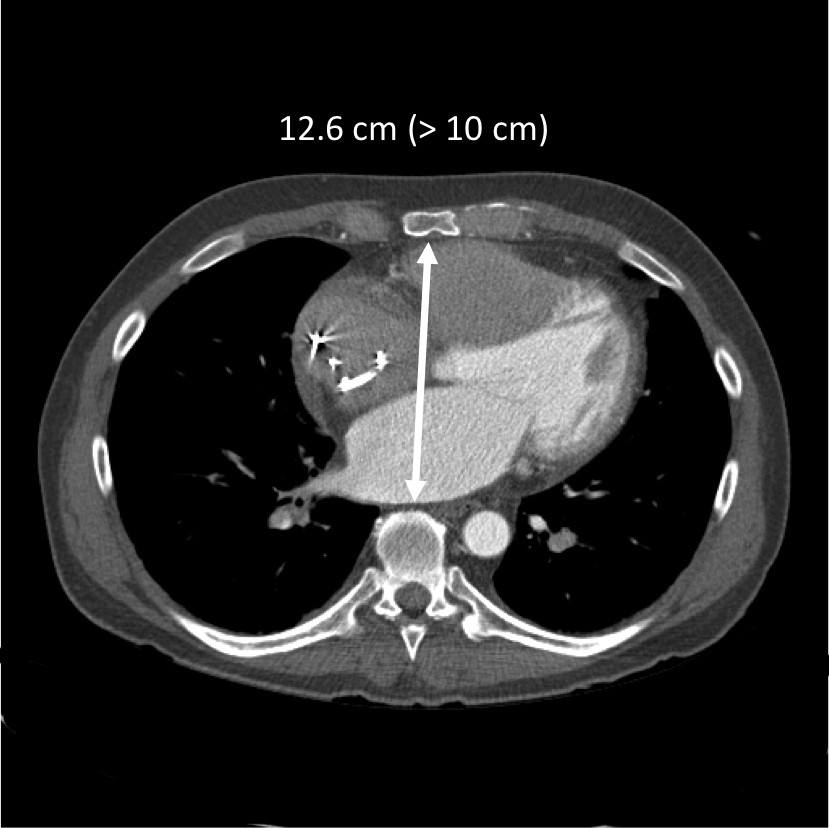
Figure 2. Measuring AP dimension by CT. A space > 10 cm between the sternum and the anterior border of the 10th vertebral body is generally required for the device to fit.
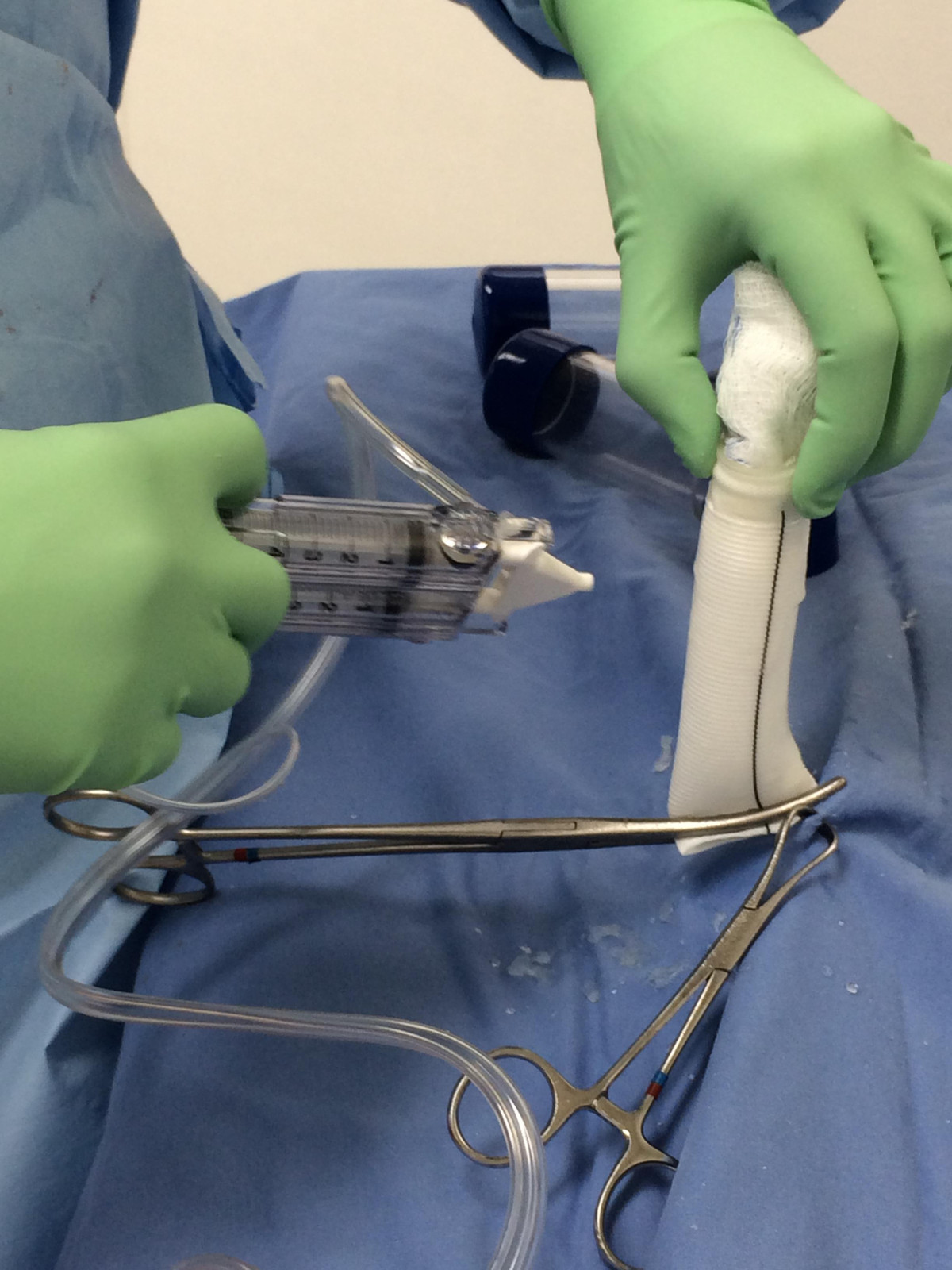
Figure 3. Pretreating the arterial outflow grafts. The aortic and pulmonary artery grafts are presealed with Coseal Surgical Sealant, a synthetic hydrogel used as an adjunctive sealant for vascular grafts. Previously