Curve di calibrazione
Panoramica
Fonte: Laboratorio del Dr.B. Jill Venton - Università della Virginia
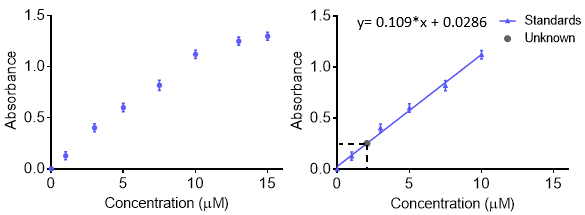
Le curve di calibrazione vengono utilizzate per comprendere la risposta strumentale a un analita e prevedere la concentrazione in un campione sconosciuto. Generalmente, una serie di campioni standard viene effettuata a varie concentrazioni con un intervallo che include l'incognita di interesse e viene registrata la risposta strumentale a ciascuna concentrazione. Per una maggiore precisione e per comprendere l'errore, la risposta ad ogni concentrazione può essere ripetuta in modo da ottenere una barra di errore. I dati sono quindi dotati di una funzione in modo da poter prevedere concentrazioni sconosciute. In genere la risposta è lineare, tuttavia, una curva può essere fatta con altre funzioni purché la funzione sia nota. La curva di calibrazione può essere utilizzata per calcolare il limite di rilevazione e il limite di quantificazione.
Quando si realizzano soluzioni per una curva di calibrazione, ogni soluzione può essere realizzata separatamente. Tuttavia, ciò può richiedere molto materiale di partenza e richiedere molto tempo. Un altro metodo per fare molte concentrazioni diverse di una soluzione è quello di utilizzare diluizioni seriali. Con le diluizioni seriali, un campione concentrato viene diluito in modo graduale per produrre concentrazioni più basse. Il campione successivo viene ricavato dalla diluizione precedente e il fattore di diluizione viene spesso mantenuto costante. Il vantaggio è che è necessaria una sola soluzione iniziale. Lo svantaggio è che eventuali errori nella creazione di soluzioni - pipettaggio, massa, ecc. - vengono propagati man mano che vengono apportate più soluzioni. Pertanto, è necessario prestare attenzione quando si effettua la soluzione iniziale.
Procedura
1. Fare gli standard: diluizioni seriali
- Crea una soluzione stock concentrata dello standard. Tipicamente, il composto viene accuratamente pesato e quindi trasferito quantitativamente in un pallone volumetrico. Aggiungere un po 'di solvente, mescolare in modo che il campione si dissolva, quindi riempire fino alla linea con il solvente appropriato.
- Eseguire diluizioni seriali. Prendere un altro matraccio volumetrico e pipettare la quantità di standard necessaria per la diluizione, quindi riempire fino
Applicazione e Riepilogo
Le curve di calibrazione sono utilizzate in molti campi della chimica analitica, della biochimica e della chimica farmaceutica. È comune usarli con spettroscopia, cromatografia e misurazioni elettrochimiche. Una curva di calibrazione può essere utilizzata per comprendere la concentrazione di un inquinante ambientale in un campione di suolo. Potrebbe essere usato per determinare la concentrazione di un neurotrasmettitore in un campione di fluido cerebrale, vitamina in campioni farmaceutici o caffeina negli alimenti. Per..
Vai a...
Video da questa raccolta:

Now Playing
Curve di calibrazione
Analytical Chemistry
797.8K Visualizzazioni

Preparazione del campione per la caratterizzazione analitica
Analytical Chemistry
84.9K Visualizzazioni

Standard interni
Analytical Chemistry
205.0K Visualizzazioni

Metodo delle aggiunte standard
Analytical Chemistry
320.4K Visualizzazioni

Spettroscopia ultravioletta/visibile (UV-VIs)
Analytical Chemistry
624.4K Visualizzazioni

Spettroscopia Raman per analisi chimiche
Analytical Chemistry
51.3K Visualizzazioni

Fluorescenza a raggi X (XRF)
Analytical Chemistry
25.6K Visualizzazioni

Gascromatografia con rivelatore a ionizzazione di fiamma
Analytical Chemistry
282.5K Visualizzazioni

Cromatografia liquida ad alta prestazione (HPLC)
Analytical Chemistry
385.3K Visualizzazioni

Cromatografia a scambio ionico
Analytical Chemistry
264.7K Visualizzazioni

Elettroforesi capillare
Analytical Chemistry
94.2K Visualizzazioni

Introduzione alla spettrometria di massa
Analytical Chemistry
112.5K Visualizzazioni

Microscopia elettronica a scansione (SEM)
Analytical Chemistry
87.3K Visualizzazioni

Misurazioni elettrochimiche di catalizzatori supportati mediante l'utilizzo di un potenziometro/galvanometro
Analytical Chemistry
51.5K Visualizzazioni

Voltammetria ciclica
Analytical Chemistry
125.5K Visualizzazioni