Method Article
Effective Oral RNA Interference (RNAi) Administration to Adult Anopheles gambiae Mosquitoes
In This Article
Summary
The oral administration of dsRNA produced by bacteria, a delivery method for RNA interference (RNAi) that is routinely used in Caenorhabditis elegans, was successfully applied here to adult mosquitoes. Our method allows for robust reverse genetics studies and transmission-blocking vector studies without the use of injection.
Abstract
RNA interference has been a heavily utilized tool for reverse genetic analysis for two decades. In adult mosquitoes, double-stranded RNA (dsRNA) administration has been accomplished primarily via injection, which requires significant time and is not suitable for field applications. To overcome these limitations, here we present a more efficient method for robust activation of RNAi by oral delivery of dsRNA to adult Anopheles gambiae. Long dsRNAs were produced in Escherichia coli strain HT115 (DE3), and a concentrated suspension of heat-killed dsRNA-containing bacteria in 10% sucrose was offered on cotton balls ad-libitum to adult mosquitoes. Cotton balls were replaced every 2 days for the duration of the treatment. Use of this method to target doublesex (a gene involved in sex differentiation) or fork head (which encodes a salivary gland transcription factor) resulted in reduced target gene expression and/or protein immunofluorescence signal, as measured by quantitative Real-Time PCR (qRT-PCR) or fluorescence confocal microscopy, respectively. Defects in salivary gland morphology were also observed. This highly flexible, user-friendly, low-cost, time-efficient method of dsRNA delivery could be broadly applicable to target genes important for insect vector physiology and beyond.
Introduction
Many diseases are transmitted by mosquitoes, making the study of mosquito physiology and genetics an important undertaking. The use of RNAi in these organisms has been prominent in the last 20 years and has allowed for the functional characterization of many mosquito genes1,2,3,4,5. The most commonly used technique for dsRNA delivery has been microinjection, which has the drawbacks that it can injure the mosquitoes and requires significant time and effort. Oral delivery methods for RNAi have been tested, but mainly in the larval stage of the mosquitoes6,7,8,9. Oral delivery of dsRNA in adult mosquitoes has not been fully explored and could be a useful tool for the study of vector biology and vector control.
Malaria is transmitted by Anopheles mosquitoes when an infected female mosquito takes a blood meal from an uninfected host and injects saliva containing malarial parasites10. To ultimately be transmitted in the saliva of a mosquito, the parasite must overcome many hurdles, including evading the mosquito immune system, traversal of the midgut barrier, and invasion of the salivary glands11. Mosquito salivary gland (SG) architecture is key to parasite invasion and that architecture is controlled both by key salivary gland-expressed transcription factors as well as determinants of sexual dimorphism. Several highly conserved transcription factors are required for cellular specification and homeostatic maintenance of the salivary glands and for the production and secretion of salivary proteins that function in blood-feeding12,13,14. Fork head (Fkh) is a winged helix transcription factor that functions as a major regulator of insect SG structure and function (based on studies in fruit flies and the silkworm moth)15,16,17,18,19,20. In the Drosophila SGs, Fkh functions with Sage, an SG-specific basic helix-loop-helix (bHLH) transcription factor, to promote SG survival and saliva production19. An important, positive co-regulator of saliva production in Drosophila is CrebA, a well-studied leucine zipper transcription factor that upregulates the expression of secretory pathway genes21,22,23. There is also a strong degree of morphological differentiation in female salivary glands that likely plays a key role, not only in blood-feeding but also in the ability of parasites to invade this tissue24.
Many of the genes involved in determining salivary gland survival, structure, physiology, and sexual dimorphism have complex spatiotemporal expression profiles25,26,27, and the traditional delivery methods of dsRNA to induce RNAi are not always efficient at targeting these kinds of genes in this or other tissues. However, oral delivery of dsRNA in the larval stage Aedes aegypti and An. gambiae mosquitoes has been used successfully to silence the female-specific form of the dsx gene9,28. Previous studies using dsRNA in mosquito salivary glands found that, although large amounts of dsRNA were required, the silencing effect was relatively long-lasting (at least 13 days)29. Here, the ability of heat-killed E. coli strain HT115 (DE3) expressing sequence-specific dsRNA for dsx, fkh, or CrebA to induce RNAi silencing of these genes in adult female mosquitoes was tested. Oral administration of dsRNA induced gene knockdown in An. gambiae, with clear reductions in mRNA levels and with phenotypes consistent with the loss-of-function of these genes. Thus, this approach will likely work to knock down the function of a variety of salivary gland genes.
Protocol
1. Cloning dsRNA into E. coli expression vector
- Select the target gene sequence to insert into an appropriate vector for the expression of dsRNA. Retrieve the expression values from Vectorbase.org using the following method.
- Search for a gene of interest (e.g., Table 1) on the homepage search box.
- In the resulting gene page, navigate to the 8. Transcriptomics section.
- Look for the listed relevant RNA-seq and microarray gene expression experiments.
- Transcribe values of interest into the spreadsheet software and create a data table.
- Select a commercially available plasmid with at least one T7 promoter to be used. If the selected plasmid has only one T7 promoter (as most commercial plasmids do), include a second T7 promoter in the reverse primer to be used for the amplification of the dsDNA for the gene of interest.
NOTE: The dsRNA sequence for the target genes can be selected using the web application E-RNAi for the design of RNAi reagents30. Either long dsRNA (approximately 400 bp) or short-hairpin dsRNA (shRNA) can be designed based on specific gene sequences. These sequences should be amplified and sequenced for identity confirmation before cloning. The selected gene regions, plasmids, and promoters used in this study are listed in Supplementary File 1. - Perform cloning according to a simple one-step procedure described previously9,31. For this purpose, purify the PCR product and ligate to the linearized plasmid DNA. Use the product of the ligation for the heat-shock transformation of competent E. coli cells32. Select the transformed cells through blue/white screening. Confirm the orientation of the insert using a T7-primer PCR and confirm the sequence using M13 primers.
NOTE: White/blue screenings can be used when the plasmid selected for transformation carries the lacZ gene that codes for β-galactosidase. White colonies should contain the desired insert within the lacZ and can be selected to further confirm the presence and orientation of the target sequence33. - Purify the plasmid from the first transformation and use it to transform competent E. coli HT115 (DE3) as previously described34. After confirmation that the plasmid with the insert is present in the competent E. coli HT115 (DE3), make glycerol stocks of bacteria for single use.
NOTE: An appropriate non-related control dsRNA should be acquired or prepared to use in every experiment. In this case, the sequence for the unrelated gene aintegumenta (ant) from Arabidopsis thaliana is used.