the grafts were preclotted with patient's own blood prior to heparinization.
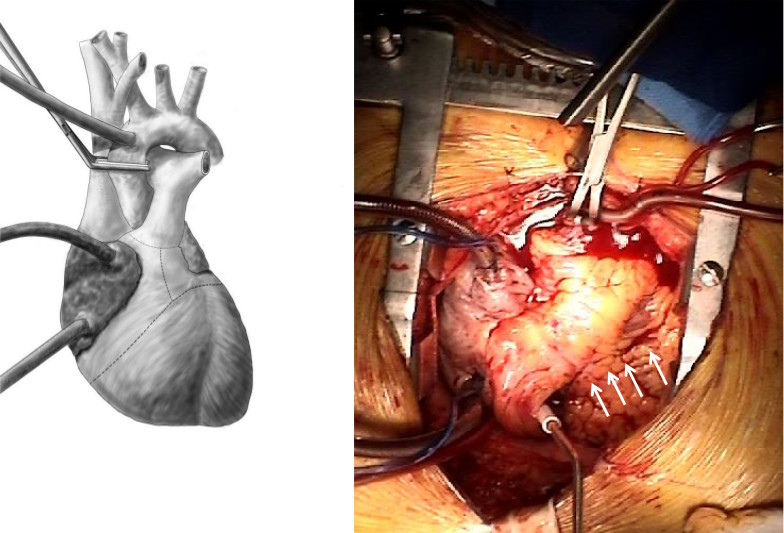
Figure 4. Excision of the right and left ventricle. The RV and LV are excised leaving a 1 cm ventricular cuff beyond the mitral and tricuspid annulus. Arrows point to the incision along the anterior wall of the RV. The incisions are extended through the left and right ventricular outflow tracts and through the aortic and pulmonic valves.
