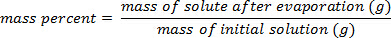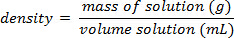Déterminer la concentration massique d'une solution aqueuse
Vue d'ensemble
Source : Laboratoire du Dr Neal Abrams — SUNY College of Environmental Science and Forestry
Détermination de la composition d’une solution est une technique analytique et médecine légale importante. Lorsque des solutions sont apportées avec de l’eau, ils sont dénommés est aqueuse, ou contenant de l’eau. La principale composante d’une solution est dénommée le solvant et la composante mineure dissoute est appelée le soluté. Le soluté est dissoute dans le solvant pour faire une solution. L’eau est le solvant plus courant dans la vie quotidienne, ainsi que presque tous les systèmes biologiques. Dans les laboratoires de chimie, le solvant peut être un autre liquide, tels que l’acétone, l’éther ou un alcool. Le soluté peut être un liquide ou un solide, mais cette expérience seule privéeLa détermination des solides.
Procédure
1. le pourcentage en masse - Direct
- Placez un petit volume d’une solution dans un plat propre et séché au four bécher ou cristallisation.
- Après avoir déterminer avec précision la masse totale exacte de la solution, faire chauffer le bécher ou plat sur une plaque chauffante ou dans un four pour chasser l’eau. Évaporation lente est la meilleure méthode, comme ébullition peut entraîner des éclaboussures de la solution.
- Une fois que le solvant s’est évaporée, refroidir le reste solide (soluté)
Résultats
À l’aide de l’exemple illustré dans la Figure 1, un ensemble de normes de chlorure de sodium a été préparé avec masse pourcentage compositions de 5,000 % 10,00 %, 15,00 %, 20.00 % et 25,00 % de soluté dans la solution. Les densités mesurées étaient de 1,025 1,042, 1.060, 1,070 et 1,090 g/mL, respectivement. Après traçage de ces données, une courbe de tendance linéaire est appliqué, montage de l’équation y = 3.446 x 10-3x + 1.0048, où y est la mass...
Applications et Résumé
Le pourcentage de sucre dans la soude, pourrait facilement être determinedusing le principe de composition masse pour cent. La procédure pour faire cette expérience serait de mesurer la masse et le volume de soude dégazé (pas de bulles) et de calculer la densité de la solution. Une courbe d’étalonnage de la masse volumique vs % massique pour plusieurs solutionswould standard de saccharose (sucre) doivent être créées, et puis l’étalonnage pourrait servir à résoudre pour le pourcentage de saccharose dans la soude. Une hypothè...
Passer à...
Vidéos de cette collection:

Now Playing
Déterminer la concentration massique d'une solution aqueuse
General Chemistry
383.5K Vues

Présentation et utilisation de la verrerie de laboratoire courante
General Chemistry
656.1K Vues

Solutions et Concentrations
General Chemistry
274.2K Vues

Déterminer la densité d'un solide et d'un liquide
General Chemistry
556.1K Vues

Déterminer la formule empirique
General Chemistry
181.9K Vues

Détermination des règles de solubilité des composés ioniques
General Chemistry
141.4K Vues

Utiliser un pH-mètre
General Chemistry
345.9K Vues

Introduction au titrage
General Chemistry
424.5K Vues

Loi des gaz parfaits
General Chemistry
78.5K Vues

Détermination spectrophotométrique d'une constante d'équilibre
General Chemistry
158.5K Vues

Principe de Le Châtelier
General Chemistry
265.2K Vues

Identification d'un composé inconnu grâce à la loi de la cryométrie
General Chemistry
160.7K Vues

Détermination de la loi de vitesse et de l'ordre de la réaction
General Chemistry
196.1K Vues

Utilisation de la calorimétrie différentielle à balayage pour mesurer les changements d'enthalpie
General Chemistry
44.5K Vues

Complexes de coordination
General Chemistry
91.5K Vues