2. Preparation of heat-killed bacteria expressing dsRNA
- Grow a culture from a single bacterial colony of E. coli strain HT115 (DE3) containing the dsRNA expressing plasmid in 50 mL of Luria Broth (LB) containing 100 µg/mL of ampicillin and 12.5 µg/mL of tetracycline, on a platform shaker (180 rpm) at 37 °C for 12 h.
- Dilute the bacterial culture (1:1000) into 2x Yeast Tryptone (2x YT) media containing 100 µg/mL of ampicillin and 12.5 µg/mL of tetracycline.
- Induce dsRNA production by adding 40 µM (final concentration) isopropyl β-D-1-thiogalactopyranoside (IPTG).
- When the cells reach an O.D.600 = 0.4, approximately after 2 h of induction at 37 °C with agitation at 180 rpm, prepare a concentrated suspension of heat-killed bacteria as described by Taracena et al 9. Pellet the cells by centrifugation (4000 x g, 4 °C, 10 min) and wash cells in one volume of sodium phosphate buffer (PBS).
- Spin again under the same conditions, re-suspend in PBS to 1/100 of the initial volume, and place at 70 °C for 1 h.
- Make 400 µL aliquots of the heat-killed bacteria and store these aliquots at -20 °C until further use (do not store for more than a week). This suspension of heat-killed bacteria contains the specific dsRNA for the RNAi experiments. Carry out this procedure both for the target-gene dsRNA-bacteria and for the un-related dsRNA-control to be used in each experiment.
3. Feeding mosquitoes with heat-killed bacteria expressing dsRNA
- Defrost one aliquot of dsRNA (HT115 (DE3) bacteria suspension) and mix with 1.6 mL of 12% sugar solution containing 0.2% methylparaben.
- Soak a small cotton ball in this solution and place the soaked cotton ball inside a cage containing 5-day-old mosquitoes. Ensure that the mosquitoes feed on this solution, picking up both the sugar and the dsRNA-containing bacteria simultaneously.
- Change the cotton ball soaked in dsRNA-sugar solution every other day for 8 consecutive days.
- Keep mosquito cages under constant conditions, i.e., 27 °C and 80% relative humidity with a photoperiod of 12 h:12 h light: dark photocycle, separated by a 30 min dawn and 30 min dusk period.
4. Assay target gene expression levels
- Cold-anesthetize the mosquitoes by placing the container on ice for a min or until the mosquitoes stop moving. Once the mosquitoes are anesthetized, place them on a cold surface to isolate females for dissection.
- Spray 70% ethanol to the mosquitoes and place them on a glass surface with PBS. With a pair of forceps, secure the mosquito head steady and pull the thorax very slowly, allowing the salivary glands to be released into the PBS.
- Keep the salivary glands in ice-cold PBS until 10 individuals have been dissected. Pool Ten SGs for RNA extraction using the guanidinium thiocyanate-phenol-chloroform method. Suspend the RNA pellet in 30 µL of RNase-free water.
- Use 1 µL aliquot of the RNA extracted from the SG in the previous step, to read absorbance at 260 and 280 nm and calculate the RNA concentration of each sample by multiplying with the dilution factor. A 260/280 ratio of ~2.0 indicates good quality RNA.
- Use 1 µg of the purified RNA to synthesize complementary DNA (cDNA) using a commercial reverse transcription kit.
- Make a 1:10 dilution of the cDNA to prepare an RT-PCR reaction according to the manufacturer's recommendations. For each sample, prepare a reaction for the target gene and in parallel, set up a reaction with the housekeeping (HK) gene. Set each gene reaction in a technical triplicate to eliminate the impact of random variation from the method.
NOTE: Here, the An. gambiae ribosomal S7 gene (GeneBank: L20837.1) and actin (VectorBase: AGAP000651) have been used as HK genes. - Use all primers at a final concentration of 300 nM, following the SYBR-green manufacturer's indications. Amplify with standard PCR conditions: 95 °C for 10 min, followed by 40 cycles of 15 s at 95 °C and 60 s at 60 °C.
NOTE: To quantify gene expression, the delta-delta-Ct method (ΔΔCt) is used. Delta Ct (ΔCt) is the difference between the Ct of the target gene and the Ct of the housekeeping gene. ΔΔCt is the difference between the ΔCt of the experimental group and the ΔCt of the control group35.
5. Phenotypic evaluation: successful blood-feeding
- To evaluate the ability to blood-feed, set groups of 15 female mosquitoes treated with target and control dsRNA on small cages (12 cm diameter) and starve them for 4 h.
- Using a circulating water bath set to 37 °C, glass mosquito feeders (24 mm diameter) and parafilm membrane, offer defibrinated sheep blood to the mosquitoes.
NOTE: Blood can be acquired from a commercial vendor who aseptically draws it from healthy, donor animals of U.S. origin and manually defibrinates without anticoagulants or additives. - By direct observation, count and record the number of probing attempts to successfully acquire a blood meal from the first five females to become fully engorged in each group.
NOTE: To avoid significant metabolic changes in the mosquitoes, that could interfere with energy resources impacting blood-seeking behavior, starvation was kept to the minimum (4 h). As a result, not every mosquito would avidly seek the blood-meal and we limited the count of the engorged females to five (a third of each group's total), to reduce the effect of time variables such as exposure to human odor, temperature change between the chambers and the feeding surfaces, etc.
6. Phenotypic evaluation: Salivary gland morphology and down-regulation of relevant proteins
- Isolate fresh tissue in 1x Phosphate-buffered saline (PBS) as described in step 4.2 and fix in ice-cold acetone for 90 s. Rinse several times in 1x PBS after removing the acetone. Incubate with primary antibodies overnight at 4 °C with antiserum (see Table of Materials) diluted into 1x PBS.
NOTE: See Table of Materials for identification of the primary antibodies used for saliva proteins (Anopheles anti-platelet protein, AAPP; Mucin 2, MUC2), SG transcription factors (Fork Head, fkh; Sage, sage; Cyclic-AMP response element-binding protein A, CrebA), and a marker of secretory vesicles (Rab11). These antibodies are used as readouts for SG form and function. However, any antibody suitable for immunofluorescence should be suitable for this protocol. - Wash in 1x PBS several times. Add secondary antibodies (fluorescent) diluted in 1x PBS, and incubate in the dark at room temperature for 2 h. Add any counterstain [such as 4′,6-diamidino-2-phenylindole (DAPI; DNA), wheat germ agglutinin (WGA; for chitin), phalloidin (for F-actin), and/or Nile Red (for lipids)] 30 min before the end of the 2 h incubation.