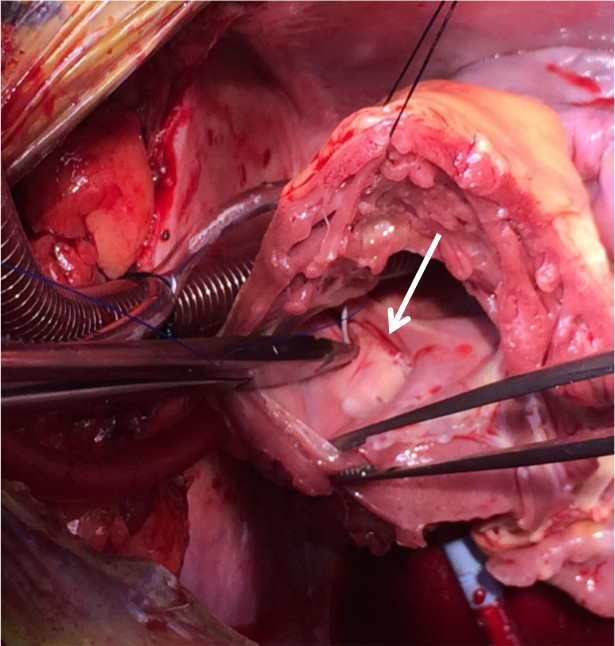
Figure 5. Oversewing the coronary sinus. The coronary sinus (arrow) is oversewn through the RV cuff (the tricuspid leaflets have been excised) for hemostasis.
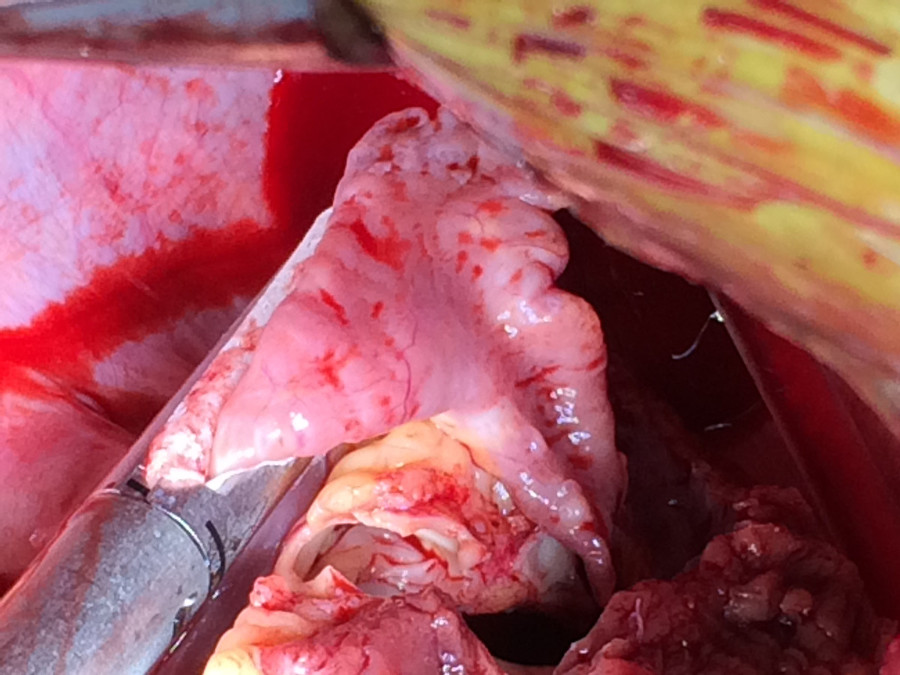
Figure 6. Ligating atrial appendage. The left atrial appendage is ligated with a Seamguard-reinforced endo GIA stapler to minimize a potential source for systemic emboli.
