Gascromatografia con rivelatore a ionizzazione di fiamma
Panoramica
Fonte: Laboratorio del Dr.B. Jill Venton - Università della Virginia
La gascromatografia (GC) viene utilizzata per separare e rilevare composti di piccolo peso molecolare nella fase gassosa. Il campione è un gas o un liquido che viene vaporizzato nella porta di iniezione. Tipicamente, i composti analizzati sono meno di 1.000 Da, perché è difficile vaporizzare composti più grandi. GC è popolare per il monitoraggio ambientale e le applicazioni industriali perché è molto affidabile e può essere eseguito quasi continuamente. GC è tipicamente utilizzato in applicazioni in cui vengono rilevate piccole molecole volatili e con soluzioni non acquose. La cromatografia liquida è più popolare per le misurazioni in campioni acquosi e può essere utilizzata per studiare molecole più grandi, perché le molecole non hanno bisogno di vaporizzare. GC è favorito per le molecole non polari mentre LC è più comune per separare gli analiti polari.
La fase mobile per la gascromatografia è un gas vettore, tipicamente elio a causa del suo basso peso molecolare e del suo essere chimicamente inerte. Viene applicata la pressione e la fase mobile sposta l'analita attraverso la colonna. La separazione avviene utilizzando una colonna rivestita con una fase stazionaria. Le colonne capillari tubolari aperte sono le colonne più popolari e hanno la fase stazionaria rivestita sulle pareti del capillare. Le fasi stazionarie sono spesso derivate del polidimetilsilossano, con il 5-10% dei gruppi funzionalizzati per sintonizzare la separazione. I gruppi funzionali tipici sono i gruppi fenile, cianopropile o trifluoropropilico. Le colonne capillari sono solitamente lunghe 5-50 m. Le colonne più strette hanno una risoluzione più elevata ma richiedono pressioni più elevate. Le colonne imballate possono essere utilizzate anche dove la fase stazionaria è rivestita su perle imballate nella colonna. Le colonne imballate sono più corte, 1-5 m. I capillari tubolari aperti sono generalmente preferiti perché consentono maggiori efficienze, analisi più veloci e hanno capacità più elevate.
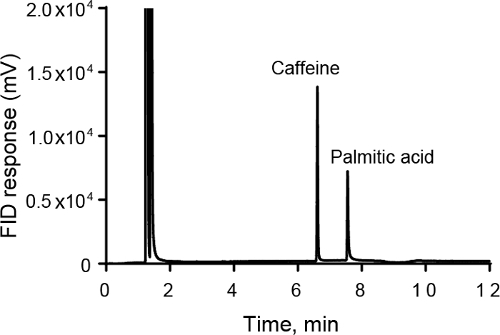
Il rilevamento della ionizzazione di fiamma (FID) è un buon rivelatore generale per composti organici in GC che rileva la quantità di carbonio in un campione. Dopo la colonna, i campioni vengono bruciati in una fiamma calda idrogeno-aria. Gli ioni carbonio sono prodotti dalla combustione. Mentre l'efficienza complessiva del processo è bassa (solo 1 su 105 ioni carbonio produce uno ione nella fiamma) la quantità totale di ioni è direttamente proporzionale alla quantità di carbonio nel campione. Gli elettrodi sono usati per misurare la corrente dagli ioni. Il FID è un rilevatore distruttivo, poiché l'intero campione è pirolizzato. Il FID non è influenzato da gas e acqua non combustibili.
Procedura
1. Inizializzazione del CG
- Accendere il gas e l'aria di trasporto dell'elio e regolare i manometri sullo strumento.
- Accendere il forno a colonna ad alta temperatura (in genere 250 °C o superiore) per cuocere nella colonna. Non superare la temperatura massima della colonna. Questo rimuoverà eventuali contaminanti. Lasciare cuocere per almeno 30 minuti prima di eseguire un campione.
2. Creazione di un file di metodi
- Nel software che controlla lo strumento, ins
Applicazione e Riepilogo
GC è utilizzato per una varietà di applicazioni industriali. Ad esempio, viene utilizzato per testare la purezza di un prodotto chimico sintetizzato. GC è anche popolare nelle applicazioni ambientali. GC viene utilizzato per rilevare pesticidi, idrocarburi poliaromatici e ftalati. La maggior parte delle applicazioni di qualità dell'aria utilizza GC-FID per monitorare gli inquinanti ambientali. GC viene anche utilizzato per l'analisi dello spazio di testa, in cui vengono raccolti e misurati i volatili evaporati da un ...
Tags
Vai a...
Video da questa raccolta:

Now Playing
Gascromatografia con rivelatore a ionizzazione di fiamma
Analytical Chemistry
282.0K Visualizzazioni

Preparazione del campione per la caratterizzazione analitica
Analytical Chemistry
84.7K Visualizzazioni

Standard interni
Analytical Chemistry
204.8K Visualizzazioni

Metodo delle aggiunte standard
Analytical Chemistry
320.1K Visualizzazioni

Curve di calibrazione
Analytical Chemistry
796.9K Visualizzazioni

Spettroscopia ultravioletta/visibile (UV-VIs)
Analytical Chemistry
623.5K Visualizzazioni

Spettroscopia Raman per analisi chimiche
Analytical Chemistry
51.2K Visualizzazioni

Fluorescenza a raggi X (XRF)
Analytical Chemistry
25.4K Visualizzazioni

Cromatografia liquida ad alta prestazione (HPLC)
Analytical Chemistry
384.5K Visualizzazioni

Cromatografia a scambio ionico
Analytical Chemistry
264.6K Visualizzazioni

Elettroforesi capillare
Analytical Chemistry
93.9K Visualizzazioni

Introduzione alla spettrometria di massa
Analytical Chemistry
112.3K Visualizzazioni

Microscopia elettronica a scansione (SEM)
Analytical Chemistry
87.2K Visualizzazioni

Misurazioni elettrochimiche di catalizzatori supportati mediante l'utilizzo di un potenziometro/galvanometro
Analytical Chemistry
51.4K Visualizzazioni

Voltammetria ciclica
Analytical Chemistry
125.2K Visualizzazioni