- Wash three times in 1x PBS. Then, mount the tissues in 100% glycerol on a standard microscope slide with a 1 mm thick coverslip and store at -20 °C until imaging using a fluorescence confocal microscope.
NOTE: To obtain quantitative data, imaging settings must be held constant. Here, only maximum intensity projection images through the entire 3D volume of the tissue were included, and all image quantification was normalized between treatments (within an experiment) based on DAPI signal in non-SG tissue remnants (fat body, cuticle, or head) also present on the slide.
Results
To begin, microarray expression data from VectorBase was used to scan potential targets across developmental stages36,37 to determine the expression status of all genes relevant to the current study (Table 1). As expected, all our chosen target genes showed expression in adult SGs. Levels of aapp and sage were particularly high (Table 1). Also of note were the high levels of expression of f-Agdsx in adult female SGs9.
Specific segments from each gene were evaluated for use as dsRNA using the web application E-RNAi for the design of RNAi reagents30. The ~400 bp regions containing sequences unique to each target gene were then cloned (Figure 1A), transformed into the appropriate bacterial strains, and used to prepare suspensions of heat-killed bacteria, which were induced to produce dsRNA. Adult mosquitoes were fed for 8 days on the sucrose-soaked cotton balls containing the bacterial suspensions of dsRNA for f-Agdsx,fkh, or ant (the unrelated negative control).
For the analysis of RNAi feeding of female mosquitos, it was first determined whether f-Agdsx or fkh dsRNA-feedings induced gene silencing. A 98.8% reduction (±2.1) in fkh transcript levels was observed in the group fed with fkh-dsRNA (Figure 1B), indicating that the dsRNA very effectively reduced the abundance of fkh transcripts in SGs. Surprisingly, fkh mRNA levels were reduced by 82.0% (±18.9) in the mosquitoes treated with dsRNA for f-Agdsx, which had an 89.86% (±4.48) of f-Agdsx reduction, suggesting that fkh could be a target of F-Dsx in the salivary gland. Concomitant with the significant reduction in fkh expression levels, the fkh-knockdown mosquitoes exhibited a significant increase in the number of probing attempts needed to blood-feed. These mosquitoes exhibited, on average, five times more feeding attempts than the control group or f-Agdsx dsRNA fed mosquitoes to be completely engorged with blood (Figure 1C). This led to asking whether the fkh knockdown RNAi treatments caused changes in localization and/or distribution of key transcriptional regulators (SG TFs Sage and CrebA) (Figure 2), secreted proteins (AAPP and mucin) (Figure 3), and secretory machinery [Nile Red (lipids) and Rab11 (secretory vesicles)] (Figure 4). Importantly, substantial differences in staining intensity were observed across different lobe regions, lobes, and individual SGs.
As predicted, levels of sage and CrebA staining were markedly reduced in all SG lobes following fkh RNAi (Figure 2B) compared to ant control RNAi (Figure 2A). Reductions in both the highest maximum intensity values (red dashed lines and numeric labels) and lowest maximum intensity values (blue dashed lines and numeric labels) in line scan profiles suggested reductions in areas of both high and low signal within the tissue (Figures 2A,B). These data suggest that An. gambiae fkh RNAi is effective and that fkh regulates the production and/or stability of the SG TFs Sage and CrebA in An. gambiae, analogous to their genetic relationship in Drosophila SGs19,38,39.
When considering highly abundant saliva-component proteins, levels of Anopheles anti-platelet protein (AAPP)40,41 were reduced in all three SG lobes following fkh RNAi, compared to control RNAi treatment (Figure 3A,B; green). On the other hand, no changes in levels of Mucin were observed (Figure 3A,B; purple). These data suggest that Fkh contributes differently to the expression of different saliva protein genes.
Finally, two markers of secretion were observed (Figures 4A,B): Rab11 (vesicles associated with apical recycling endosomes)42 and Nile Red (lipids). Reduced Rab11 fluorescence was observed in distal lateral (DL) lobes following fkh RNAi treatment (Figure 4A v vs. 4B v; green). However, increased Rab11 signal in the medial (M) and proximal lateral (PL) lobes (Figure 4A vii, ix vs. 4B vii, ix; green) also occurred. No discernible difference was observed in Nile Red signal (Figures 4A,B; purple) after fkh RNAi compared to the control RNAi treatment. These data suggest that fkh reduction may alter some secretory machinery action in a complex manner that differs between SG lobes.
| Dataset: | Goltsev | Neira Oviedo | Neira Oviedo | Baker | Baker | Baker | Baker | ||
| gene symbol | function | AGAP ID | embryo (25 hr.) | L3 larvae | L3 SG | adult female body (3 day) | adult male body (3 day) | adult female SG (3 day) | adult male SG (3 day) |
| AAPP | saliva protein | AGAP009974 | 3.92 | 4.38 | 4.33 | 3.81 | 2.46 | 11.92 | 2.69 |
| CrebA | txn factor | AGAP001464 | 6.28 | 5.22 | 5.92 | 2.99 | 2.96 | 3.27 | 3.13 |
| " | txn factor | AGAP011038 | 4.50 | 4.46 | 5.23 | 2.96 | 2.86 | 3.05 | 2.88 |
| dsx | txn factor | AGAP004050 | 4.91 | 5.39 | 5.55 | 3.72 | 4.00 | 4.57 | 4.01 |
| fkh | txn factor | AGAP001671 | 5.18 | 4.67 | 5.25 | 2.99 | 3.09 | 3.21 | 3.05 |
| MUC2 | saliva protein | AGAP012020 | 4.59 | 5.53 | 5.63 | 2.96 | 3.07 | 3.08 | 3.26 |
| Rab11 | vesicular trafficking | AGAP004559 | 10.21 | 7.47 | 8.60 | 4.90 | 3.79 | 3.38 | 2.96 |
| sage | txn factor | AGAP013335 | 5.32 | 5.96 | 8.89 | 3.40 | 3.33 | 7.37 | 7.23 |
Table 1: Mean log2 microarray expression profiles for An. gambiae genes of interest. Shown are gene names, functional category, Vectorbase (AGAP) identifiers, and mean log2 microarray expression data gathered from Vectorbase. These data indicate that our genes of interest (involved in salivary gland (SG) cell biology and secretion) are expressed and enriched in larval stage 3 (L3) and adult SGs, as compared to whole individuals.