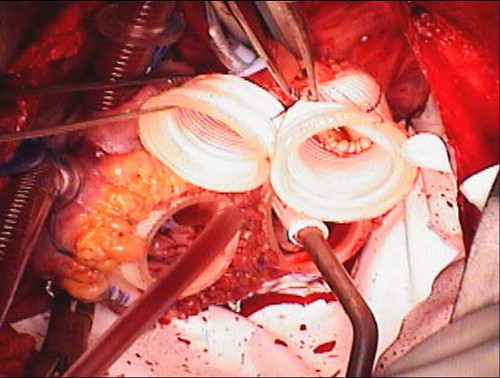
Figure 7. Quick connects and Goretex Preclude implanted. The atrial quick connects and vascular grafts are sewn to their respective orifices. The pericardium is lined with Goretex membrane to facilitate subsequent reentry for transplantation.
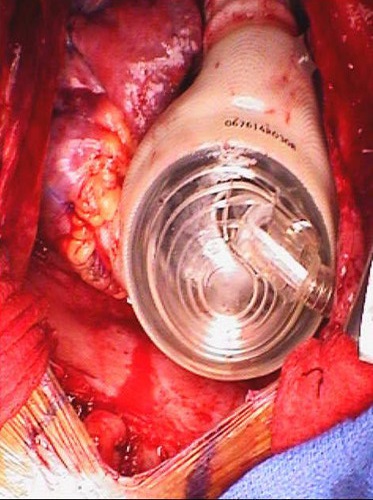
Figure 8. Device implanted. The device implanted just prior to chest closure.
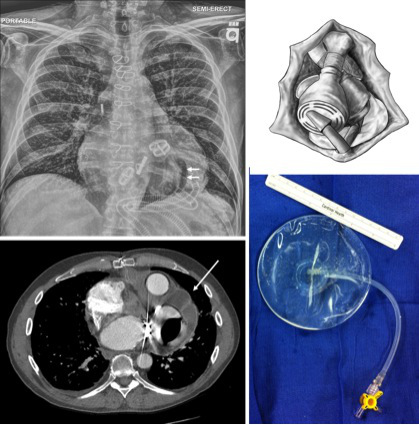
Figure 9. Pericardial apical saline implant as demonstrated on CT imaging and CXR. A saline implant (single arrows) is used to maintain the pericardial apical space for transplantation. In the CXR, the edge of the TAH (where the pericardium would otherwise contract) is delineated by the central air bubble (double arrows).
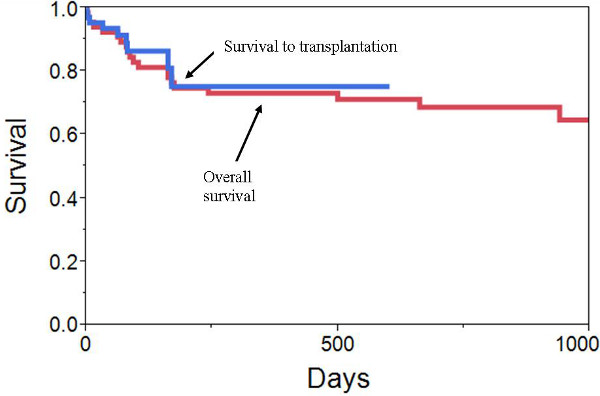
Figure 10. VCU Outcomes. Kaplan-Meier survival curves demonstrating survival to transplantation and overall survival following TAH implant.
Discussion
Artificial replacement of the human heart has long captured the public's imagination. Early experience with the total artificial heart was marked by suboptimal outcomes1-3. The design and implantation techniques for the TAH used today have not changed significantly since Dr. DeVries' original description6. However, refinements in patient selection (as a bridge to transplant) and understanding of perioperative management have led to significant improvement in outcomes. In 2004, a landmark study of 81 TAH implants was published. The trial established the efficacy of the TAH as a bridge to transplantation and led to FDA approval of the Syncardia TAH. In this nonrandomized study, 79% were successfully bridged to transplantation. Overall survival at 1 year was 70%. In a matched cohort of 35 patients who met study criteria but did not undergo TAH implantation, 46% survived to transplantation and 1 year survival was 31%7.
