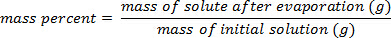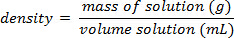Determining the Mass Percent Composition in an Aqueous Solution
Source: Laboratory of Dr. Neal Abrams — SUNY College of Environmental Science and Forestry
Determining the composition of a solution is an important analytical and forensic technique. When solutions are made with water, they are referred to as being aqueous, or containing water. The primary component of a solution is referred to as the solvent, and the dissolved minor component is called the solute. The solute is dissolved in the solvent to make a solution. Water is the most common solvent in everyday life, as well as nearly all biological systems. In chemistry labs, the solvent may be another liquid, like acetone, ether, or an alcohol. The solute can be a liquid or a solid, but this experiment only addressesthe determination of solids.
1. Percent by Mass - Direct
- Place a small volume of a solution into a clean and oven-dried beaker or crystallization dish.
- After accurately determining the precise total mass of the solution, heat the beaker or dish on a hotplate or in an oven to drive off the water. Slow evaporation is the best method, as boiling can result in splattering of the solution.
- Once the solvent has evaporated, cool the remaining solid (solute) and determine the mass.
- Calculate the mass percent as:
Using the example shown in Figure 1, a set of sodium chloride standards was prepared with mass percent compositions of 5.000%, 10.00%, 15.00%, 20.00%, and 25.00% of solute in solution. The measured densities were 1.025, 1.042, 1.060, 1.070, and 1.090 g/mL, respectively. After plotting these data, a linear trendline is applied, fitting the equation y = 3.446 x 10-3x + 1.0048, where y is the density and x is the mass percent composition.
Next, the volume o
The percentage of sugar in soda, could easily be determinedusing the principle of mass percent composition. The procedure for doing this experiment would be to measure the mass and volume of degassed soda (no bubbles) and to calculate the solution's density. A calibration curve of density vs. percent by mass for several standard sucrose (sugar) solutionswould need to be created, and then that calibration could be used to solve for the percent of sucrose in the soda. One assumption is that sucrose is the major contributor to a change in d
Pular para...
Vídeos desta coleção:

Now Playing
Determining the Mass Percent Composition in an Aqueous Solution
General Chemistry
382.7K Visualizações

Vidraria de laboratório comuns e seus usos
General Chemistry
651.2K Visualizações

Soluções e Concentrações
General Chemistry
271.8K Visualizações

Determinando a densidade de um sólido e um líquido
General Chemistry
553.0K Visualizações

Determinação da Fórmula Empírica
General Chemistry
178.1K Visualizações

Determinação das Regras de Solubilidade de Compostos Iônicos
General Chemistry
140.8K Visualizações

Usando um medidor de pH
General Chemistry
343.3K Visualizações

Introdução à Titulação
General Chemistry
422.0K Visualizações

Lei dos gases ideais
General Chemistry
77.5K Visualizações

Determinação espectrofotométrica de uma constante de equilíbrio
General Chemistry
158.0K Visualizações

Princípio de Le Châtelier
General Chemistry
262.9K Visualizações

Depressão do ponto de congelamento para determinar um composto desconhecido
General Chemistry
159.4K Visualizações

Determinação das Leis de Velocidade e da Ordem de Reação
General Chemistry
195.4K Visualizações

Uso de Calorimetria de Varredura Diferencial para Medir Mudanças na Entalpia
General Chemistry
44.3K Visualizações

Complexos de Química de Coordenação
General Chemistry
90.6K Visualizações
Copyright © 2025 MyJoVE Corporation. Todos os direitos reservados