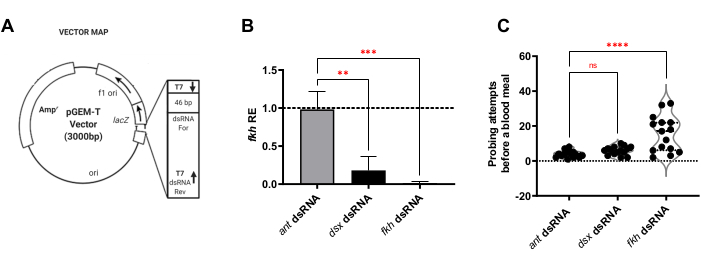
Figure 1: f-Agdsx and fkh knockdown in adult An. gambiae reduces fkh mRNA levels in the SGs and affects the female ability to blood-feed. (A) Representative image of the plasmid design utilized for dsRNA production in this methodology. The second T7 promoter sequence is added to the plasmid by including it in the 3' primer used to amplify the insert to be cloned into the pGEMT plasmid. The plasmid is then transformed into E. coli HT115 (DE3) bacteria and a feeding solution is made of a suspension of induced heat-killed bacteria in 10% sugar water. (B) Animals fed with a dsRNA feeding solution for either f-Agdsx or fkh, showed significantly lower levels of fkh transcripts (one-way ANOVA with multiple comparisons; n=15). However, only the group fed with fkh dsRNA (C) showed a significant difference in the number of biting attempts needed to acquire a blood meal. Mosquitoes in this group needed, on average, five times the number of probing attempts to obtain a successful blood meal than needed by the control or the dsx-dsRNA fed groups (one-way ANOVA with multiple comparisons; n=15). Error bars indicate the Standard Error of the Mean (SEM). Each experiment was conducted in three separate biological replicates. Please click here to view a larger version of this figure.
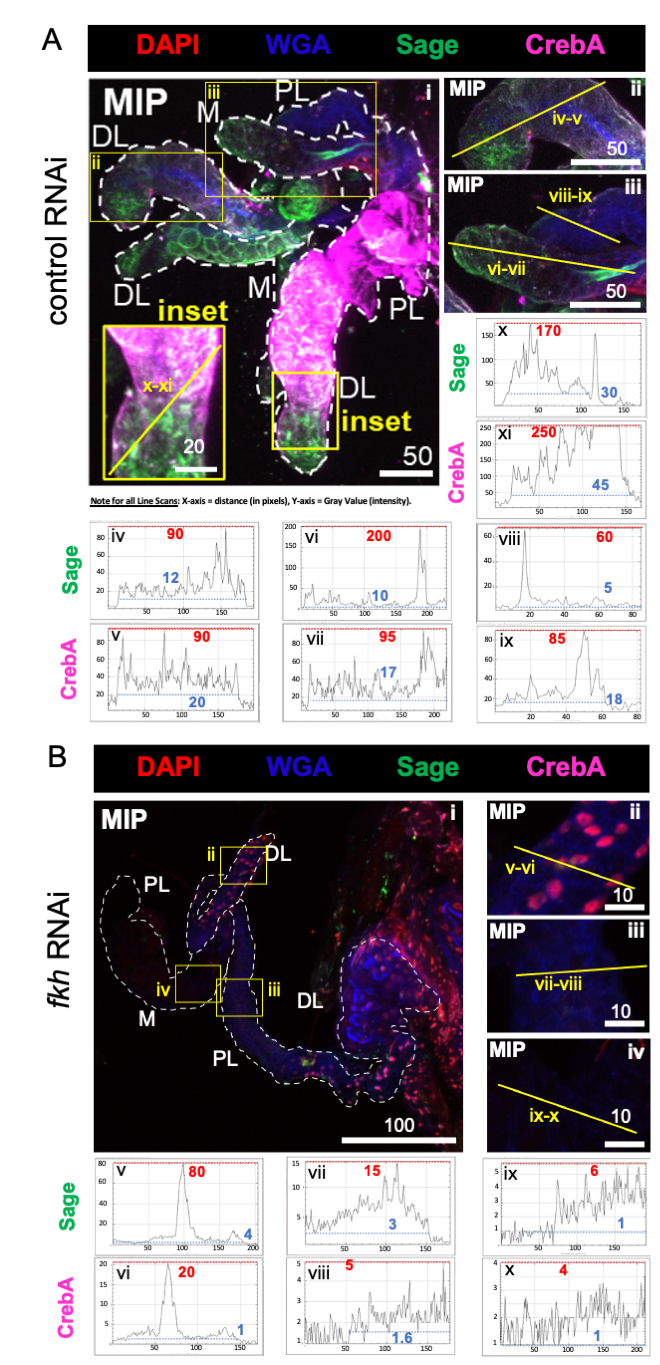
Figure 2: fkh knockdown in adult An. gambiae salivary glands reduces SG transcription factor levels. Shown are representative images from day 13 adult female An. gambiae SGs after 8 days (days 5-13) of oral exposure to either (A) non-related dsRNA control (ant) or (B) dsRNA targeting the SG TF fork head (fkh, AGAP001671) in 10% sucrose stained with the dyes DAPI (DNA; red), labeled wheat germ agglutinin (WGA, chitin/ O-GlcNAcylation; blue), antisera against the SG TFs Sage (green) and CrebA (purple). Scale bar lengths shown are microns. SGs (i) are outlined with white dashes. Yellow lines in zoomed lobe images (of the regions enclosed by yellow boxes, and labeled "inset") indicate where the line scans of signal intensity were conducted. Green and purple channel intensities corresponding to line scans for each zoomed lobe are plotted (always from left to right in the SG) in the graphs below the images; X-axis = distance (in pixels) and Y-axis = gray unit (pixel intensity). The pixel intensity's dynamic range is delimited by red (maximum) and blue (minimum) dotted lines and the corresponding values are shown on each graph. MIP = maximum intensity 3D projection through the entire SG depth. DL: distal lateral lobe; M: medial lobe; PL: proximal lateral lobe; SD: salivary duct. Please click here to view a larger version of this figure.