Patient selection is critical to good outcomes8,9. It is important to identify patients that have not yet developed irreversible end organ failure or other complications which would limit their likelihood for resuscitation or transplant candidacy. At our institution, findings of cirrhosis by liver biopsy, chronic dialysis dependency, or other psychosocial factors that would preclude transplant candidacy would also preclude candidacy for a TAH. Conversely, patients with acute decompensation or other evidence for potential end organ recovery are considered.
It is equally important to identify patients with biventricular failure whose right ventricular (RV) dysfunction will improve with isolated LV unloading and thus do not need biventricular circulatory support. RV failure following LVAD implantation is associated with increased morbidity and mortality10. Early use of biventricular support compared to delayed rescue for RV failure following LVAD is also associated with improved outcomes11. A number of risk factors for RV failure have been identified and several risk scoring systems have been developed. The need for inotropic / intra-aortic balloon pump support, evidence of renal and hepatic dysfunction (elevated creatinine, aspartate aminotransferase, bilirubin), hemodynamic evidence of RV dysfunction (decreased RV stroke work index, increased right atrial / wedge pressures), and echocardiographic evidence of RV dysfunction (RV dilatation, decreased RV ejection fraction / tricuspid annular motion, increased tricuspid regurgitation) have all been identified as risk factors12-15. Nonetheless, determination of risk for RV failure after LVAD placement remains difficult. One recent small series demonstrated no predictive value from several of the scoring systems in predicting the need for RV support16.
Timing of surgery is an important consideration. Once the decision has been made that biventricular mechanical support is necessary, early implantation offers the most effective way to restore blood flow and resuscitate a patient. However, patients can present abruptly with profound acute cardiogenic shock, severe malperfusion, and minimal prior evaluation. Liberal use of temporary support options (such as ECMO / IABP) for 24-48 hrs can start the process of resuscitation while questions regarding neurologic status or other questions of transplant candidacy are being answered. Prolonged use of high dose inotropic support may increase the risk for irreversible end organ failure or other complications.
The general techniques of implantation have not changed dramatically since the device was first introduced6,17. However, the benefits of taking the time to protect and maintain the pericardial space should be emphasized18. The device appears to incite an intense inflammatory thickening of the pericardium. Lining the pericardium with Goretex and maintaining the apical space with a saline implant greatly facilitates the re-entry for transplantation. Perioperative bleeding remains the most frequent perioperative complication. Packing the chest with delayed sternal closure is an effective strategy to limit the amount of perioperative blood products needed to reverse underlying coagulopathy and minimize the risk for tamponade. Despite the relatively rigid shell of the artificial ventricles it is possible for enough mediastinal fluid to accumulate and impede venous inflow causing tamponade. Surface echocardiography following TAH implantation has limited utility. CT imaging if often limited by underlying renal dysfunction and need to avoid IV contrast. We recommend early mediastinal re-exploration in any TAH patient who is otherwise doing poorly for unknown reasons.
Following implantation, the device is adjusted to maximize cardiac output. It is capable of generating an output > 9 lpm. To minimize stasis, device parameters are adjusted for "partial filling and complete eject." Typical early postoperative TAH parameters are: Left drive pressure 180-200 mmHg, right drive pressure 30-60 mmHg, HR 100-120 bpm, % systole 50, and vacuum 15 mmHg. In patients with long standing pulmonary hypertension, the higher RV drive pressures required for full right sided ejection may be detrimental. It is possible to "overdrive" the right sided output. In 2 patients, this resulted in profound pulmonary edema requiring temporary venovenous ECMO support. One patient had progressive multisystem organ failure and expired. The other was supported until the edema subsided and was successfully weaned off of ECMO.
Anticoagulation is generally initiated 24 hrs after chest closure. Patients are started on bivalirudin (0.005 mg/kg/h), aspirin (81 mg daily), and dipyridamole (50 mg tid). Outside of the operating room, heparin is avoided to minimize the risk of heparin induced thrombocytopenia. Bivalirudin is generally not titrated and once stable transitioned to oral warfarin. Goals of therapy are an INR 2-3 and platelet function 20-40% normal by optical aggregometry. Patients with evidence of increased hemolysis (LDH > 1000) may benefit from the addition of pentoxifylline (400 mg TID)19,20.
Renal failure in acutely ill patients is clearly multifactorial. However, we have noticed a disproportionate tendency towards renal failure following TAH implantation. We hypothesize that this is in part related to the abrupt decrease in native natriuretic peptide production associated with ventricular excision. Perioperative supplementation with a low dose nesiritide infusion (0.005 mcg/kg/min) appears to reduce the incidence of renal failure21. Furthermore, infusion of nesiritide has a profound effect for increasing urine output after TAH implantation22. Once patients have recovered we have been able to discontinue the infusion in most patients. However, we have had a few patients who could not be weaned from the infusion until they were transplanted.
TAH patients often demonstrate significant chronic anemia. Causes include low grade hemolysis and ineffective erthryopoesis. Despite the anemia, TAH patients demonstrate good exertional tolerance and minimal symptoms even with hemoglobin concentrations of 5-6 g/dL. In order to avoid HLA sensitization and other associated complications, transfusions are avoided unless the patient is symptomatic or there are other sides of end organ malperfusion23.
Beyond the early postoperative period, care is focused on aggressive physical rehabilitation. Despite the severe presentation, the majority of patients were able to initiate physical therapy in the first postoperative week and most were able to start treadmill exercise by the second week. However, we have found that TAH patients demonstrate an abnormal blunting of the blood pressure response to exercise. This is in part related to the use of vasodilators to limit afterload24. While this could limit the amount of physical recovery, most patients go on to transplant before this is reached.
Until recently, TAH patients were hospital bound and tethered to a 418 lb console. As waiting times for donor organs continue to increase, this was accompanied by significantly reduced quality of life as well as increased financial costs. The introduction of a portable driver allowing discharge to home and even returning to work has been a major advance in the practical utility of the TAH. The Syncardia Freedom Driver (under clinical trial and not FDA approved) is a 14 lb, backpack sized driver with an electrically driven pneumatic piston25. Early experience with the driver has demonstrated that it is sensitive to afterload conditions and that an aggressive anti hypertensive drug regimen is necessary26.
In summary, current results have established the TAH as an effective device for resuscitation and subsequent bridge to transplantation. This cohort of patients represents an extreme end of the spectrum of end stage heart failure patients. Often there is no other suitable durable therapeutic option.
Disclosures
Production of this video-article was sponsored by SynCardia. MH and VK have served as consultants for Syncardia Systems Inc. The other authors have nothing to disclose.
Materials
| Name | Company | Catalog Number | Comments |
| Total artificial heart (implant kit) |  Syncardia Syncardia | 500101 | |
| PRECLUDE pericardial membrane |  Gore Medical Gore Medical | 1PCM101 | |
| Smooth Round Saline Implant |  Mentor Mentor | 350-1645 | |
| CoSeal Surgical Sealant |  Baxter Baxter | 93407 |
References
- Cooley, D. A., Liotta, D., Hallman, G. L., Bloodwell, R. D., Leachman, R. D., Milam, J. D. Orthotopic cardiac prosthesis for two-staged cardiac replacement. American Journal of Cardiology. 24 (5), 723-730 (1969).
- Cooley, D. A. The total artificial heart. Nat. Med. 9 (1), 108-111 (2003).
- DeVries, W. C., Anderson, J. L., Joyce, L. D., Anderson, F. L., Hammond, E. H., Jarvik, R. K., Kolff, W. J. . Clinical use of the total artificial. 310 (5), 273-278 (1984).
- . Syncardia Systems. 1000th Implant of the World's Only Approved Total Artificial Heart Performed. [Press release]. , (2012).
- Joyce, L. D., et al. Nine year experience with the clinical use of total artificial hearts as cardiac support devices. ASAIO Transactions. 34, 703-707 (1988).
- DeVries, W. C. Surgical Technique for Implantation of the Jarvik-7-100. Total Artificial Heart. JAMA. 259 (6), 875-880 (1988).
- Copeland, J. G., et al. Cardiac replacement with a total artificial heart as a bridge to transplantation. N. Engl. J. Med. 351, 859-867 (2004).
- Kasirajan, V., Tang, D. G., Katlaps, G. J., Shah, K. B. The total artificial heart for biventricular failure and beyond. Curr Opin Cardiol. 27, (2012).
- Copeland, J. G., et al. Risk factor analysis for bridge to transplantation with the CardioWest total artificial heart. Ann Thorac Surg. 85, 1639-1644 (2008).
- Kormos, R. L., et al. Right ventricular failure in patients with the HeartMate II continuous-flow left ventricular assist device: incidence, risk factors, and effect on outcomes. J Thorac Cardiovasc Surg. 139, 1316-1324 (2010).
- Fitzpatrick, J. R., et al. Early planned institution of biventricular mechanical circulatory support results in improved outcomes compared with delayed conversion of a left ventricular assist device to a biventricular assist device. J Thorac Cardiovasc Surg. 137, 971-977 (2009).
- Matthews, J. C., Koelling, T. M., Pagani, F. D., Aaronson, K. D. The right ventricular failure risk score a pre-operative tool for assessing the risk of right ventricular failure in left ventricular assist device candidates. J Am Coll Cardiol. 51, 2163-2172 (2008).
- Ochiai, Y., et al. Predictors of Severe Right Ventricular Failure After Implantable Left Ventricular Assist Device Insertion: Analysis of 245 Patients. Circulation. 106 (suppl I), (2002).
- Drakos, S. G., et al. Risk factors predictive of right ventricular failure after left ventricular assist device implantation. Am J Cardiol. 105, 1030-1035 (2010).
- Fitzpatrick, J. R., et al. Risk score derived from pre-operative data analysis predicts the need for biventricular mechanical circulatory support. J Heart Lung Transplant. 27, 1286-1292 (2008).
- Pettinari, M., et al. Are right ventricular risk scores useful. Eur J Cardiothorac Surg. 42 (4), 1-6 (2012).
- Arabia, F. A., Copeland, J. G., Pavie, A., Smith, R. G. Implantation Technique for the CardioWest Total Artificial Heart. Ann Thorac Surg. 68, 698-704 (1999).
- Copeland, J. G., Arabia, F. A., Smith, R. G., Covington, D. Synthetic membrane neopericardium facilitates total artificial heart explantation. J Heart Lung Transplant. 20, 654-656 (2001).
- Crouch, M. A., et al. Successful use and dosing of bivalirudin after temporary total artificial heart implantation: a case series. Pharmacotherapy. 28, 1413-1420 (2008).
- Ensor, C. R., et al. Antithrombotic therapy for the CardioWest temporary total artificial heart. Tex Heart Inst J. 37, 149-158 (2010).
- Stribling, W. K., et al. Use of nesiritide and renal function following total artificial heart implantation. J Heart Lung Transplant. 30, 96 (2011).
- Shah, K. B., et al. Impact of low dose B-type natriuretic peptide infusion on urine output after total artificial heart implantation. J Heart Lung Transplant. 31 (6), 670-672 (2012).
- Mankad, A. K., et al. Persistent anemia in patients supported with the total artificial heart: hemolysis and ineffective erythropoiesis. J Card Fail. 17, 42-42 (2011).
- Kohli, H. S., et al. Exercise blood pressure response during assisted circulatory support: comparison of the total artificial heart with a leftventricular assist device during rehabilitation. J Heart Lung Transplant. 30, 1207-1213 (2011).
- Jaroszewski, D. E., Anderson, E. M., Pierce, C. N., Arabia, F. A. The SynCardia freedom driver: a portable driver for discharge home with the total artificial heart. J Heart Lung Transplant. 30, 844-845 (2011).
- Yankah, L., Shah, K. B., Hess, M. L., Kasirajan, V., Tang, D. G. Response of the Syncardia TAH Pneumatic Drivers to Afterload Challenge on a Mock Loop. ASAIO Journal. 58 (7), (2012).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved