מתחמי כימיה של תיאום
Overview
מקור: המעבדה של ד"ר ניל אברמס — מכללת SUNY למדעי הסביבה ויערנות
מתכות מעבר נמצאות בכל מקום, החל תוספי ויטמינים לאמבטיות electroplating. מתכות מעבר גם מרכיבות את הפיגמנטים בצבעים רבים ומרכיבות את כל המינרלים. בדרך כלל, מתכות מעבר נמצאות בצורה הקטיקטית מכיוון שהן מתחמצןות בקלות, או מאבדות אלקטרונים, ומוקפות בתורמי אלקטרונים הנקראים ליגנדים. ליגנדים אלה אינם יוצרים קשרים יוניים או קוולנטיים עם מרכז המתכת, אלא הם לוקחים על סוג שלישי של קשר המכונה קואורדינטות קוולנט. הקשר הקואורדינט-קוולנטי בין ליגנד למתכת הוא דינמי, כלומר ליגנדים מחליפים ומתאמים מחדש ללא הרף סביב מרכז המתכת. הזהויות של המתכת והליגנד מכתיבות אילו ליגנדים יקשרו באופן מועדף על פני אחר. בנוסף, תכונות צבע ומגנטיות נובעים גם מסוגי המתחמים שנוצרים. תרכובות התיאום שנוצרות מנותחות באמצעות מגוון מכשירים וכלים. ניסוי זה בוחן מדוע כל כך הרבה מתחמים אפשריים ומשתמש בשיטה ספקטרוכימית (צבע וכימי) כדי לסייע בזיהוי סוג קומפלקס הקואורדינציה שנוצר.
Procedure
1. מתחמי ניקל וצבעים
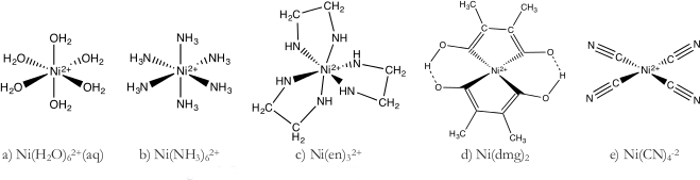
- Ni(H2O)62+ קומפלקס(איור 1a)
- הכן פתרון 1 M של Ni(H2O)62+ על ידי המסת NiSO4 בנפח המתאים של מים.
- לדלל עוד יותר את הפתרון Ni(H2O)62+על ידי הוספת 70 מ"ל של פתרון 1 M ל 1,000 מ"ל של מים deionized.
- חלק את Ni(H2O)62 + בין שבעה 400 מ"ל כות.
- תמיסה ניקל מימי לוקח על צבע ירוק בהיר מאז מים הוא ליגנד שדה חל
Application and Summary
מפיגמנטים ועד אנשים, מתכות מעבר נמצאות בתחומי הכימיה, הביולוגיה, הגיאולוגיה וההנדסה. הבנת ההתנהגות של מתכות מעבר תחת מצבים כימיים שונים יכולה להיות פשוטה כמו ניטור צבע או התנהגות מגנטית. כמעט כל מתכת מעברתלת-ממדית (שורה 4) חיונית לתפקוד הפיזיולוגי, ובכל המקרים, מתכות אלה כבולות על ידי...
References
- Shakhashiri, B. Z.; G. E. Dirreen, G. E; Juergens, F. Color, Solubility, and Complex Ion Equilibria of Nickel (II) Species in Aqueous Solution. J. Chem. Ed. 52 (12), 900-901 (1980).
Tags
Skip to...
Videos from this collection:

Now Playing
מתחמי כימיה של תיאום
General Chemistry
91.7K Views

כלי זכוכית ושימושים נפוצים במעבדה
General Chemistry
658.3K Views

פתרונות וריכוזים
General Chemistry
275.1K Views

קביעת הצפיפות של מוצק ונוזל
General Chemistry
556.8K Views

קביעת הרכב אחוז המסה בפתרון מימי
General Chemistry
383.8K Views

קביעת הנוסחה האמפירית
General Chemistry
183.7K Views

קביעת כללי המסיסות של תרכובות יוניות
General Chemistry
141.6K Views

שימוש במד pH
General Chemistry
346.7K Views

מבוא לתמצית
General Chemistry
425.4K Views

חוק הגז האידיאלי
General Chemistry
79.0K Views

קביעת ספקטרופוטומטריה של קבוע שיווי משקל
General Chemistry
158.7K Views

עקרון לה שאטלייה
General Chemistry
265.8K Views

דיכאון נקודת הקפאה כדי לקבוע תרכובת לא ידועה
General Chemistry
160.8K Views

קביעת חוקי התעריפים וסדר התגובה
General Chemistry
196.3K Views

שימוש בסריקה דיפרנציאלית קלורימטריה למדידת שינויים באנטלפיה
General Chemistry
44.7K Views
Copyright © 2025 MyJoVE Corporation. All rights reserved