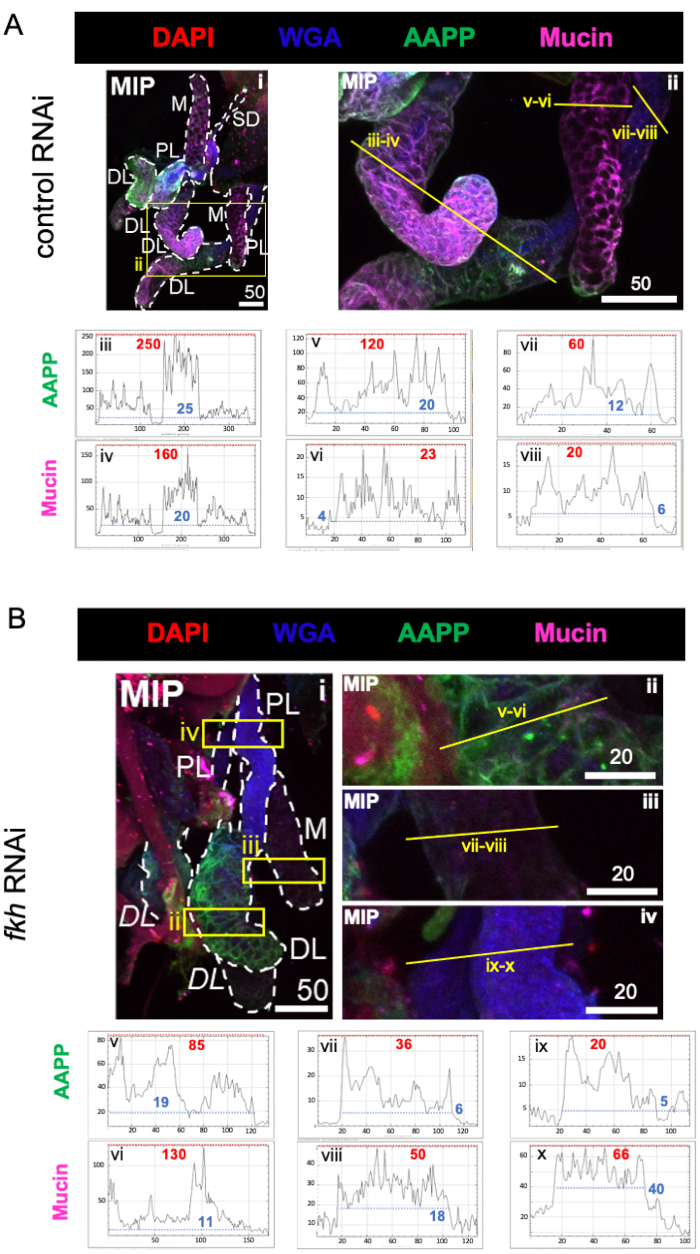
Figure 3: fkh knockdown in adult An. gambiae salivary glands reduces SG secreted protein levels. Shown are representative images from day 13 adult female An. gambiae SGs after 8 days (days 5-13) of oral exposure to either (A) non-related dsRNA control (ant), or (B) dsRNA targeting the SG TF fork head (fkh,AGAP001671) in 10% sucrose stained with the dyes DAPI (DNA; red), labeled wheat germ agglutinin (WGA, chitin/ O-GlcNAcylation; blue), and the saliva proteins AAPP (green) and Mucin (MUC2, purple). Scale bar lengths shown are microns. SGs (i) are outlined with white dashes. Yellow lines in zoomed lobe images (of the regions enclosed by yellow boxes) indicate where the line scans of signal intensity were conducted. Green and purple channel intensities corresponding to line scans for each lobe are plotted (always from left to right in the SG) in the graphs below the images; X-axis = distance (in pixels) and Y-axis = gray unit (pixel intensity). The pixel intensity's dynamic range is delimited by red (maximum) and blue (minimum) dashed lines and the corresponding values are shown on each graph. MIP = maximum intensity 3D projection through the entire SG depth. DL: distal lateral lobe; M: medial lobe; PL: proximal lateral lobe; SD: salivary duct. Italic "DL" labels (Bi) indicate two visible regions of the same DL lobe. Please click here to view a larger version of this figure.
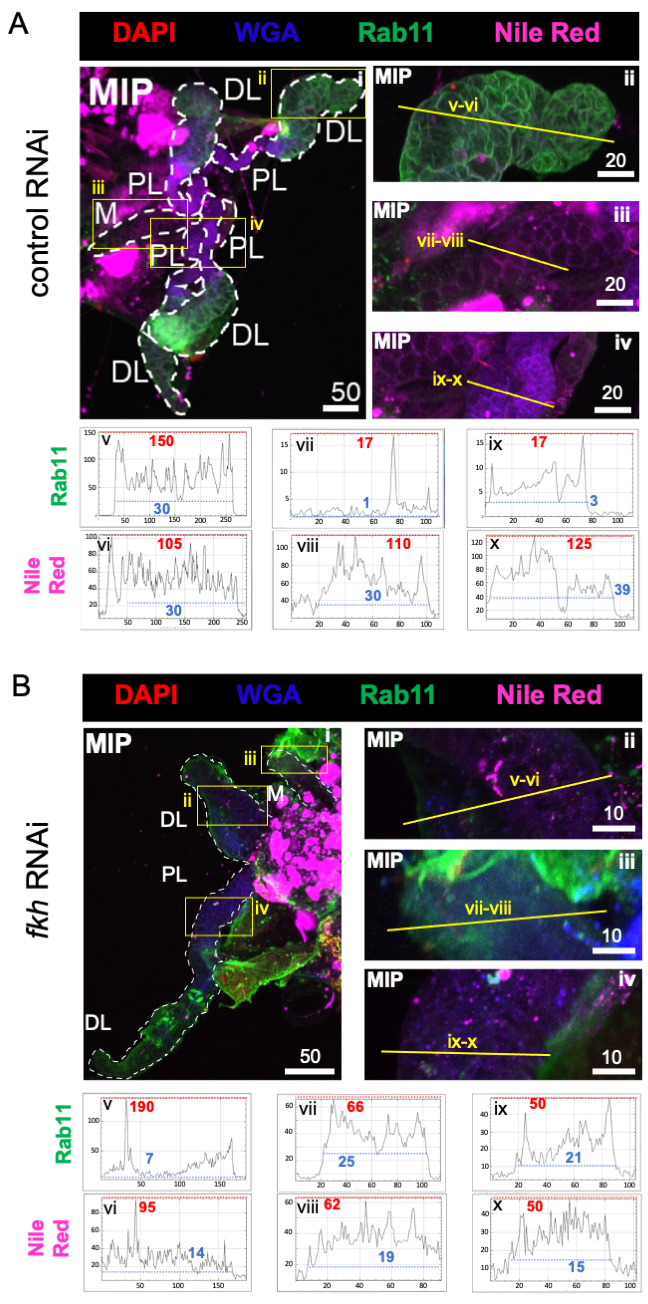
Figure 4: fkh knockdown in adult An. gambiae salivary glands reduces SG secretion markers. Shown are representative images from day 13 adult female An. gambiae SGs after 8 days (days 5-13) of oral exposure to either (A) non-related dsRNA control (ant), or (B) dsRNA targeting the SG TF fork head (fkh, AGAP001671) in 10% sucrose stained with the dyes DAPI (DNA; red), labeled wheat germ agglutinin (WGA, chitin/ O-GlcNAcylation; blue), Nile Red (lipids; purple), and antisera against the recycling endosome vesicle marker Rab11 (green). Scale bar lengths shown are microns. SGs (i) are outlined with white dashes. Yellow lines in zoomed lobe images (of the regions enclosed by yellow boxes) indicate where the line scans of signal intensity were conducted. Green and purple channel intensities corresponding to line scans for each lobe are plotted (always left to right in the SG) in the graphs below the images; X-axis = distance (in pixels) and Y-axis = gray unit (pixel intensity). The pixel intensity's dynamic range is delimited by red (maximum) and blue (minimum) dashed lines and the corresponding values are shown on each graph. MIP = maximum intensity 3D projection through the entire SG depth. DL: distal lateral lobe; M: medial lobe; PL: proximal lateral lobe; SD: salivary duct. Please click here to view a larger version of this figure.
Supplementary File 1. Please click here to download this File.
Discussion
The ability to effectively deliver dsRNA to An. gambiae mosquitoes by oral feeding has broad implications for studies of vector biology both in the laboratory and in the field. Microinjection has long been accepted as the preferred mode of delivery of chemicals, antibodies, RNAi, and genetic modification strategies in mosquitoes43,44. The consequence of substantial physical manipulation, cellular damage, and stress can be avoided by the use of oral delivery, which could also be potentially suitable for large-scale or field applications. Previous work has suggested that RNAi acts ubiquitously within an individual adult mosquito29, allowing for effects in all tissues, including salivary glands. By feeding mosquitoes with large numbers of dsRNA-expressing E. coli that are digested asynchronously over a long timeframe, one can potentially achieve consistent and uniform exposure to the RNAi across all individuals in a cage. This method allows to feed large numbers of mosquitoes and analyze potential variability of the resulting phenotypes depending on the target gene. However, one important consideration is the possibility of heterogeneous distribution of the bacteria, and hence dsRNA, in the cotton fiber. The 400 µL of bacteria used daily for mosquito sugar-feeding would contain approximately ≤4.6 µg of dsRNA, as described and calculated previously9 but the amount of dsRNA ingested by each mosquito was not individually determined. If building dsRNA constructs becomes routine, this simple treatment protocol allows for rapid assimilation of this technique by any mosquito researcher. A priori, the time expenditure during treatment (30 min per day) is trivial compared to the time taken to learn and apply microinjection to similar sample sizes.
Feeding dsRNA is routinely used for reverse genetics studies in the model organism Caenorhabditis elegans45. This heavy level of use underscores the value of the oral delivery approach. Construction of an An. gambiae genome-wide library in transformed E. coli, similar to that which exists in C. elegans46,47, would allow for rapid reverse genetic screening in mosquitoes at an increased scale. However, it is important to note that the efficiency of the method depends in great measure on the endogenous levels of transcript and if the expression is not limited to the target tissue but expressed more broadly4,8,44. Additionally, there is evidence that some insecticides could induce behavioral avoidance from mosquitoes48, and feeding with bacteria that potentially induce adverse effects in them could trigger similar patterns of avoidance. In the controlled setting of the laboratory, where the mosquitoes did not have an alternative food source, they did not have a choice to avoid the sugar water with E. coli and the need for a nutritious source would probably override the instinct to avoid the bacteria. However, this should be considered if the strategy were meant to be used in less controlled settings.
It may be possible to target multiple genes simultaneously (using one construct, multiple constructs, or a mixture of transformed bacterial isolates), but further studies are needed to assess effectiveness. Another important consideration to this point is the evaluation of possible off-target or synergistic effects when using single or multiple targets. The establishment of appropriate control genes and groups is an important part of the experimental design. Further, it is tempting to speculate that this approach could be used to target other pathogens or viruses49. Previous work toward RNAi induction in mosquitoes was performed under conditions where the reagent was directly injected, so E. coli were not present. The E. coli may provide a protective compartment allowing for the slower release of dsRNA over time, ensuring that exposure is more or less continuous over a much longer period29.
Finally, these results show that the effects of this technique are tunable by adjusting the time frame (length and starting day) of exposure and the quantity of E. coli used. This feature allowed us to study the functions of essential genes (dsx and fkh) by identifying optimal knockdown conditions by trial and error. This greatly enhances the likelihood that target genes of interest can be investigated using this technique.
In summary, it was found that oral delivery of RNAi to adult mosquitoes can be simple, versatile, and a powerful approach to studying mosquito gene function and for the creation of novel and malleable tools for vector control of mosquito-borne diseases.
Disclosures
The authors report that they have no conflicts of interest to disclose.
Acknowledgements
The authors wish to thank the staff and scientists within the Entomology Branch and the Division of Parasitic Diseases and Malaria at the CDC, and Brian Trigg and Michelle Chiu for assistance with bacteria preparation at JHU and/or helpful discussions of this work. We thank the JHMRI Insectary and manager Chris Kizito for access to, and rearing of, An. gambiae mosquitoes. We thank Wei Huang (JHSPH) for assistance in obtaining plasmids PJet GFP and pPB47 GFP for use in this study. Funding for this work was provided by: NIH R21AI153588 (to DJA), a Johns Hopkins Malaria Research Institute Postdoctoral Fellowship (to MW); and by a grant from the Good Ventures Foundation and the Open Philanthropy Project to the CDC Foundation entitled Support cryopreservation and suppression of female development in mosquitoes to assist research for malaria, Open Philanthropy Project, 2017. We deeply appreciate assistance from the JHU Microscope Facility staff and applicable NIH grant support for the microscope used (NIH Grant #: S10OD016374). The findings and conclusions in this manuscript are those of the authors and do not necessarily represent the views of the CDC. Use of trade names is for identification only and does not imply endorsement by the Centers for Disease Control and Prevention, the Public Health Service, or the US Department of Health and Human Services.
Materials
| Name | Company | Catalog Number | Comments |
| 1 Kb Plus DNA Ladder | Thermo Fisher Scientific | 10787018 | |
| 2x Yeast Extract Tryptone (2xYT) Medium | BD Difco | DF0440-17 | |
| AAPP | n/a | n/a | Antisera. 1:50 dilution (rabbit); gift from Fabrizio Lombardo |
| AccuStart II PCR Supermix | Quantabio | 95137-100 | |
| Agarose | Millipore Sigma | A9539 | |
| Ampicillin | Millipore Sigma | A5354 | |
| Anopheles gambiae G3 | BioDefense and Emerging Infections (BEI) Malaria Research and Reference Reagent Resource Center (MR4) | MRA-112 | |
| BugDorm | BioQuip | 1452 | |
| Centrifuge 5810R | Eppendorf | P022628181 | |
| CrebA | DSHB | CrebA Rbt-PC | Antisera. 1:50 dilution (rabbit); generated by the Andrew Lab |
| Damiens diet | BioServ | ||
| DAPI | Life Technologies | n/a | 4′,6-diamidino-2-phenylindole; 1:200 dilution. |
| Defibrinated sheep blood | HemoStat | DSB050 | |
| Escherichia coli HT115 (DE3) | |||
| Ethidium bromide | Millipore Sigma | E7637 | |
| High-Capacity cDNA Reverse Transcription Kit | Thermo Fisher Scientific | 4368814 | |
| Isopropyl β-D-1-thiogalactopyranoside | Millipore Sigma | I5502 | |
| JM109 Competent cells | Promega | L2005 | |
| Luria Broth Media | Thermo Fisher Scientific | 10855001 | |
| Mucin 2 | Proteintech | Muc2; 27 675-1-AP | Antisera. 1:100 dilution (mouse). |
| Nanodrop 2000 | Thermo Fisher Scientific | ||
| Nile Red | Sigma | n/a | Lipid dye; 1:50 dilution. |
| Owl EasyCast B2 Mini Gel Horizontal Electrophoresis | Thermo Fisher Scientific | Model B2 | |
| pGEMT easy | Promega | A3600 | |
| Power SYBR-green PCR master MIX | Applied Biosystems | 4367659 | |
| PureLink PCR purification kit | Thermo Fisher Scientific | K31001 | |
| QuantaStudio 6 | Applied Biosystems | ||
| QuantStudio6 Real Time PCR System | Applied Biosystems | ||
| Rab11 | n/a | n/a | Antisera. 1:100 dilution (rabbit); generated by the Andrew Lab |
| Rh-WGA | Vector Labs | n/a | Rhodamine-conjugated wheat germ agglutinin (chitin, O-GlcNAcylation dye); 1:40 dilution |
| Sage | n/a | n/a | Antisera. 1:50 dilution (rat); generated by the Andrew Lab |
| T4 DNA ligase | Promega | M1801 | |
| Tetracycline | Millipore Sigma | 87128 | |
| Trizol | Thermo Fisher Scientific | 15596018 | |
| Zeiss LSM700 fluorescence confocal microscope | Zeiss | ||
| ANTIBODIES | |||
| Chicken anti-Rat IgG (H+L), Alexa Fluor 647 | Thermo Fisher Scientific | A21472 | |
| Goat anti-Mouse IgG (H+L), Alexa Fluor 647 | Thermo Fisher Scientific | A28181 | |
| IgG (H+L) Goat anti-Rabbit, Alexa Fluor 488 | Thermo Fisher Scientific | A27034 | |
| Rabbit anti-Goat IgG (H+L), Alexa Fluor 488 | Thermo Fisher Scientific | A27012 | |
| PRIMERS | |||
| ACT-2f: TACAACTCGATCATGAAGTGCGA | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| ACT-3r: CCCGGGTACATGGTGGTACCGC CGGA | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| FKH_RNAi_F: GCCGACTTATGCTTAGCCCA | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| FKH_RNAi_R: TAGCCGTCAATTCCTCCTGC | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| newDSX-f: AGAGGGCGGGGAAATTCTAGT | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| newDSX-r: GGGCTTGTGGCAGTACGAATA | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| S7qf1: AGAACCAGCAGACCACCATC | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
| S7qr1: GCTGCAAACTTCGGCTATTC | CDC Biotechnology Core Facility Branch | n/a | qRT-PCR primer |
References
- Hoa, N. T., Keene, K. M., Olson, K. E., Zheng, L. Characterization of RNA interference in an Anopheles gambiae cell line. Insect Biochemistry and Molecular Biology. 33, 949-957 (2003).
- Caplen, N., Zheng, Z., Falgout, B., Morgan, R. Inhibition of viral gene expression and replication in mosquito cells by dsRNA-triggered RNA interference | Elsevier enhanced reader. Molecular Therapy. 6, 243-251 (2002).
- Brown, A. E., Catteruccia, F. Toward silencing the burden of malaria: progress and prospects for RNAi-based approaches. BioTechniques. , 38-44 (2006).
- Airs, P. M., Bartholomay, L. C. RNA interference for mosquito and mosquito-borne disease control. Insects. 8, (2017).
- Blandin, S., et al. Reverse genetics in the mosquito Anopheles gambiae: targeted disruption of the Defensin gene. EMBO Reports. 3 (9), 852-856 (2002).
- Garver, L., Dimopoulos, G. Protocol for RNAi assays in adult mosquitoes (A. gambiae). Journal of Visualized Experiments: JoVE. (5), e230 (2007).
- Whyard, S., et al. Silencing the buzz: a new approach to population suppression of mosquitoes by feeding larvae double-stranded RNAs. Parasites & Vectors. 8, 96 (2015).
- Wiltshire, R. M., Duman-Scheel, M. Advances in oral RNAi for disease vector mosquito research and control. Current Opinion in Insect Science. 40, 18-23 (2020).
- Taracena, M. L., Hunt, C. M., Benedict, M. Q., Pennington, P. M., Dotson, E. M. Downregulation of female doublesex expression by oral-mediated RNA interference reduces number and fitness of Anopheles gambiae adult females. Parasites & Vectors. 12, 170 (2019).
- Grassi, B. Studi di uno zoologo sulla malaria. Real Accademia dei Lincei. 3, 229 (1901).
- Smith, R. C., Jacobs-lorena, M. Plasmodium - Mosquito interactions: A tale of roadblocks and detours. Advances in Insect Physiology. 39, (2010).
- Das, S., et al. Transcriptomic and functional analysis of the Anopheles gambiae salivary gland in relation to blood feeding. BMC Genomics. 11, 1-14 (2010).
- Francischetti, I. M. B., Valenzuela, J. G., Pham, V. M., Garfield, M. K., Ribeiro, J. M. C. Toward a catalog for the transcripts and proteins (sialome) from the salivary gland of the malaria vector Anopheles gambiae. Journal of Experimental Biology. 205, 2429-2451 (2002).
- Henderson, K. D., Isaac, D. D., Andrew, D. J. Cell fate specification in thedrosophila salivary gland: The integration of homeotic gene function with the DPP signaling cascade. Developmental Biology. 205, 10-21 (1999).
- Mach, V., Ohno, K., Kokubo, H., Suzuki, Y. The Drosophila fork head factor directly controls larval salivary gland-specific expression of the glue protein gene Sgs3. Nucleic Acids Research. 24 (12), 2387-2394 (1996).
- Weiserova, M., et al. Mini-Mu transposition of bacterial genes on the transmissible plasmid. Folia Microbiologica. 32 (5), 368-375 (1987).
- Abrams, E. W., Mihoulides, W. K., Andrew, D. J. Fork head and Sage maintain a uniform and patent salivary gland lumen through regulation of two downstream target genes, PH4αSG1 and PH4αSG2. Development. 133, 3517-3527 (2006).
- Myat, M. M., Isaac, P. P., Andrew, D. J. Early genes required for salivary gland fate determination and morphogenesis in Drosophila melanogaster. Advances in Dental Research. 14, 89-98 (2000).
- Fox, R. M., Vaishnavi, A., Maruyama, R., Andrew, D. J. Organ-specific gene expression: the bHLH protein Sage provides tissue specificity to Drosophila FoxA. Development of Cell Biology. 140, 2160-2171 (2013).
- Maruyama, R., Grevengoed, E., Stempniewicz, P., Andrew, D. J. Genome-wide analysis reveals a major role in cell fate maintenance and an unexpected role in endoreduplication for the Drosophila FoxA gene fork head. PLOS ONE. 6, 20901 (2011).
- Johnson, D. M., et al. CrebA increases secretory capacity through direct transcriptional regulation of the secretory machinery, a subset of secretory cargo, and other key regulators. Traffic. 21, 560-577 (2020).
- Fox, R. M., Hanlon, C. D., Andrew, D. J. The CrebA/Creb3-like transcription factors are major and direct regulators of secretory capacity. Journal of Cell Biology. 191, 479-492 (2010).
- Abrams, E. W., Andrew, D. J. CrebA regulates secretory activity in the Drosophila salivary gland and epidermis. Development. 132, 2743-2758 (2005).
- Wells, M. B., Andrew, D. J. Anopheles salivary gland architecture shapes plasmodium sporozoite availability for transmission. mBio. 10 (4), 01238 (2019).
- Pei-Wen, L., Xiao-Cong, L., Jin-Bao, G., Yan, L., Xiao-Guang, C. Molecular cloning, characterization and expression analysis of sex determiantion gene doublesex from Anopheles gambiae (Diptera: Culicidae). Acta Entomologica Sinica. 58 (2), 122-131 (2015).
- Scali, C., Catteruccia, F., Li, Q., Crisanti, A. Identification of sex-specific transcripts of the Anopheles gambiae doublesex gene. The Journal of Experimental Biology. 208, 3701-3709 (2005).
- Price, D. C., Egizi, A., Fonseca, D. M. Characterization of the doublesex gene within the Culex pipiens complex suggests regulatory plasticity at the base of the mosquito sex determination cascade. BMC Evolutionary Biology. 15, 1-13 (2015).
- Mysore, K., et al. siRNA-mediated silencing of doublesex during female development of the dengue vector mosquito Aedes aegypti. PLoS Neglected Tropical Diseases. 9, 1-21 (2015).
- Boisson, B., et al. Gene silencing in mosquito salivary glands by RNAi. FEBS Letters. 580, 1988-1992 (2006).
- Horn, T., Boutros, M. E-RNAi: a web application for the multi-species design of RNAi reagents-2010 update. Nucleic Acids Research. 38, 332-339 (2010).
- Taracena, M. L., et al. Genetically modifying the insect gut microbiota to control chagas disease vectors through systemic RNAi. PLoS Neglected Tropical Diseases. 9, (2015).
- Sambrook, J., Fritsch, E. F., Maniatis, T. . Molecular Cloning: A Laboratory Manual. , (1989).
- Ullmann, A., Jacob, F., Monod, J. Characterization by in vitro complementation of a peptide corresponding to an operator-proximal segment of the β-galactosidase structural gene of Escherichia coli. Journal of Molecular Biology. 24, 339-343 (1967).
- Timmons, L. Bacteria-mediated RNAi-General outline. Carnegie Institution of Washington. , (2000).
- Pfaffl, M. W. Relative quantification. Real-time PCR. , 63-82 (2004).
- Neira-Oviedo, M., et al. The RNA-Seq approach to studying the expression of mosquito mitochondrial genes. Insect Molecular Biology. 20, 141-152 (2011).
- Baker, D. A., et al. A comprehensive gene expression atlas of sex- and tissue-specificity in the malaria vector, Anopheles gambiae. BMC Genomics. 12, (2011).
- Loganathan, R., Hoon, J., Wells, M. B., Andrew, D. J. Secrets of secretion - How studies of the Drosophila salivary gland have informed our understanding of the cellular networks underlying secretory organ form and function. Cellular Networks in Development. , 143 (2021).
- Chung, S., Hanlon, C. D., Andrew, D. J. Building and specializing epithelial tubular organs: The Drosophila salivary gland as a model system for revealing how epithelial organs are specified, form and specialize. Wiley Interdisciplinary Reviews: Developmental Biology. 3, 281-300 (2014).
- Yoshida, S., et al. Inhibition of collagen-induced platelet aggregation by anopheline antiplatelet protein, a saliva protein from a malaria vector mosquito. Blood. 111, 2007-2014 (2008).
- Wells, M. B., Villamor, J., Andrew, D. J. Salivary gland maturation and duct formation in the African malaria mosquito Anopheles gambiae. Scientific Reports. 7 (1), 601 (2017).
- Takahashi, S., et al. Rab11 regulates exocytosis of recycling vesicles at the plasma membrane. Journal of Cell Science. 125, 4049-4057 (2012).
- Catteruccia, F., Levashina, E. A. RNAi in the malaria vector, Anopheles gambiae. Methods in Molecular Biology. 555, 63-75 (2009).
- Balakrishna Pillai, A., et al. RNA interference in mosquito: understanding immune responses, double-stranded RNA delivery systems and potential applications in vector control. Insect Molecular Biology. 26, 127-139 (2017).
- Fire, A., et al. Potent and specific genetic interference by double-stranded RNA in caenorhabditis elegans. Nature. 391, 806-811 (1998).
- Kamath, R. S., Ahringer, J. Genome-wide RNAi screening in Caenorhabditis elegans. Methods. 30, 313-321 (2003).
- Kamath, R. S., et al. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature. 421, 231-237 (2003).
- Carrasco, D., et al. Behavioural adaptations of mosquito vectors to insecticide control. Current Opinion in Insect Science. 34, 48-54 (2019).
- Magalhaes, T., et al. Induction of RNA interference to block Zika virus replication and transmission in the mosquito Aedes aegypti. Insect Biochemistry and Molecular Biology. , 111 (2019).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved